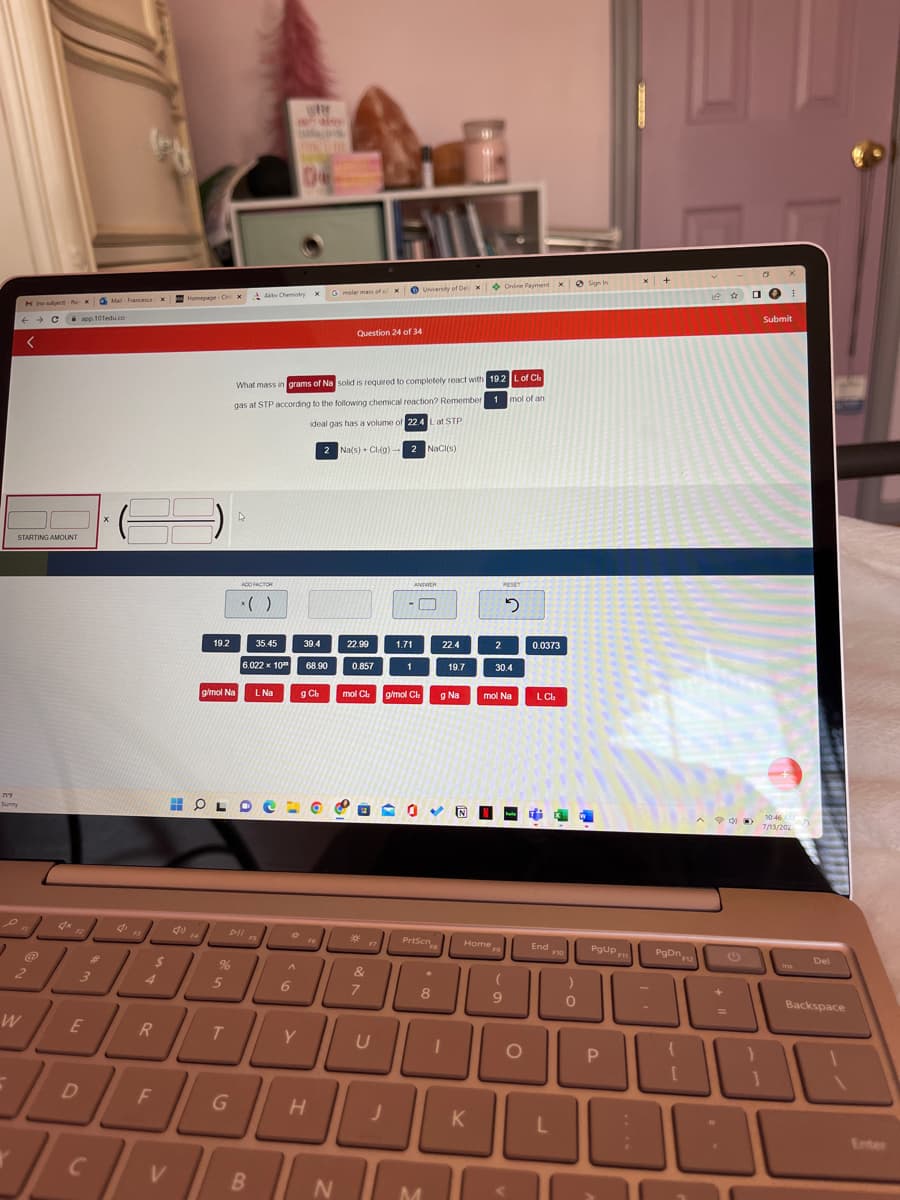
What mass in grams of Na solid is required to completely react with 19.2 L of Ch gas at STP according to the following chemical reaction? Remember 1 mol of an ideal gas has a volume of 22.4 L at STP 2 Na(s) Cl(g)- 2 NaCl(s)
What mass in grams of Na solid is required to completely react with 19.2 L of Ch gas at STP according to the following chemical reaction? Remember 1 mol of an ideal gas has a volume of 22.4 L at STP 2 Na(s) Cl(g)- 2 NaCl(s)
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter10: Energy
Section: Chapter Questions
Problem 3ALQ: ou place hot metal into a beaker of cold water. ol type='a'> Eventually what is true about the...
Related questions
Question
Transcribed Image Text:717
Sunny
2
Mo subject-RxMail-Francesca
← C app.101edu.co
→
STARTING AMOUNT
F
2
W
4x
3
E
D
عالی
C
X
F3
4
$
R
F
x Homepage-CHI
V
19.2
14
g/mol Na
%
5
T
X
G
ADD FACTOR
HOLDE
Alte Chemistry XG molar mass of ox University of Deli
What mass in grams of Na solid is required to completely react with 19.2 L of Ch
gas at STP according to the following chemical reaction? Remember 1 mol of an
ideal gas has a volume of 22.4 L at STP
2 Na(s) + Cl(g) 2 NaCl(s)
x()
35.45
6.022 x 10
Dll s
FS
B
L Na
wa
W
A
6
39.4
68.90
g Ch
Y
Question 24 of 34
H
N
22.99
0.857
30
1.71
&
7
1
ANDWER
J
□
mol Ch g/mol Ch g Na
030R
PrtScn
22.4
✰
MOD
U
M
19.7
8
Home
K
Online Payment xSign In
2
RESET
2
30.4
mol Na
(
9
O
0.0373
LCI:
End
L
F10
)
0
PgUp
P
x +
Pgon12
+
D
31
X
I
Submit
10:46 AM
7/13/202
Del
Backspace
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning