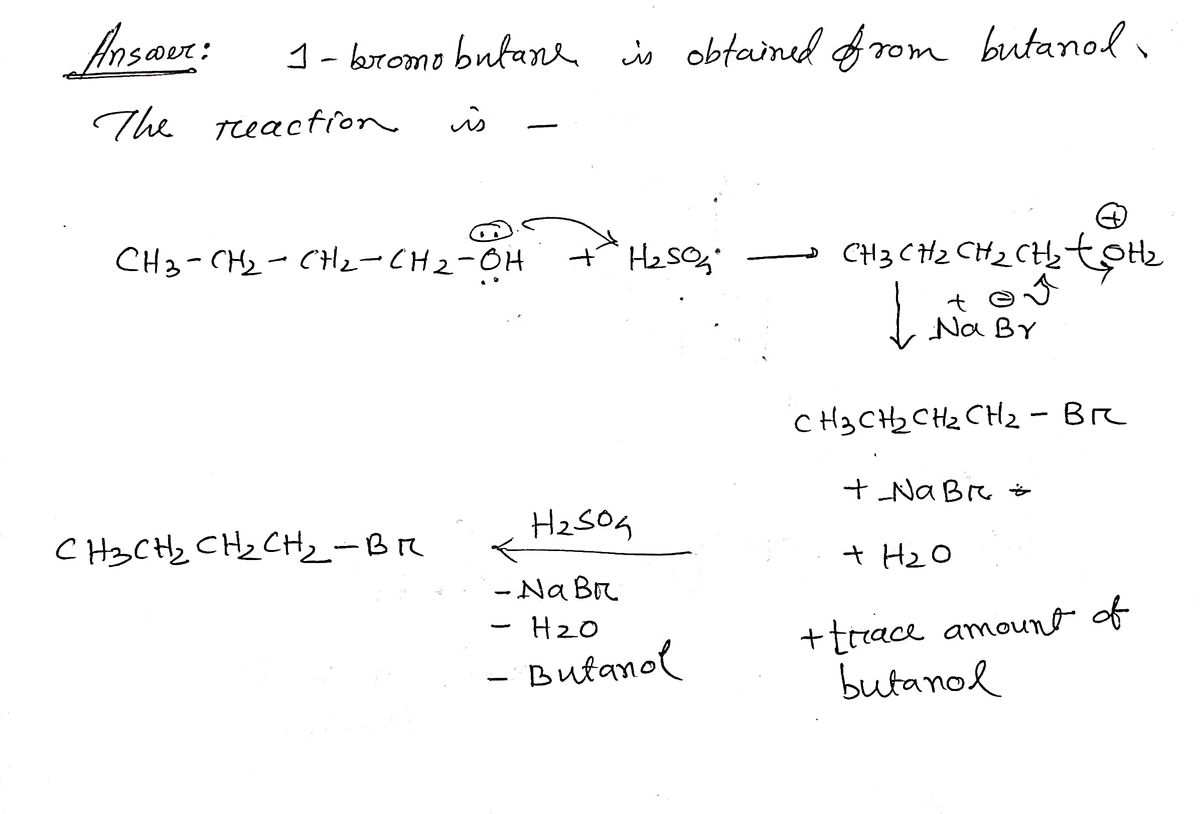
what is the purpose of adding concentrated sulfuric acid to the product, 1-bromobutane after distillation? Write the chemical equation to illustrate the role of sulfuric acid.
Q: Consider the table below Refer to image What is the molecular shape of the following molecules?…
A: we have to determine the molecular shape of the given molecules
Q: Potassium ferrocyanide (K4Fe(CN)6) is used in the tempering of steel and in process engraving. It is…
A: No. Of moles of Compound = Mass of Compound/ Molar mass of compound Molar mass of Potassium…
Q: 0.700 mol Na₂SO STARTING AMOUNT 0.700 mol Na₂SO ADD FACTOR x( ) 0.700 0.0833 mL Convert the…
A: Conversion factor - When we have to convert one unit to another unit we must use conversion factor.…
Q: Determine the number of atoms of O in 57.7 moles of Al₂(CO₃)₃.
A: Given number of moles of Al2(CO3)3 = 57.7 mole Number of oxygen atom =?
Q: In a laboratory experiment, students synthesized a new compound and found that when 11.52 grams of…
A:
Q: The molecular weight of glucose is 180.156 g/mol. If you wish to administer 205.00 grams of glucose…
A:
Q: Using IUPAC guidelines, name the following organic compound HHHH |||| H-C-C-C-C-H AAAA Answer:…
A: IUPAC nomenclature: In IUPAC nomenclature a molecule's longest chain of carbons is taken as parent…
Q: Knowing the following at a certain temperature, 2 NaHCO₃ (s) ⇄ Na₂CO₃ (s) + H₂O (g) + CO₂ (g)…
A:
Q: How many mL of 0.600 M LiCl would be required to make a 0.0855 M solution of LiCl when diluted to…
A:
Q: The following endothermic reaction is allowed to reach equilibrium. NH4HS(s) NH3(g) + H₂S(g) a)…
A:
Q: What volume of 3.0M acetic acid will form from the reactian of calcium acetate with 125mL of 1.0M hy…
A: Answer: This question is based on stoichiometric calculation where by using stoichiometric…
Q: Pentane (C5H12) and hexane (C6H14) form an ideal solution. At 25°C the vapor pressures of pentane…
A: (b) Given that, vapour pressure of the pentane at 25oC is 511 torr. A solution is prepared by mixing…
Q: Two heat capacity values are given in the task. Why are two different heat capacities needed?
A: Heat capacity is the amount of heat to be supplied to a substance to increase its temperature by 1…
Q: 0.30h+0.50(15-h)=6.30
A: Given : equation
Q: Draw the 2D formula for the following condensed formulas
A:
Q: Humidity is the percentage of partial pressure of water in air over the total possible water in the…
A: Introduction Humidity is an important concept in chemistry. It refers to the amount of water vapor…
Q: 15. Which of the compounds in each reaction will act as the acid and which as the base? Draw the…
A:
Q: -ОН О
A:
Q: How many grams of NH 3 are in 2.5 mol of NH3 3?
A:
Q: Dust falls on Chicago at a rate of 65 mg m-2 day-1. Major metallic elements in the dust include…
A: Given data is Rate of fall of dust on Chicago=65 mg m-2 day-1 Rate of accumulation of Lead=0.029 mg…
Q: Given the information A+B C 2D D calculate AG at 298 K for the reaction A + B 2C ᎪG° = AH = -641.9…
A: Given : reaction and thermodynamic data
Q: The equation P=1+(d)/(33) gives the pressure, P, in atmospheres (atm ) at a dopth of d feot in the…
A: The problem is based on general chemistry. The pressure value determines the corresponding depth…
Q: Consider the following system at equilibrium where K = 0.159 and AH = -111 kJ/mol at 723 K. N₂ (9) +…
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on…
Q: * 4. CH₂ Circle the pairs of organic solids which can be separated from each other by solvent…
A: Solvent extraction: It is a method of separating compounds based on their relative solubilities in…
Q: Calculate the kilojoules released when 90.0 g of steam condenses at 100 °C, the liquid cools to 0…
A: Given data, mass of steam = 90.0 g Temperature at which it condenses = 100 °C Temperature till it…
Q: a. CH3CH=CHCH3 c. CH3CH2CH3 b. d. Н. ΗΙ Н H на н -С-СЕС-С Н HA 'H H нн 6-bromo-4-methylhex-2-yne C-t…
A: We will determine bond line formula for each of the formula given in the question.
Q: No Plagiarism Please! In one to two sentences, describe the process by which the ionic compound,…
A: Lithium chloride is a chemical compound with a chemical formula “LiCl”. The salt is a normal ionic…
Q: Please use two Friedel-Crafts acylation reactions to produce:
A: Friedel-Crafts acylation reaction: Benzene gives an aromatic electrophilic substitution reaction.…
Q: Write the electron configuration of the following atoms and identify what Family and period do they…
A: The electronic configuration and the class of the elements are given below
Q: 4 PH3(g)→→P4(g) + 6 H₂(g) the following data have been obtained: [PH3], M time, s 8.86x10-2…
A:
Q: How many different ways can 4 identical items be put into 6 possible spaces? How about 4 identical…
A: The number of ways to put "n" identical items into "k" possible spaces can be calculated using the…
Q: Aromatic compounds are classified as saturated. Select one: O True O False Aromatic compounds are…
A: Answer: Aromatic compounds have obtained their name from the word aroma that means pleasant smell…
Q: 1.72. Calculate the average grade using equation 1.32 if the scores on a 10-point quiz are 5, 5, 5,…
A: Since, Average of the given sample or data is calculated as sum of all the data divided by the…
Q: In order to prepare a buffer of pH 4.60, you start with 500.0 mL of 0.100 M benzoic acid (C6H5COOH)…
A: 1 mole benzoic acid react with 1 mole NaOH. Henderson equation for buffer solution, pH = pKa + log…
Q: The freezing point of water is 0.00 °C at 1 atmosphere. How many grams of zinc chloride (136.3…
A:
Q: Draw the structures of the following Alkynes: 2,5-Dimethyl-3-hexyne Ethyne 1-Propyne…
A: Alkynes are a class of hydrocarbons that contain a carbon-carbon triple bond. The basic formula for…
Q: Select which of the following hydrocarbons would exhibit stronger London Dispersion Forces. Select…
A: London Dispersion forces are the weak forces present in the non polar molecules. This forces…
Q: A chemistry graduate student is studying the rate of this reaction: 2NH, (g)-N₂ (g) + 3H₂(g) He…
A: We have find out the rate law and rate constant of the given reaction.
Q: 4. A certain gas has a volume of 2.50 L and a pressure of 3.00 atm at a constant temperature. a. If…
A:
Q: D. Give the quantum numbers for the last electron of each element given in Part II following the…
A:
Q: Name each of the following NO₂ A OH
A: For naming following steps are required First of all identify the longest carbon chain containing…
Q: The percent yield of this reaction is 78.0%. 2Al(s) + 3 F2 (g) →→→ 2AlF3 (s) Determine the mass, in…
A:
Q: 7. Using the bond dissociation energies provided below, draw an energy diagram for the two…
A: Suppose a reaction A2 + B2 –> C2 Then ΔHrxn given as ΔHrxn = Σ Bond energy of reactants -Σbond…
Q: How many atoms of Ca are in 2.00 mol of Ca? Show your work. Be sure each step is written out…
A:
Q: The nonvolatile, nonelectrolyte aspirin, C₂H8O4 (180.1 g/mol), is soluble in diethyl ether,…
A:
Q: 2a) The molecule ethene (or ethylene), which has the molecular formula C₂H4, contains two carbon…
A: In the lewis structure of ethene, there is a double bond between carbon atoms, four C-H bonds. There…
Q: What mass of H₂ gas is produced during the electrolysis of water if a current of 12.7 A is used for…
A: Given, 2H+(aq) + 2e- ----> H2(g) Current (I) = 12.7 A Note: 1 min = 60 s Time (t) = 30 min = 30…
Q: Difluoroethene has two isomers with very different properties despite having the same chemical…
A: Given, Enthalpy of vaporization (∆Hvap) = 21.4 kJ/mol = 21400 J/mol Note: vapour pressure of isomer…
Q: Consider an aqueous solution containing 1.0 g/L of a protein (molar mass = 7.5 x 104 g/mol) with a…
A: Answer: Properties of the solution those depend on the number of solute particles present in the…
Q: Match each of the elements on the left to an elemental form on the right. Xe Cu N C(fullerenes) Ge…
A:
what is the purpose of adding concentrated sulfuric acid to the product, 1-bromobutane after distillation? Write the chemical equation to illustrate the role of sulfuric acid.
Step by step
Solved in 2 steps with 2 images

- List the advantages and constraints of reactive distillation by explaining methyl acetate production.give at least three characteristics of dichloromethane that make it a good extracting solvent for alkaloidHow do you expect the composition of cyclohexane and toluene to change over the course of a distillation?
- Write equations for the preparation of n-pentane from:i) Write the chemical equation including reagent(s) and condition(s) for preparation of 2-pentyne from ethyne.ii) Determine the chemical equation for hydration of butyne including all reagent(s),condition(s) and intermediate(s).why is fractional distillation a better method of separating 1-chlorobutane and toluene than simple distillation
- Discuss the “green” implications of using dichloromethane (methylene chloride) as your primary extraction solvent. Consider its hazards and toxicities, process for manufacture, cost, efficiency of extraction, ease of recovery and reuse, and cost of waste disposal.Why is it bad to put too much water as the solvent when recrystillizing benzoic acid?1. What type of reaction explains the solubility of phenol in Sodium Hydroxide? Explain the organic product formed 2. How do the reactions of phenol samples with FeCl3 compare? Which structural component of the phenols account for the observation? 3. What compound is the precipitate formed in the Bromine water test? 4. Write the reaction formed in the formation of phenolphthalein. Identify the functional group in phenolphthalein , which is responsible for the indicator property. 5. What is the significance of Millon's test?
- What is the purpose of Chromic acid test? a. What are the reagents used? b. Write oxidation reaction of Primary Alcohols and Secondary AlcoholsWrite the reactions involved in synthesizing Benzanol1-Bromopropane is synthesized by treating 1-propanol with sulfuric acid (H2SO4) and potassium bromide (KBr) through a simple distillation. What is the inorganic by-product left in the reaction flask following the distillation?

