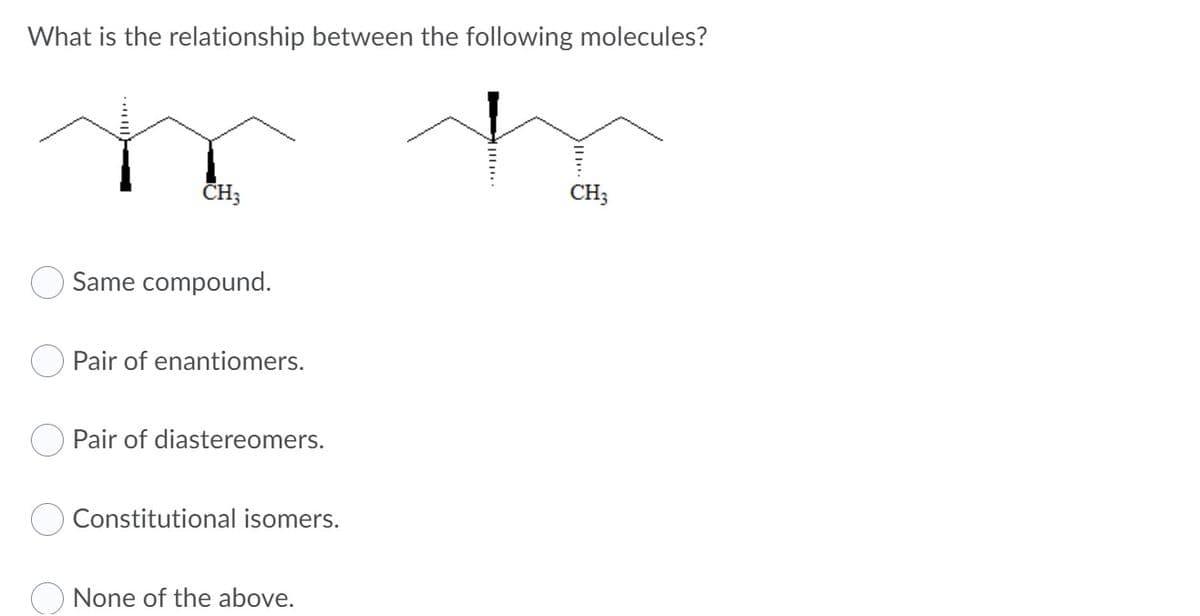
What is the relationship between the following molecules? CH; CH; Same compound. Pair of enantiomers. Pair of diastereomers. Constitutional isomers. None of the above.
Q: Any enantiomers, stereocenters, diastereomers or meso compounds for dibutylcyclohexane?
A: Since position of butyl group is not mentioned, hence 1,1 position is being considered. The…
Q: What is the name for a 50:50 mixture of enantiomers such as a 50:50 mixture of (S)- -utanol and…
A: a 50:50 mixture of enantiomers such as 50:50 mixture of (S)-2-butanol and (R)-2-butanol are called…
Q: How are these molecules related? OH CH3 CH3 ZOH and A. conformational isomers В. C. enantiomers…
A: Isomerisms: Two compounds having similar chemical formula but have different structures are known as…
Q: For Tylenol (Acetaminophen), what is the primary and secondary carbon? Any enantiomers? (R and S)
A: The primary, secondary and tertiary nature of carbon atom depends on the number of substituents…
Q: The molecules shown below are A) Identical molecules B) Stereoisomers C) Constitutional isomers D)…
A: ->Cis ,trans isomers are stereoisomer.
Q: Draw three-dimensional representations of the following compounds. Which have asymmetric carbon…
A: Given: 1-chloro-3-methylbuta-1,2-diene
Q: What is relationship between the two structures below? CI O they are enantiomers O they are…
A:
Q: Q6.7 B and D CH3 CH H;C, CH CH3 H3C H3C" CH3 CH3 CH3 H;C "CH, H3C D E F What is the relationship…
A: Given molecules:
Q: CH3 H3C JCH3 CH3 H3C CH3 ICH3
A: Given structures,
Q: Q1:- Which of the isomeric alcohols having formula CSH120 are chiral? Which are achiral? Draw the…
A: The question is based on the concept of isomers. molecules having same molecular formula but…
Q: 6. Enantiomers, diastereomers or same molecule? CH3 City Br -H Br CH3 CH3 City H- Br
A: Enantiomers : They are one type of stereoisomers having mirror image relationship. Enantiomers are…
Q: How many degrees of unsaturation does 3,4-dicyclopropyl-1-heptyne have? 1 2 3 4
A: The index of hydrogen deficiency or degree of unsaturation means the determination of shortage of…
Q: What is the relationship? Identical, constitutional isomers, enantiomers, or diastereomers?
A:
Q: Which of the following is the enantiomer of the given compound? HO CHS OH H. CH3 E locA CH2Br H. H.…
A: Two enentiomers have opposite absolute configuration
Q: Mark each chiral center in the following molecule with an asterisk. How many stereoisomers are…
A: Chiral center is the one which is attached to four different substituents and knowing the number of…
Q: H,C „CH3 H,SO,, H,0 CH3 The two major products are a) enantiomers b) diastereomers c) the same…
A: Hydration of double bond occurs via carbocation intermediate formation. Most stable intermediate…
Q: For 2-chloro-3-fluorocyclohexane, draw all possible stereoisomers, and identify their stereochemical…
A: It is given that 2-chloro-3-fluorocyclohexane stereoisomers needs to be drawn, and needs to be…
Q: N-
A:
Q: Indicate whether each of the structures in the second row is an enantiomer of, is a diastereomer of,…
A: Stereoisomers are isomers which have different spatial arrangement in spite of same bond…
Q: X- enantiomers?
A: Enantiomers: These are the chiral molecules which are mirror image to each other and they are non…
Q: Which statement about these Fischer projections is correct? CH3 CH3 CH3 CH, Но -H- Но -- H- OH H- Но…
A: The correct fisher projection is as follows
Q: Which Fischer projection below ( A,B,C,D, or E) represents the D stereoisomer for 2-butanol? А. В.…
A: Detail description is given below
Q: Draw three-dimensional representations of the following compounds. Which have asymmetric carbon…
A: Given, 1-chlorobuta-1,3-diene
Q: 4-Chloro-2-pentene has a double bond that can have either the E or the Z configuration and a…
A: Isomers that are formed by the spatial arrangement of atoms are known as stereoisomers.…
Q: Br Br CH3
A: The given compound has three chiral centers. Thus total eight (2n = 23 = 8) stereoisomer is…
Q: Indicate whether the pair of structures shown represent stereoisomers, constitutional isomers,…
A: Given : two different structures of one compound. To find : type of conformations or isomers…
Q: 5) For each of the pairs of molecules drawn below, place letters in the corner boxes corresponding…
A: The isomers which have the same connectivity of atoms in the molecules but different spatial…
Q: Label them based on the type of stereoisomerism they exhibit in. OPTICAL. NEITHER OPTICAL OR…
A: Chiral carbon: a carbon having 4 different atoms or groups attache to it. A molecule with chiral…
Q: II
A: The compound with the same molecular formula but different structures are known as an isomer.…
Q: Consider the pair of compounds shown below then select the word or phrase that best describes the…
A: For enantiomers the configuration should be RR and SS. For distereoisomer the configuration may be…
Q: 2 Identify the following pairs of compounds as identical or isomers. If they are isomers, are they…
A: Stereoisomer - is the same structural formula but different arrangements. Enantiomer- the all…
Q: Draw the diastereomer of a trans-1,2-dimethylcyclopentane
A: Diastereomers are the stereoisomers that are not mirror images and non superimposible. Diastereomers…
Q: KMNO4 hot
A:
Q: Do the following structures represent identical compounds or a pair of enantiomers?
A:
Q: Of the isomers shown, which are chiral? Which ones are constitutional isomers of each other?…
A:
Q: Hexane and 3-methylpentane A) B) C) D) None of these are examples of: enantiomers. stereoisomers.…
A: Ans. D) constitutional isomers. Explanation: Constitutional or structural isomers are the…
Q: H3C 2 HO CH3 CH3 H. CH3 brevisamide O:
A:
Q: i) BH3, THF i) H,О,NaOH ? CH3 CH3 -CH3 FHO- он -CH3 HO + + enantiomer enantiomer I II III IV
A:
Q: These two structures, shown as Fischer projections, are: Br H3C- H- H-N- H- NH2 Br the same compound…
A: Stereisomers are those which have same molecular formula but different arrangement of atoms in…
Q: Indicate whether the pair of structures shown represent stereoisomers, constitutional isomers,…
A:
Q: Which statement is true about these Fischer structures? CH3 CH3 CH3 HO CH3 it: -H HO- -H H- -OH H-…
A: Given that,
Q: Draw three-dimensional representations of the following compounds. Which have asymmetric carbon…
A: Asymmetric carbon atoms are those carbon atoms on which the attached 4 groups are different from one…
Q: 5) For each of the pairs of molecules drawn below, place letters in the corner boxes corresponding…
A: The isomers which have the same connectivity of atoms in the molecules but different spatial…
Q: Describe the relationship between each of the pairs of isomers shown by recording Constitutional,…
A: -> After flipping we get conformation isomer.
Q: Consider the molecule shown below: OH [1] Complete the IUPAC name of the molecule below by…
A: Conformation - Is the spatial arrangement which the atom in a molecule may adopt and freely…
Q: Which is MORE BASIC? || CH3CH2CH2-C-NH2 CH3CH2CH2CH2NH2
A:
Q: the following compound? CHCIBICH:CHBrCH3 a) 0 b) 1 c) 2 d) 3 will not show optical activity? a)…
A: Note: Since you have posted a question with multiple subparts, we will solve the first three…
Q: How are the compounds in attached pair related to each other? Are they identical, enantiomers,…
A: To identify whether the below pair are identical, enantiomers, diastereomers, constitutional…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- 1) Are the molecules A and B... conformational isomers? Diastereomers? Enantiomers? Position isomers? Non-related? 2) What about the molecules B and C? 3) What about the molecules C and D?-Tetrahedral representation of the enantiomers of the structure below -tetrahedral representations of both R and S enantiomers of the structure below -what functional groups does the given structure posses?Locate with an asterisk the stereogenic centers (if any) in the following structure: B. C6H5CH2CH(OH)CO2H
- Locate with an asterisk the stereogenic centers (if any) in the following structure: A. HOCH2CH(OH)CH(OH)CHOWhich of the following structures represent the same stereoisomer? A. only 1 and 2 B. only 1 and 3 C. only 2 and 3 D. 1, 2 and 3 Please explain.1. Name and draw all possible isomers of trimethylcyclopropane. Which of the isomers is chiral?2. If this is chiral, then is it an R or S isomer?
- Draw the bond-line dash-wedge structure and the Fischer projection of (R)-2-bromopentane draw the Newman Projection down the C2-C3 bond in the lowest energy conformation. Pls make it neat and explain in simple termsDraw three-dimensional representations of the following compounds. Which have asymmetric carbon atoms? Which have no asymmetric carbons but are chiral anyway? 1-chlorobuta-1,3-diene(a) meso-hexane-3,4-diol, CH3CH2CH(OH)CH(OH)CH2CH3, draw any diastereomers.
- H3C Cl \ / C=C / \ H2N I (R)-enantiomer (Z) isomer (S)-enantiomer (E) isomer meso compoundDoes 2,3-dichlorobutane have optical activity?(1). No.(2). Only in solution.(3). Yes, in any state (solid, liquid and gas).(4). It depends on the stereoisomer we are referring to.Draw all the isomeres of a thioester with molecular formula C4H8OS

