Q: Draw a plausible mechanism for each of the following transformations: a. b. C. N- N H H₂O* H₂O* H₂O…
A: We have to give the mechanisms of the following transformations.
Q: The conjugate acid of HSO 4 is
A:
Q: If a sample of a certain solution is determined to have a pH of 8.9, what is the concentration of…
A: pH is defined as the negative logarithm of hydronium ion, H3O+ concentration of base ten .The…
Q: 10. Your local radio station Jump! 106.9FM operates at a radio frequency of 106.9 MHz. Your cars…
A:
Q: ? H
A: The Kornblum oxidation is an oxidation reaction that converts alkyl halides into carbonyl compounds.
Q: A large sport utility vehicle has a mass of 2600 kg. Calculate the mass of CO2 emitted into the…
A: Mass of SUV = 2600 Kg Initial speed, V1 = 0 Final speed, V2 = 66 mph = 66 mile per hr 1 mile/hr =…
Q: The major product that completes the following reaction is:
A: An acid anhydride is a carboxylic acid derivative and it is readily hydrolyzed when it reacts with…
Q: Predict the product(s) for the reaction shown. OH + enantiomer 1. BH3-THF 2. H₂O2/NaOH/H₂O OH +…
A:
Q: What are the appropriate reagents to carry out the conversion shown? LOH 0=0° O KMnO4/NaOH/H₂O O…
A: we have to select the reagents needed to make the given conversion
Q: Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any…
A:
Q: 34. Pick the true statements from the list below. * 1. Electrons occupy any vacant valence orbitals…
A: In this question, we have to choose the correct option for the given questions.
Q: For each system listed in the first column of the table below, decide (if possible) whether the…
A: Answer: Entropy change is the measure of system's disorderliness. In more possible states, system…
Q: a) For the three Lewis structures shown below, determine the formal charge of each of the three…
A: Formal charge ---> number of valence electron in neutral atom or element —number of valence…
Q: Based on the results of the KMnO4 and Lucas tests on the representative alcohols (alcohol, phenol,…
A: Lucas test: Given below is a table describing the positive Lucas test observations for different…
Q: You have 20.0 mL of a Na₂CO3 solution of unknown concentration. To this solution you add excess HCI…
A: Molarity is number of moles present in litre of a solution.
Q: f 5.42 g of CuNO3 is dissolved in water to make a 0.410 M solution, what is the volume of the…
A:
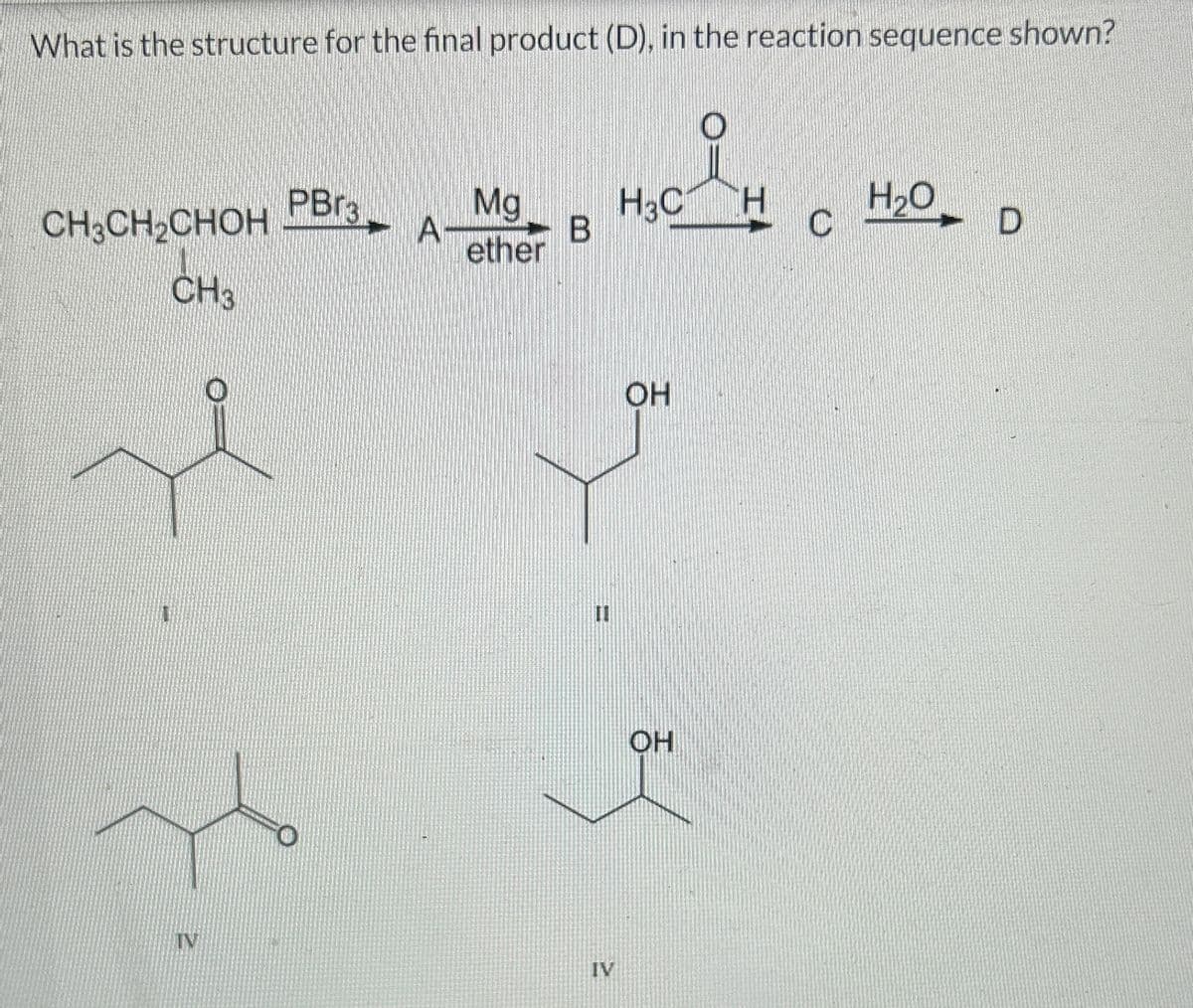
Q: For the following reacrion sequence, what are the correct structures of products A through D? OH or…
A: Synthesis of alcohol Trimethyl silane act as protecting group for alcohol.TBAF act as deprotecting…
Q: The standard reaction free energy AG-835. kJ for this reaction: 2 Al(s) + Fe₂O3(s)-Al₂O3(s) + 2…
A: Answer: Given chemical reaction: 2Al(s)+Fe2O3(s)→Al2O3(s)+2Fe(s) Gibbs free energy change for this…
Q: A solution contains 8.38×10-³ M potassium hydroxide and 1.36x10-2 M ammonium carbonate. Solid…
A: We need to find the Mn2+ concentration at which carbonate begins to precipitate. Then, we can find…
Q: 23.0 g of Z2 are mixed with excess X2. How many grams of product are produced? 2 X2 + 3 Z2 →…
A:
Q: 6. A student performs the reaction below in three experiments studying initial concentrations and…
A: The order of the given chemical reaction can be determined by measuring the rate of reaction by…
Q: 5. The human eye can detect as little as 2.5*10-16 joules of light. How many photons of green…
A:
Q: For each of the metabolic transformations (e) through (h), determine whether the compound on the…
A:
Q: A typical chest X ray exposes a patient to an effective dose of 0.05 mSv. A) How many rem is this?…
A: The part A is to convert the given quantity into rem. Part B is to count number of chest X rays.
Q: So what are there equilibrium constant values?
A: In this question, what are there equilibrium constant values in the preceding question.
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A:
Q: Which of the following ionic compounds is expected to have the smallest lattice energy? O AICI 3 O…
A:
Q: What are the appropriate reagents to carry out the following conversion? OH O KMnO4/NaOH/H₂O O…
A:
Q: A 50.0 mL solution of Mg(OH)2 of unknown concentration is titrated with 0.348 M HCI. The equivalence…
A: Volume of Mg(OH)2 = Concentration of HCI = 0.348 M Volume of HCl = 62.8 ml
Q: A 23.2 g piece of metal (C = 0.455 J/g oC) initially at 14.2 oC is placed in 35.2 g of water…
A:
Q: 3. Provide the product(s) or reagent(s) as appropriate. ? 1. H₂O, H₂SO4 2. H₂CrO4 1. NaOH 2. H₂CRO4…
A: We have find out the products or reagents in the given reactions. Note -as per our company…
Q: How would you find the equilibrium constant value?
A:
Q: The octane rating of gasoline is a relationship of the burning efficiency of the given gasoline…
A: 18mole of water producing in this reaction.
Q: Instant cold packs, often used to ice athletic injuries on the field, contain ammonium nitrate and…
A: It is an application of Principle of calorimetry. Here we are required to find the Enthalpy of…
Q: Classify the substance shown in the sketch below. You can click the other tabs in the sketch to get…
A: There are three forms of matter that is solid, liquid, and gas based on the internuclear…
Q: Which set of reagents is necessary to carry out the following conversion? CH3CH₂CH₂CHCH3 Br +…
A: The required conversion consists of elimination reaction followed by hydroboration oxidation of…
Q: How do you find the net ionic equation of a solution NaC2H3O2. Is it a Ka or Kb equation?
A: In this question, we have to write the net ionic equation of a solution NaC2H3O2. Is it a Ka or Kb…
Q: Assign the hybridization and bond angles at each of the labelled atoms H. H C. H H C C HIN: H H 4 I…
A:
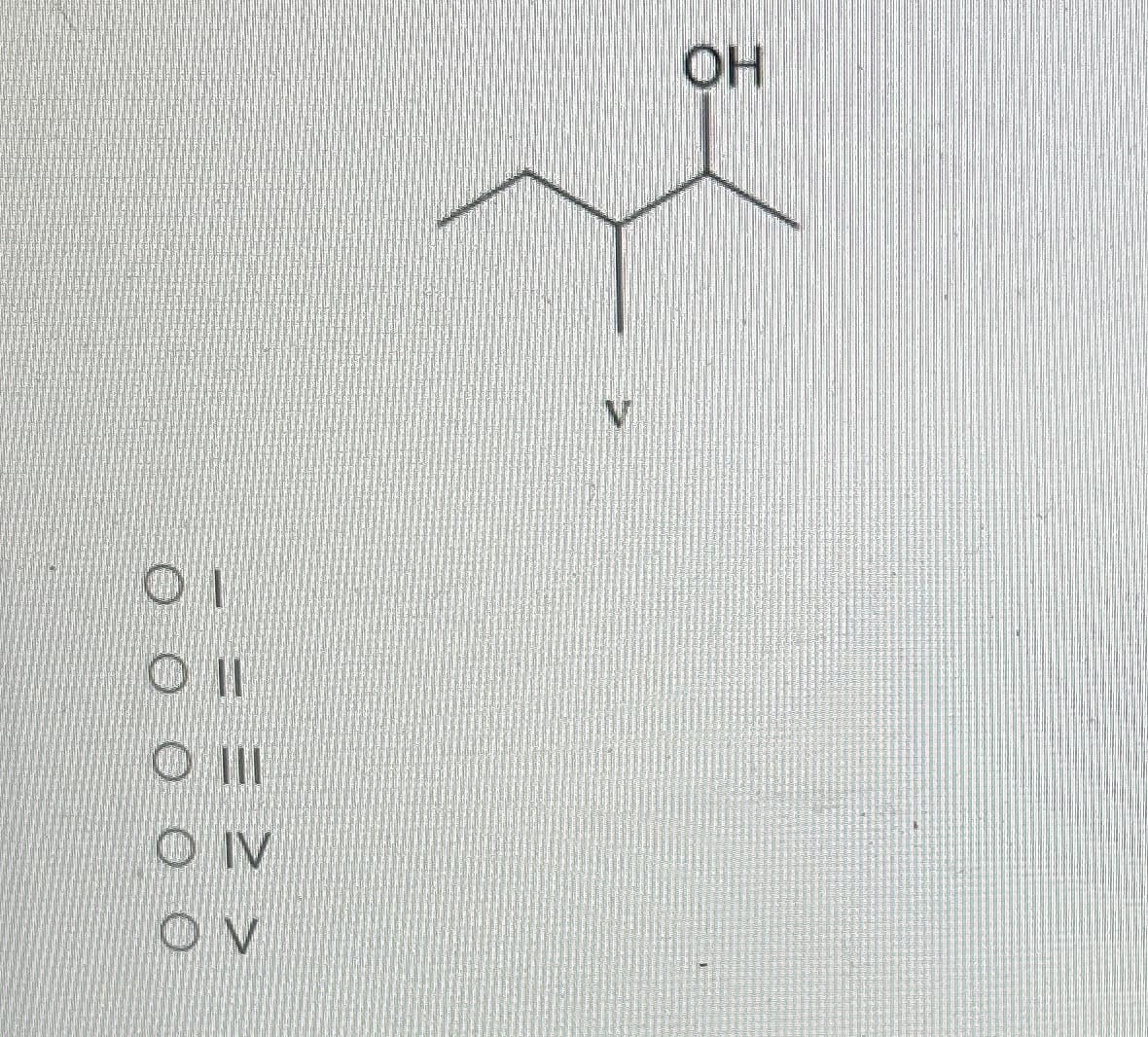
Q: What is the likely product of the reaction shown? لیلی میگن میلی لیکن ملیٹی کمیٹی OT 11 O III OIV OV…
A: Aldehyde and ketone both can be reduced by NaBH4 to corresponding alcohol. First the lone pair of…
Q: Use the phase diagram of Substance x below to find the pressure at which the boiling point of…
A: Boiling point: The temperature at which the vapor pressure of the liquid becomes equal to the…
Q: Which of the resonance structures below is least significant? O .. :0: structure C structure A…
A: The least stable resonating structure is least significant
Q: A solution of volume 0.500 L contains 1.68 g NH3 (Kb = 1.8 x 10^-5) and 4.05 g (NH4)2SO4. If 0.88 g…
A: Given : Mass of NH3 = 1.68 g Mass of (NH4)2SO4 = 4.05 g Mass of NaOH = 0.88 g Volume of solution…
Q: If 250.0 mL of HNO3 are reacted with 34.8 mL of 0.721 M Ba(OH)2, to reach the equivalence point,…
A: Given -> Volume of HNO3 = 250.0 ml Volume of Ba(OH)2 = 34.8 ml Molarity of Ba(OH)2 = 0.721 M
Q: Use the table of standard reduction potentials from your textbook to complete the following table.…
A:
Q: you change the number of each subatomic particle neutrons✔ f protons of electrons Choose... Jay…
A: Atom is smallest particle which can not be further divided in to any other particle. Element is a…
Q: The number of unsaturated bonds in this fatty acid is | ball & stick + labels
A: Lauric acid, also known as dodecanoic acid, is a 12-carbon saturated fatty acid that shares many…
Q: ОН SOCI₂/pyridine це IV OH ОН Ө
A: Alcohol are organic compound which have OH functionality. Here we are required to find the product…
Q: If the mass of the baby is 9lb and 8.0 oz, how many kilograms does the baby weigh?
A: The conversion factors are 1 lb =0.453592 kg 1 oz =0.0283495 kg
Q: When a liquid is in dynamic equilibrium with its vapour at a given temperature, T, which of the…
A: The state of a system in which a reversible reaction is occurring ceases altering the ratio of…
Q: 1.500.0 liters of a gas in a flexible-walled container are prepared at 700.0 mmHg and 200.0 °C. The…
A:
Step by step
Solved in 3 steps with 3 images

- Show the SN2 mechanism in the reaction between (S) 2-bromobutane and iodide ion. Label the product and draw a enery diagram for this reaction.I need help drawing the product of this reaction. The starting material is vanillin and it is being treated with SO3 & H2SO4 in order to generate a product with the formula C8H8SO6.Give the structure of the product(s) for each step of the following reaction sequence.
- What is the main product from the reaction sequence below?The synthesis above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order. Be sure to consider stereochemistry and regiochemistry.A. NaOHB. mCPBAC. OsO4, tBuOOHD. NaHE. H2, LindlarF. CH3CH2CH2BrG. CH3CH2ClH. NaNH2I. H3O+J. Na, NH3(l)Which mononitro product is the result of the nitration reaction of the compound whose open structure is given below? or consists of products? Show it.
- What is the product of each of the following reactions? complete both parts A and B.What is the product of the reaction sequence shown in Image 24?A benzene ring reacts with these reagents in order: 1) Cl2, AlCl3 2) Mg 3) CH3CH2CHO 4) H + 5) conc. H2SO4, heat, -H2O I am confused about what happens after the first step.
- Show how to make these deuterium-labeled compounds, using CD3MgBr and D2O as your sources of deuterium, and anynon-deuterated starting materials you wish.(a) CH3CH(OD)CD3Complete the following reaction equations :(i) C6H5Cl + CH3COCl →(ii) C2H5NH2 + C6H5SO2Cl →(iii) C2H5NH2 + HNO2 →i need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,

