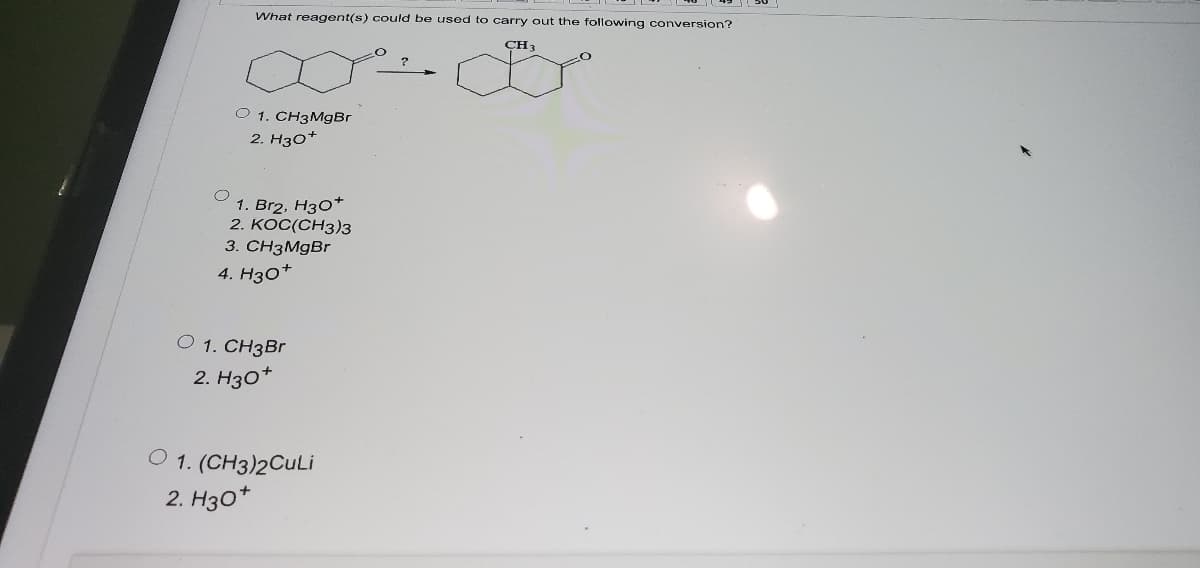
What reagent(s) could be used to carry out the following conversion? CH3 O 1. CH3MgBr 2. H30* 1. Br2, H30* 2. KOC(CH3)3 3. CH3MGBR 4. Нзо* O 1. CH3Br 2. Нзо* O 1. (CH3)2CULİ 2. Нзо*
Q: Which reagents best lead to this transformation? A B 1. SO3, H₂SO4 2. CH3CH₂CI, AICI3 1.CH3CH₂CI,…
A: Here we have to determine the best reagents among the following which can convert benzene to…
Q: Arrange the alkenes in each set in order of increasing rate of reaction with HI and explain the…
A: The species alkene in the front of the acid hydrogen iodide HI to generate the alkyl halide through…
Q: Fill in ALL the missing parts to the organic transformations in this sequence - T tBuOK CH,CH;CH,…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: Which reagents would you choose to complete the following conversion? Step , step and finally step…
A:
Q: D (C3H1204) H30* heat heat E
A: A scheme is given to us . In this scheme we have to identify unknowns
Q: CH,CH, (CH;),CHCH,C=CH (CH;),CHCH,
A: synthesis is the process by which one or more chemical reactions are performed with the aim of…
Q: 2. Answer the following questions based in the scheme below: (0) Determine the principle involved in…
A:
Q: Select the starting material for the transformation below. MCPBA CH3 CH3 CH2 C Oc
A: Given,
Q: What series of reagents are necessary to carry out the following conversion? CH3 `Br CH,ca / AICa…
A: Mechanism is given below in next step
Q: Which compound will produce an irritating burnt fat odor when upon heating together with KHS04? %3D…
A:
Q: a) Explain in detail why reaction of methylcyclopentane with Cl2/light gives a mixture of products…
A: Reactivity of hydrogens : tertiary > secondary > primary. Reactivity of halogens: Chlorine…
Q: Which of the following reagents can be used to accomplish each of the transformation? la-l OH CH3LI…
A:
Q: What reagents are needed to carry out the conversion shown? OH OPCC/CH₂Cl2: H3O+/Br2: NaCN O…
A:
Q: What reagents are needed to carry out the conversion shown? EtO Et Et O 1. HOCH2CH2CH3/H2SO4; 2.…
A:
Q: What reagents are needed to carry out the conversion shown? N- OCH3 CI- OCH3 Cl2/FeCl3; HNO3/H2SO4;…
A:
Q: Show the reagents and experimental conditions necessary to bring conversion in good yield.
A: Given conversion reaction is:
Q: Which of the following within a tetrahedral intermediate leaves most readily as a nucleofuge? *…
A: A nucleofuge is a species that acts as a leaving group in the reaction. It is a leaving group…
Q: Choose the reagents that can be used to achieve the following conversion. More than one reaction is…
A: This reaction involves conversion of ketone into alkene. It can be brought about by first…
Q: The correct reagent for the following transformation is... CH3 H3C H ??? Select one: O BH3 then…
A: When an alkene is treated with Hydrogen gas in presence of palladium metal as catalyst, then cis…
Q: show the reaction of 2-methyl-2-butene and 2-methyl-1-butene with Br2 in CH2Cl2
A:
Q: Select the correct reagent for the following transformation* ?? CH3CH2CHCH3COCI/Alcl3 O…
A: Benzene is an aromatic compound that is planar, cyclic, conjugation of pi-electrons and, obeys…
Q: B- Instructions: What reagent and/or catalysts are necessary to bring about each conversion? Br 1.…
A:
Q: This compound was synthesized from benzene by the scheme outlined. CH3 Br ČH3 1. CH3CI, AICI3 2.…
A: The first step of this synthesis is called Friedel craft alkylation . It is an electrophilic…
Q: Pick the reactant or solvent in each part that gives the faster elimination reaction.a. reaction of…
A: The elimination reactions are of two types such as unimolecular and bimolecular elimination…
Q: (CH3)3CHOH is reacted with CH3-O-C--C6H5 in a transesterification reaction. What is the M+ of the…
A: Given reaction is trans esterification reaction.
Q: 1,2 addition of Br2 to an alkene results in a _______________. A. alcohol B. vicinyl dihalide C.…
A: 1,2 addition of Br2 to an alkene results in a ?
Q: Complete the following reactions by adding the massing major product: CH3 H3C-CH2B NaOH 1. H. Br…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Encircle the most stable alkene. CH3 CH3 CH3 a) CH3 CH2CH3 b)
A: A question about the stability of alkene, which is to be accomplished.
Q: Which of the following series of reagents could be used to carry out the conversion shown? NO2 Br O…
A: Conversion of benzene into m-nitrobromobenzene.
Q: dentify reagents that can be used to achieve the following transformation: HO, dilute H2SO4 1) BH3 •…
A: Alkenes are hydrolyzed into alcohol. The hydration reaction can be proceeded by the addition of a…
Q: Which will give the most effective yield of ether? CH3CH20 + CH3-CH-CH2BR CH3 PhO- + CH3-CH-Br CH3…
A: Synthesis of ether by Williamson Ether Synthesis.
Q: (b) Give the reagents and reaction conditions necessary for the following conversions (i)…
A:
Q: Which is the method of choice for preparing the ether (CH3CH2),COCH3? (a) (CH3CH2), COH + CH3MGBT…
A: Since you have asked multiple questions, we will answer only first question for you. In order to get…
Q: Show the reagents and experimental conditions necessary to bring conversion in good yield.
A: A chemical reaction is symbolic representation of the conversion of substances to new substances. In…
Q: Which reagent would be best suited for the conversion below? H3CH2C
A: A reactant is alpha, beta unsaturated carbonyl compound, and it shows 1,2 addition and 1,4 addition…
Q: Which pair of reactants is most likely to undergo an E2 reaction? O CH3SH + CH3CH₂CBr(CH3)2 at 20°C…
A: The given problem is based on the chemical properties of alkyl halides.
Q: Assuming a 56.8% yield, how kany ml of alkene is required to produce 22.5 mL of…
A: Acid hydrolysis of alkenes will result in the formation of alcohol. The acid hydrolysis of…
Q: Which reagents will give the transformation shown? LOH (1) Вн3: (2) Н2О2 a. (1) Hg(OAc)2; (2) NaBH4…
A:
Q: 4. In the following examples (where R represents the rest of the molecule), tell which is the best…
A: A leaving group is the one ,which departs with a pair of e- in heterolytic cleavage. A good leaving…
Q: Show the reagents and experimental conditions necessary to bring conversion in good yield.
A: The reagents and experimental condition has to be given for given convertion.
Q: Provide the correct set of reagents that would complete the following rxn? a) ) LAH, 2) HzO d) No,…
A: The reaction taking place is given as,
Q: Complete the following reactions. Only include the major products. а. CH3 c=C + HF CH3 H
A: This addition will take place according to tha markonikove's rule , which states that in…
Q: IV. For the given reaction, show which product is formed and why? 2-chloro-1,1-dicydlohexylethane…
A: Carbocation is any cation containing an even number of electrons in which a significant portion of…
Q: The reagent required for the following conversion is CH3. CH3-C= C- CH, → C=C H CH3 Na/Liquid NH3…
A: Conversion of alkyne to alkene means the addition of hydrogen to the triple bond. When hydrogen gets…
Q: (a) CH3 CH3CH2CH2CH2-0-¢ + HI CH3 (b) H2SO4 + H20 (C) H3C 10-CH2CH2CH3 HBr
A:
Q: What reagents are needed to carry out the conversion shown? OH هده O 1. HNO3/H₂SO4: 2. Br2/FeBr3: 3.…
A: Solutions Hence option (D) is correct answer...
Q: Show how to bring about each conversion in good yield.
A: The given compound is acyl chloride. It reacts with NaCH(COOC2H5)2 and further undergoes hydrolysis…
Q: What reagents are needed to carry out the conversion shown? HO HO 1 mole / H2SO4 2 moles /H30* O…
A: The reaction taking place is given as,
Q: Which reagent can be used to achieve the transformation shown? * C,H5. C,H5. 1.) ? 2.) H,O* CH3 CH,…
A:
Q: What reagents would be needed to complete the following reaction? Hz CH3 CH3 H2/Pd AIC13/CH3CI…
A: When acetophenone treated with Zinc-mercury in Presence of Hydrochloric acid, then due to reduction,…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- 6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, PdAssuming a 56.8% yield, how kany ml of alkene is required to produce 22.5 mL of 2,3-dimethyl-2-butanol? MW of alkene: 84.16, d: 0.653 MW of 2,3-dimethy-2-butanol: 102.17, d:0.823Which reaction corresponds to the Kb for HSO4− ? a. H2SO4 ⇌ H+ + HSO4− b. SO42− + H2O ⇌ OH− + HSO4− c. HSO4− + H2O ⇌ OH− + H2SO4 d. HSO4− ⇌ H+ + SO42− e. HSO4− + OH− ⇌ H2O + SO42−
- 7. Choose the best reagent from the list below for carrying out each transformation. Place the letter of the reagent in the blank to the left of the reaction. (see attached screenshot). a. 1. O3 2. Zn, H3O+ b. 1. BH3, THF 2. H2O2, NaOH, H2O c. H2O, H2SO4, heat d. 1. OsO4 2. NaHSO3, H2O e. KMnO4, acid f. 1. Hg(OAc)2, H2O 2. NaBH4For the nitration of a monosubstituted benzene, which substituent, when attached to benzene, produces the fastest reaction rate? —Br —CH3 -OCOCH3 —OCH3Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heat
- What is the rate law implied by the mechanism given below? CH3COCH3(aq) + H+(aq) ←→ CH3C(OH)CH3+(aq) (fast, reversible) CH3C(OH)CH3+(aq) → CH3C(OH)=CH2(aq) + H+(aq) (slow) CH3C(OH)=CH2(aq) + Br2(aq) → CH3C(OH)CH2Br+(aq) + Br-(aq) (fast) CH3C(OH)CH2Br+(aq) → CH3COCH2Br(aq) + H+(aq) (fast) A. Rate = k[CH3COCH3][H+] B. Rate = k[CH3COCH3] C. Rate = k[CH3COCH3][Br2] D. Rate = k[CH3COCH3]2 E. Rate = k[CH3COCH3][Br2]/[H+] (Answer is A, looking for explanation why!).1,2 addition of Br2 to an alkene results in a _______________. A. alcohol B. vicinyl dihalide C. alkyl halide D. geminal dihalideWhich reagent is required to complete the reaction in Figure 11? [Generate a SMILES notation: https://jsme-editor.github.io/dist/JSME_test.html. Copy the notation (CTRL+C) and paste it as the answer to this question (CTRL+V).] * Your answer
- Rank (1-4) from most reactive to least reactive (in decreasing reactivity in an elimination reaction with NaOH in DMF at 100 degrees celsius)4. Show how you could make the following conversion. More than one step may be neededPick the reactant or solvent in each part that gives the faster elimination reaction.a. reaction of -OH with 1-chloro-1-methylcyclohexane or 1-chloro-3-methylcyclohexaneb. reaction of H2O with CH3CH(Cl)CH2CH3 or (CH3)2C(Cl)CH2CH3c. reaction of (CH3)3CCl with -OH in H2O or DMSO

