Q: None
A: Step 1:Part (a): Step 2:Part (b):Step 3:Part (c):Step 4:Part (d):
Q: For an ideal gas, calculate the following quantities. (a) The pressure of the gas if 0.155 mol…
A: The ideal gas law equation is:PV = nRTwhere:P is the pressure of the gas .V is the volume of the gas…
Q: d) Calculate the molar solubility of Fe(OH)3 in a solution of 0.0125 M NaOH.
A: ### 1. Write the Dissolution EquationIron(III) hydroxide, Fe(OH)3, dissociates in water according to…
Q: If an electron has an uncertainty in its velocity of 2.30 m/s, what is the uncertainty (in meters)…
A:
Q: Question 10 A photon is quantum of electromagnetic radiation. A True B False
A:
Q: Select any and all Diels-Alder endo products from this set of molecules. Choose one or more: H H H…
A: In endo product the electron withdrawing group from the dienophile is directed towards the newly…
Q: F NO2 + NaOCH2CH3
A: Given reaction:Sodium ethoxide (NaOCH2CH3) with p-fluoro nitrobenzene (1-fluoro-4-nitrobenzene)We…
Q: Which of the following pairs of substances can NOT be used to make a buffer solution? a. HF & KOH…
A: Buffer systems are used in chemical systems to maintain the pH of the system constant. They are…
Q: A gas system has initial volume and temperature of 8160mL and 467.0K. If thetemperature changes…
A:
Q: None
A: Ans part 1 Given Mass of UO2 = 2.26 kg= 2260 g Write the balanced equation UO2 (g) + 4 HF (g) →…
Q: Fix this for me
A:
Q: 1. Convert ethyne to 2,3-pentanedione (a compound responsible for the aroma of cereal coffee brew).…
A: Step 1: Step 2: Step 3: Step 4:
Q: Q5. Predict the major product of the following reaction. MeO CO₂Me 1) NaOH, H₂O at 50 °C 2) H3O+ B…
A: The nucleophile OH- attacks the carbonyl carbon to form a tetrahedral intermediate and finally, the…
Q: What is the molar solubility of NiS in 0.40 M NH3? The Ksp for NiS is 3.0 × 10-19. The K₁ for…
A: Two equations can be represented as; NiS ⇌ Ni2+ + S2- ...KspNi2+ + 6NH3 ---->…
Q: 7. Draw the product of the following reaction explain if there is any stereochemistry. NaOH -HBr Br
A: Step 1: Step 2:In the 1st step there is formation of 3° carbocation by elimination of Br- as leaving…
Q: Please don't provide handwriting solution
A: The objective of the question is to identify the correct statements that describe the energy…
Q: sample size
A: The resulting polymer will be a random copolymer with a blocky characteristic. Chemical composition…
Q: Write a balanced half-reaction for the oxidation of bismuth ion (Bi³+) to bismuth oxide ion…
A: Half-reaction is a part of the overall reaction representing either an oxidation or a reduction…
Q: How is primary hypocortisolism (Addison disease) differentiated from secondary hypocortisolism…
A: The objective of the question is to differentiate between primary and secondary hypocortisolism,…
Q: d. Give the mechanism for the reaction between 1,3 cyclohexadione and chlorine in the presence of…
A: The objective of this question is to understand the mechanism of the reaction between 1,3…
Q: 14. Why does the rate of the reaction decrease over time? A. Exothermic reactions lose heat which…
A: Write the general rate law form. The order with respect to each reactant is not known.For the…
Q: dont provide handwriting solution ...
A: The objective of this question is to calculate the change in internal energy (ΔU) of the reaction…
Q: Modify the structure of the compound provided to show the skeletal structure of the product that…
A: Popoff’s rule: According to this rule oxidation of an unsymmetrical ketone takes place in such a way…
Q: For the following reaction, select the major product below. A) B Oc OO D B) хох Хл NaOCH3 CH3OH ахл…
A:
Q: 6. What is the overall order of the following reaction, given the rate law? NO(g) + O(g) -> NO2(g) +…
A: Answer:Power of concentration term of a reactant in the rate law is equal to the order of reaction…
Q: Draw the major organic product for the reaction shown. Do not draw counterions or byproducts. Draw…
A: Step 1: Step 2: Step 3: Step 4:
Q: Given that ΔHvap = 40.66 kJ/mol for water at 100 °C, calculate Kb of H2O.
A: Enthalpy of vaporization (ΔHvap) for water = 40.66 kJ/molTemperature (T) = 100 °CConverting to…
Q: Please provide appropriate reagents to achieve the conversion of B into α,ẞ-unsaturated ester C over…
A: Given is organic synthesis reaction.Here, we need to find out reagents for conversion of B to C.
Q: 3. Based on the pH results obtained in this experiment, complete the following equation for the…
A: Step 1:
Q: Indicate the units of the reaction rate for the kinetics of order 0, 1, 2 and 3. Are they the same?
A: The order of the reaction indicates the relationship between the concentration of the reactant and…
Q: 15.18 Determine the number and symmetry designations of the infrared-active C-O stretching modes in…
A: Infrared (IR) spectroscopy is useful for determining the stretching modes of chemical bonds,…
Q: Consult the table below and answer the questions that follow. The table below shows the equilibrium…
A: Le Chatelier's principle suggests that when a system at equilibrium is disturbed by changing factors…
Q: Choose the best reagents to complete the following reaction. Br2 A excess Br2, NaOH B 0 CH3CO2H D OH…
A: When methyl ketone is treated with excess halogen and base a reaction occurs leading to the…
Q: dont provide handwriting solution...
A: Step 1:- When , we have a solution of CuSO4 , Copper(II) sulfate (CuSO4) dissociates completely in…
Q: Draw a detailed mechanism for the Grignard reaction performed in this experiment, starting from…
A:
Q: Add explaintion each step
A: Let's go through each step with explanations:Alanine (Ala, A): Alanine is a nonpolar amino acid,…
Q: Select the correct arrow-pushing mechanism for the formation of the more stable carbocation…
A: Alkenes are nucleophiles. The π bond is localized above and below the C−C σ bond. The double bond…
Q: What quantity in moles of NaOH need to be added to 200.0 mL of a 0.200 M solution of HOCl (Ka = 3.5…
A: 1. **Understand the problem and what we're trying to achieve:** - We have a solution of HOCl, a…
Q: dont provide handwriting solution ....
A: The objective of the question is to find the concentration of OH- ions in a basic solution given the…
Q: Fix this for me
A: Step 1: Step 2: Step 3: Step 4:
Q: Please explain mechanism. G.
A: Step 1: Step 2: Step 3: Step 4:
Q: A neutral compound has a partition coefficient of 5 5 between ether and water. What percentage of…
A: The objective of the question is to determine the percentage of a neutral compound that would be…
Q: oul on 1. NaOEt Br✓ Br 2. H3O+, heat
A: Given an active methylene compound, base , a dihalide, Daw the major product.
Q: 2. A buffer solution contains 0.15 mol of propionic acid (HC3H5O2) and 0.10 mol of sodium propionate…
A: Calculate the new concentration of the conjugate:= 0.10 mol + 0.01 mol = 0.11 mol Calculate the new…
Q: A buffer solution contains 0.329 M NaH2PO4 and 0.324 M Na2HPO4. Determine the pH change when 0.080…
A: The chemical reactions involved are:1. H2PO4^- <-> H^+ + HPO4^2-2. HCl -> H^+ + Cl^-When…
Q: energy heat + H3CO A B reaction coordinate
A: The objective of the question is find out the mechanism of both the product formation and mention…
Q: D
A: The objective of the question is to identify the correct terms that can be used to describe the…
Q: None
A: The Diels-Alder reaction involves cycloaddition of a conjugated diene (a molecule containing two…
Q: None
A: Thank You.
Q: A solution has [H+] = 4.2 x 103 M. The [OH] in this solution is O 4.2 x 10-11 M O 3.7 x 10-13 M O…
A: Answer:Product of hydrogen ion concentration and hydroxide ion concentration is always equal to the…
dont provide handwriting solution ....
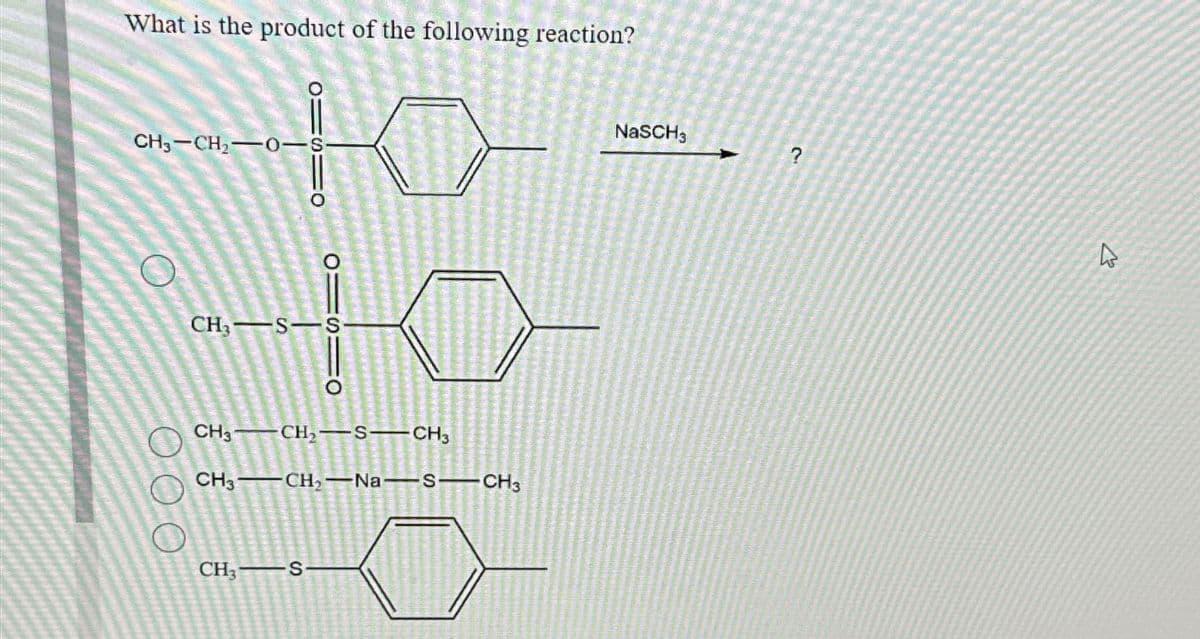
Step by step
Solved in 2 steps with 1 images

- What are the products of the following reactions? a.C5H11Cl + OCH3 b.C4H9O- + CH3-XDraw the products of the following reactions: a. CH3OCH3 + BF3 b. CH3OCH3 + H Cl c. CH3NH2 + AlCl3What reagent is necessary to complete the reaction? CH3-CH2-CH-C-OH ? CHỊCH,CH C-0- Na* CH3 CH3 NaO O NaCl O Na O NaOH + H₂O
- Draw the products formed when (CH3)2C=CH2 is treated with each reagent.a. HBrb. H2OH2SO4c. CH3CH2OH, H2SO4d. Cl2e. Br2, H2Of. NBS (aqueous DMSO)g. [1]BH3;[2]H2O2, HO-What is the product of the reaction P4O6 + H2O? A) H3PO4 B) H2PO4 C) H3PO3 D) H2PO3 E) H2P4O7In an SN1 reaction, how many rearranged carbocations will be formed from CH3-CHCl-CH=CH-CH3? 4 3 2 1 0
- Bicyclo[2.2.1]heptan-7-one + PCC (in CH₂Cl₂) => A.) Bicyclo[2.2.1]heptan-7-ol B.) Bicyclo[2.2.1]heptanoic acid C.) Cyclohexanecarbaldehyde D.) All the given choices are possible products E.) No reactionChoose the best pair of reactants to form the following product by an SN2 reaction. (CH3)2CHOCH2CH2CH3The stereochemistry of the products of reduction depends on the reagent used, with this in mind, how would you convert 3,3-dimethylbutan-2-one [CH3COC(CH3)3] to: (a) racemic 3,3-dimethylbutan-2-ol [CH3CH(OH)C(CH3)3]; (b) only (R)-3,3- dimethylbutan-2-ol; (c) only (S)-3,3-dimethylbutan-2-ol?
- What is the major product(s) of the following reaction: (R)-2-chlorobutane, only (S)-2-chlorobutane, only 1-chlorobutane, only (R)-1-chlorobutane, only (S)-3-chlorobutane, only NdaDraw the product of each of the following Sn2 reactions: a) Chloropentane and NaSH b) (R)-3-lodohexane and NaClWhat is the product of the SN2 reaction of (R) 3-bromo-2-methylhexane with CH3CO- Select one: a. (S) 2-methyl-3-ethoxy-hexane b. (S) 2-methyl-2-ethoxy-hexane c. (S) 3-methyl-2-ethoxy-hexane d. (S) 2-methy-3-hexanol e. (R) 2-methy-3-hexanol



