Q: pH = %3D
A:
Q: Based on the absorption spectrum below, if n=1 for this element occupies an energy level of -21 eV,…
A: The expression of energy of a specific orbit for hydrogen like atoms is: En=-kZ2n2 = -13.6 eV ×Z2n2…
Q: Calculate, in units of Dₒ, the LFSEs of the following (show all work): a. [FeF6]3- b. [Co(H2O)6]3+…
A:
Q: A Gouy balance is used to determine the magnetic susceptibility of a substance. A strong…
A: We have to predict the nature of elements.
Q: An experiment research would produce a 1200 gram magnet material Neodymium-Iron-Boron Nd2 Fe14 B…
A: First of all calculate the numberof moles of each of the molecule by using the formula -mole = mass…
Q: 9. Which of the following is paramagnetic? Z of Zn = 30, Cr = 24, O =8 Group of answer choices a)…
A: a) Zn+2 = The atomic number of Zn is 30. Here +2 means 2 electrons removed from the outermost shell.…
Q: Which one of the following atoms is diamagnetic in the ground electronic state? Drawing an…
A: d) Mg is diamagnetic
Q: Reducedicr 7) 8KCIO, + C„H„O 8KCI + 11H,O + 12CO, 8) 3H,C,O, + 2K,MnO, 6CO, + 2K,O+Mn,O, + 3H,O 9)…
A: 7. 8KClO3 + C12H22O11 <----> 8KCl + 11H2O + 12CO2 Reducing agent; C12H22O11 Atom reduced :…
Q: Calculate the effective nuclear charge on a 5s, a 5p, and a 4d clectron in a tin atom.
A: Effective nuclear charge (Z* ) is calculated using the formula: Z* = Z - σ Z= Atomic number σ =…
Q: Calculate the energy (in eV) of the process: Br(g) + Na(g) → Br- + Na . Data: Ionization energy of…
A: The given process involves the ionization of gaseous sodium atom to sodium ion and gain of electron…
Q: Rank the elements by effective nuclear charge, Zef, for a valence electron. Highest Zeff Lowest Zeff…
A:
Q: what is the dn electron configuration of Cu(II) ion
A: We are authorised to solve only one question at a time. Please post rest of the questions…
Q: ) Cirele the ions with the 4s orbitals empty? Sc Tit Cr Mn
A: The electronic configuration of the given ions are: Sc :[Ar] 3d14s2Sc3+ : [Ar]Ti :[Ar] 3d24s2Ti4+:…
Q: Given the following selectivity coefficients for a K+ ISE, which of the below ions would cause the…
A:
Q: What is the Ka for CH,CO,H (pKa = 4.75)? Select the best answer. 1.00 M 15. %3D a. 4.19 x 103 M 4.19…
A: Given, pKa of CH3COOH = 4.75 Acid dissociation constant of CH3COOH = Ka = ? Options are : a. 1.00…
Q: Compute the ionization potential for five-times ionized (Mg11+) and also compute the wavelength of…
A: In the ground state , electron in Mg11+ present at n = 1 energy of electron in Mg11+ =…
Q: If A is an m x n matrix, and B is an n x m matrix, then A + B is ann x n matrix. (Assume m + n.) No.…
A: For addition of matrix size of both matrix should be same
Q: 7. Indicate if whether the specie is paramagnetic c Specie Magnetism Ge Kr Fr Al*
A: The question is based on the concept of atomic structure. it involves writing of electronic…
Q: Calculate the spin-only moment for the following: a. Cr b. an element in the chalcogen family
A: Concept introduction: Spin only magnetic moment is the magnetic moments arises due to the spin of…
Q: Ca* is paramagnetic. True O False
A:
Q: ii) Calculate the screening constant (sigma) on a 2p electron of N and N and deduce the values of Z*…
A: Introduction- Screening constant σ and the effective nuclear charge (Z*) is calculated by the help…
Q: +3 16- Electron spectrum Cu (H20),* compared to the electron spectrum Ti (H2O)6*³is +3. very wide…
A: The question is based on the concept of coordination chemistry. we have to describe the nature of…
Q: What's a hypothesis about the experiment Colorimetric Determination of Iron?
A: The colorimetric analysis is the determination of change in the intensity of the color of the…
Q: S PeriodicTable O ALEKS Link A ALEKS - Rachel West - Learn X K VHL Central | Log in…
A: Calculation:Let a be the first brand gallons of 45% antifreeze needed and b be the second brand…
Q: photo electric effect is maximum in a Cs b Na c K d Li
A: When light falls on any material plates, it emits electron or free carriers. This phenomenon is…
Q: 22. How does the answer in question 21 explain the splitting pattern of H,? Does the splitting…
A: Given compound,
Q: The spin and parity of J/psi meson J/y : JP О а. 1 O b. 1* Oc. 0* 33 d. 0
A: Concept: The J/ψ(J/psi) meson or psion is a type of subatomic particle. It is a flavor-neutral meson…
Q: i…
A: We have given that The electron configuration of an atom is the representation of the arrangement…
Q: with the exception of ICP- MS , all elemental analysis methods measure some form of electomagnetic…
A: Analysis of elements is performed has become really important nowadays in order to monitor the…
Q: If t = 2, what can be deduced about n? %3D
A: There are total four set of quantum numbers to give address of electron in an atom…
Q: A line in the Balmer series occurs at 434 nm. What is n high for this transition ? 3
A:
Q: -E a.164eV Consider a silicon crystal at 300° K doped with arsenic atoms. Depending on the following…
A: Given: ni= 1.45 x 1010 cm-3 ne= electron concentration , n0 nh= hole concentration , p0 Temperature…
Q: Compare between Electron ionization (El) and Chemical ionization methods.
A: Chemical ionization requires a lower amount of energy compared to electron ionization (EI), but this…
Q: Determine the complete electronic configuration of Ni. Z of Ni = 28
A: Ans
Q: -Define: mole, electropositive element, polymorphism, Anisotropy, Heisenberg's uncertainty…
A: Atom is defined as smallest particle which can not be further divided into small particles.
Q: 1. Give the set of quantum numbers for the following: a. The e- is in the 3rd energy level,…
A: Given,
Q: Show the electron configuration of vanadium (V). Drag the appropriate labels to their respective…
A: Part A = Electronic configuration of Vanadium = The atomic number of Vanadium is 23. Hence, the…
Q: How do we get X=3 for ClF3
A: I think you are asking about how Formula of the compound ClF3 formed means where that 3 atoms of…
Q: There areow many unpaired d electrons in Ni3+ (Z = 28).
A: Ni have 28 electrons . After giving 3 electrons Ni3+ have 25 electrons
Q: Which of the following is considered a key feature of a relative method? A) Based on the interaction…
A: Relative technique is the strategy for contrasting present information and reference upsides of…
Q: 4. For the n=6 to n=2 transition: v = A in nm = A in Å = Color of corresponding line in the hydrogen…
A: When transition of electron from higher energy level to lower energy level occurs, then energy will…
Q: 2.14 The net potential energy between two adja- cent ions, EN, may be represented by the sum of…
A: Given: The net potential energy between two adjacent ions, EN, may be represented as; EN=-Ar+Brn
Q: 1089
A:
Q: K =- %3D
A:
Q: Ca20 Au79 I53 FIND THE FOLLOWING: A. AUFBAU PRINCIPLE B. HUNDS RULE
A: 1) Ca (20) -
Q: 3. For the n=5 to n=2 transition: E = _ % = A in Å = A in nm = Color of corresponding line in the…
A:
Q: Calculate the effective core load for 58Ce, 59Pr and 60Nd containing 4f electrons.
A: Introduction: Effective nuclear charge: It is a function of total nuclear charge which is…
What is the Z effective of Ni 1-
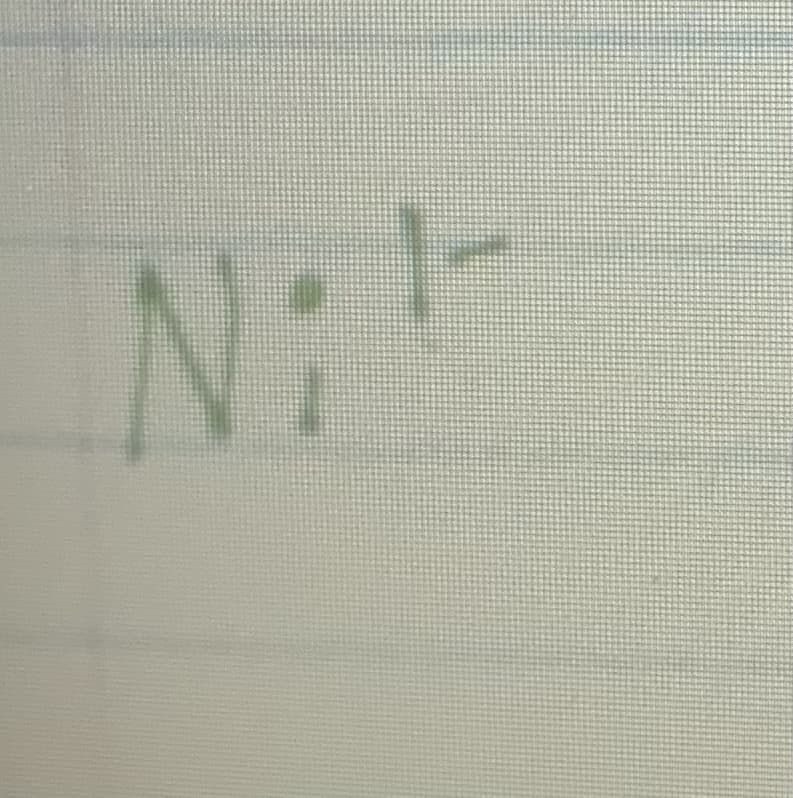
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


