What orbitals are involved in forming the indicated bond in the following compound? CH3 Link ds-ds pue ds-ds O O sp3-sp3 and 2p-2p O sp³-sp³ O sp2-sp? and 2p-2p
What orbitals are involved in forming the indicated bond in the following compound? CH3 Link ds-ds pue ds-ds O O sp3-sp3 and 2p-2p O sp³-sp³ O sp2-sp? and 2p-2p
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter17: Conjugation And Molecular Orbital (mo) Theory
Section: Chapter Questions
Problem 10E
Related questions
Question
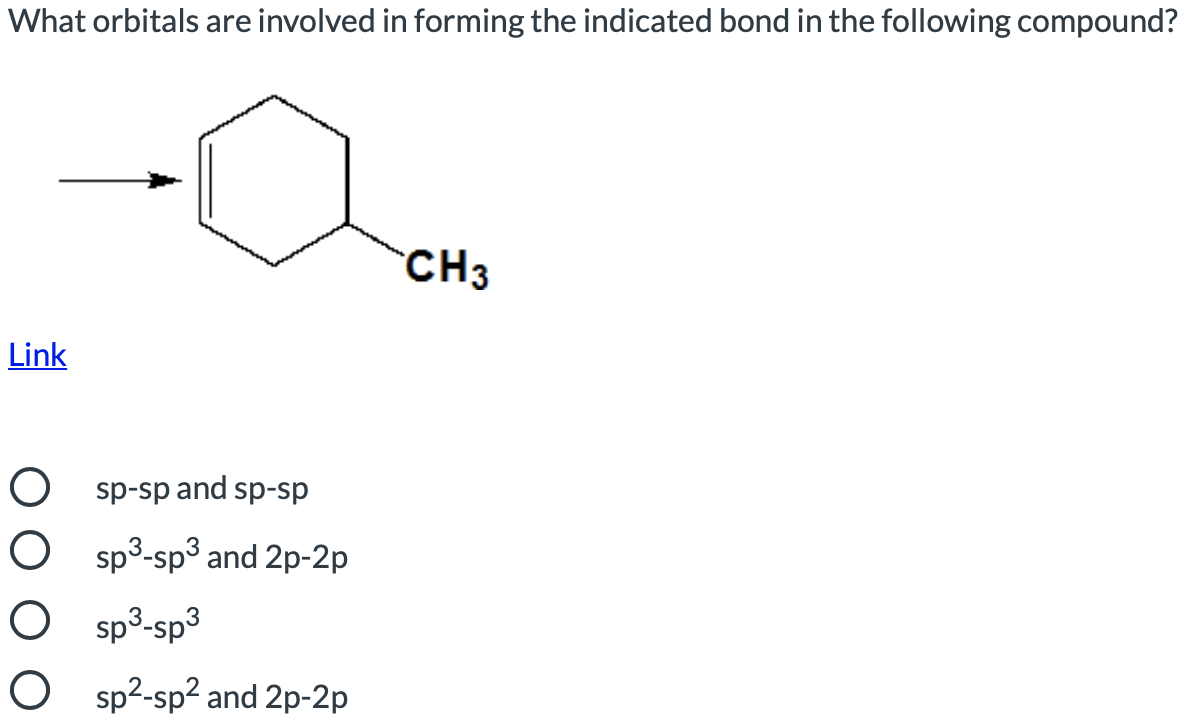
Transcribed Image Text:What orbitals are involved in forming the indicated bond in the following compound?
CH3
Link
ds-ds pue ds-ds O
O sp3-sp3 and 2p-2p
O sp³-sp³
O sp2-sp? and 2p-2p
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning