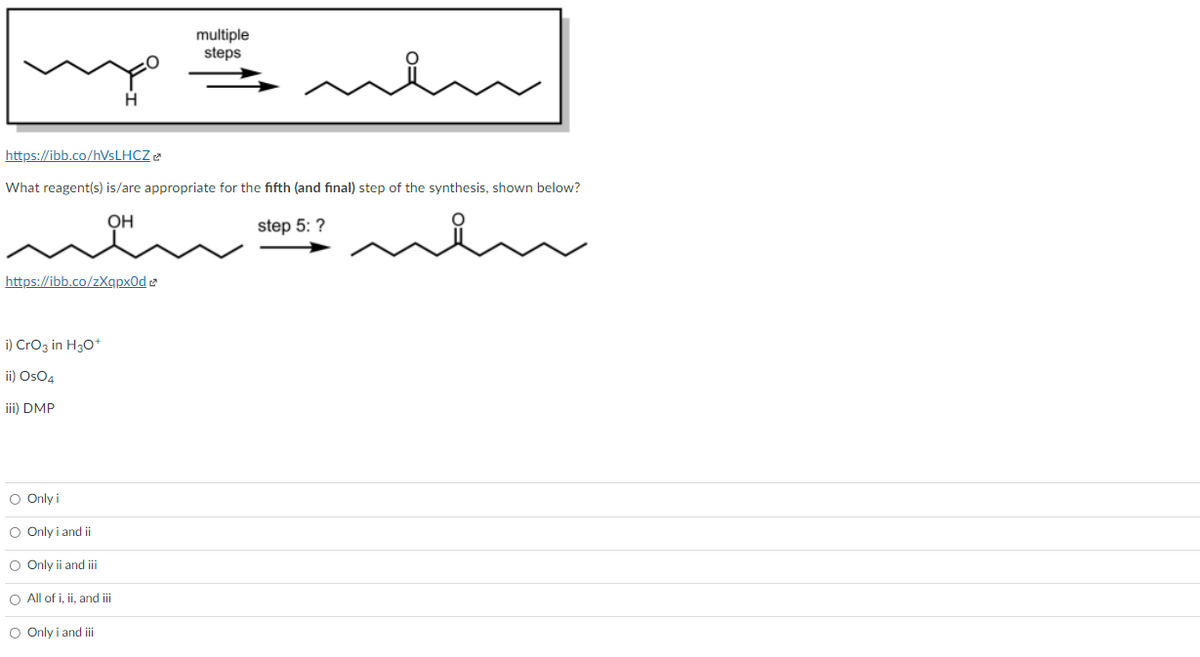
What reagent(s) is/are appropriate for the fifth (and final) step of the synthesis, shown below? step 5: ? https://ibb.co/zXqpx0d e i) CrOz in H30* ii) OsO4 iii) DMP Only i O Only i and i O Only i and i O All of i, i, and i Only i and i
Q: 1.Provide information regarding directing ability and strength of all substituents. 2What is the…
A:
Q: The following statements are true, EXCEPT: (a) The purpose of a titration is to determine the…
A: Titration is a analytical method to fnd out the unknown concentration of a solution by using another…
Q: Half-Life 1189 years Decay Rate, k Complete the table shown to the right for the half-life of a…
A: Given, Half life = 1189 years Decay rate, k = ?
Q: certain nucleus has a an atomic mass of 248.8 u and can release 178 MeV if it undergoes fission. How…
A:
Q: Calculate the pH, the pOH, and the molarity of all species in solution for a 6.15 M methylammonium…
A: We have asked to calculate pH, the pOH and molarity of all species in solution. The answer to this…
Q: List 3 common strong bases. Include both chemical formula and chemical name
A: Strong base are those which completely dissociates in aqueous solution and produce hydroxide ion.…
Q: Calculate the hydronium ion concentration for each of these aqueous solutions: a) solution has a pH…
A: Please find your solution below : pH is the measure of extent of how acidic or basic a solution is.…
Q: Phosphorus-32 is a radioisotope used in the treatment of leukemia and pancreatic cancer. If the…
A: Given, Phosphorus-32 in a sample decreases from 1.2g to 0.30g in 28 days . We have to calculate half…
Q: Calculate the potential of each electrode below: 1. a zinc electrode immersed in 0.0500 M Zn(NO3)2…
A: Since you have asked multiple questions, we will solve, first one for you. For remaining questions,…
Q: II What occurs in the initiation step of an addition polymer? Adding hydrogen gas Cross-linking Free…
A: The correct answer about initiation step of addition polymer is given below
Q: Part A For the hatl-reaction Cr"eCr, E- 0.424V excess Fe(s) is added to a solution in which (Cr"]…
A:
Q: 8. What mass of propane, C3H3, must be burned in order to produce 76,000 kJ of energy? Circle one:…
A: Given, C3H8 + 5O2 ➝ 3CO2 + 4H2O + 2200 kJ mass of propane, C3H8 must be burned in order to produce…
Q: Calculate the cell potential for the galvanic cell in which the reaction Cd(s)+Cu2+(aq) ->…
A:
Q: 3 NO2 (g) + H20(g)2 HNO3 (B) + NO (e) If 5.0 mol of NO21E) and 2.6 mol H20 (e) were placed in a 3.0…
A: Initial mole of NO2 = 5.0 mole Initial mole of H2O = 2.6 mole Volume = 3.0 L Equilibrium molarity of…
Q: Fill in the missing chemical formulas in the tables below: acid conjugate base base conjugate acid…
A: conjugate acid–base pair consists of two substances that differ only by the presence of a proton…
Q: dentify the monosaccharides present in each of the follow. ng disaccharides as glucose, galactose,…
A: a. Glucose+glucose b. Galactose+ fructose c. Glucose+ galactose
Q: Using an experimentally determined value (2.4×10−10) of Ksp, determine the value for the reaction…
A: we need to calculate the value of Q for Ag2CrO4
Q: Consider the following reaction and its AG" at 25.00 °C. Mg(s) + Ni" (aq) – Mg²* (aq) + Ni(s) AG =…
A:
Q: Product: DRY SENSITIZED SOLAR CELL (a) How was it produced using nanotechnology? (b) What are the…
A: (a) A dye-sensitized solar cell ( DSSC, DSC, DYSC or Grätzel cell) is a low-cost solar cell…
Q: According to Bronsted-Lowry, what is the definition of an BASE? Choose the right answer in the…
A: According to Bronsted-Lowry, what is the definition of an BASE? Choose the right answer in…
Q: Consider the electrochemical cell that is pictured below, that can be used as a battery. ) In which…
A:
Q: QUESTION 7 If the value of Kc for a given reaction at a given temperature is a small number, the…
A: Ans is : b
Q: sodium carbonate reacts with nitric acid according to the following equation…
A: Answer: part1) 0.59 mol Na2CO3, part2) 0.071 mol CO2
Q: 1. ) Complete and balance the following half-reactions: Cl, C a. b. НОВГ > HBr (acid soluti W,O, WO,…
A: Complete and balanced the following half-reactions:
Q: 10. Which one of the following is best associated with carbohydrates? a. amino acids b. phosphate…
A:
Q: Consider the following equation: 3C2H2(g) ↔ C6H6(g) ΔGof(C2H2, g) = +209.2 kJ/ mol; ΔGof(C6H6, g) =…
A:
Q: What is the objective of the Separation Technique: Paper Chromatography ?
A: A separation process is a method that converts a mixture or solution of chemical substances into two…
Q: Balance the following redox raction under acidic conditions. H2S(g) + NO3 (ag) → (3)ON In the final…
A: After balancing the electrons and atoms other than H and O , add water molecules to O deficit side…
Q: Predict the changes in volume of a helium-filled balloon at different temperatures. When you took…
A: Given Number of miles (n) = 0.200 moles Pressure (P) = 1.00 atm Temperature (T) = 0.00
Q: Calculate the molar concentration of a solution containing 18 g of NaOH in 100 mL water. (Density…
A: Mass of NaOH = 18 g Volume of water= 100 ml = 0.1 L Molar mass of NaOH = 40 g/mol
Q: Complete the table shown to the right for the half-life of a certain radioactive substance. …
A: Here we are required to find the decay constant for the given radioactive substance
Q: 4) Draw the MAJOR product of the following reactions. Show stereochemistry when appropriate. Circle…
A: We have to give the major products and also tell the type of the mechanism.
Q: What amount of heat, in KJ , would be involved in condensing 8.8 g of CH3OH? (Hvap = 38.0 KJ/mol)
A:
Q: Which balanced equation represents an oxidation-reduction reaction? Choose the right answer…
A: When oxidation and reduction takes place in the same reaction then it’s called redox reaction. We…
Q: what is acid-base equilibrium or electrochemistry?
A: Acids and bases have a chemical equilibrium in solution. At chemical equilibrium, the products and…
Q: For the following polar bonds, draw the partial positive (8*) and partial negative (8) charges on…
A: Polarity in a covalent bond is due to the electronegativity difference between the combining atoms.…
Q: NABH CH,OH b) 1. Hg(OAc)2, H;0 2. NaBHa H;O, H
A: Regioselectivity is the uni directional reaction.
Q: What is the change in entropy when one mole of calcite (CaCO3) decomposes at 298.2 K to CaO(s) and…
A:
Q: N2(g) + 202(g) → 2NO2(g) AH = +66.4 kJ Complete the table for Reaction 3. Assume the process occurs…
A:
Q: Which is an industrial reducing agent frequently used to reduce iron from its ore? O Oxygen O…
A: Answer: Iron is found in nature in form of oxide ores. During the reduction process we take off this…
Q: Which of the following processes are EXOTHERMIC? Select all correct answers. Hand warmers…
A: The exothermic processes are given below
Q: 20.) Industrial production of methanol uses a synthesis gas which is a mixture of carbon monoxide…
A:
Q: 4. Using the Lewis dot structure, predict the molecular geometry of the following and state the…
A: Hybridization, bond angle and geometry of molecule is depend on number of bonding pair of electrons…
Q: orm hydrogen ions and cyanide ions, the equilibriu constant at 25°C is 5 x 10-10. What does this…
A: Chemical kinetics is branch of chemistry in which we deal with speed at which rate of reaction…
Q: What is the conjugate base in the following reaction?
A:
Q: The equilibrium concentration for a reaction were found to be the following 0.54M [ HI] , 0.5M […
A: Kc is the ratio of the equilibrium concentrations of product over equilibrium concentrations of…
Q: Draw the Lewis dot structure for each of the following molecules or ions. Determine the number of…
A:
Q: What is quantity of heat (in KJ) associated with cooling 212.7 g of water from 25.60 C to ice at…
A:
Q: The concentration of your sodium thiosulfate is 0.520 M. You use a sample of 100 mL of your tap…
A: Given that - Molarity of sodium thiosulfate, Na2S2O3 = 0.520 M Volume of tap water used = 100 mL…
Q: Lalaulate the eH of a 3 lox1D M la/OH),s Selution.CafoH)is. isa strory buse.
A:
Step by step
Solved in 2 steps

- Which compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaFUseful Information: sodium metam: Commercial grade sodium metam is 33% pure sodium metam by weight and has a density of 1.2 g/mL. Hydrolysis of metam (Methyl isothiocynate) + H2S Rate constants for hydrolysis for metam (base catalyzed hydrolysis is negligible) = 300 mol -1 L. sec -1 , = 1 x 10-8 sec-1 Metam also undergoes photolysis to methyl isothiocyanate with a half-life of 1.6 hrs. Data for Sacramento River discharge Q = 75,000 L/min mean depth = 0.30 m pH = 7.8 mean width = 3.2 m dispersion coefficient D = 1.6 x 102 m2/min Assuming the spill acts acts as a single point source. How long does it take for the maximum contaminant concentration to get to Lake Shasta? If Na+ acts as conservative (i.e., it is not transformed) tracer, calculate the maximum concentration (in g/L) at Lake Shasta. (Hint: remember that sodium only constitutes a…Fast pls in 5 min will give u like for sure Create a synthesis of the heterocyclic compound 5 from starting materials each containing no more than six carbon atoms.In the solution you should use retrosynthetic analysis to justify your choice.
- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- ΔH=-75 kJ/mol Rate = k [CH3CH2Br][CH3COO-] Which reaction energy profile would be the best representative of the data provided?Why does the final product has the opposite configuration compared to the reactant? Shouldn’t it form OMs first, then OMs gets substituted by Cl- via Sn2 (the 1st inversion of configuration) then the Cl- gets substituted by OCH3- (the 2nd inversion of configuration? To my understanding 2 inversions = same configurationPlease help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)
- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?What would the final products look like? Pls specify stereochemistry if neededplease quickly thanks ! 3.Please write out the major reaction and side-reaction in the preparation of ter-butyl.chloride, and write out the key points to use separation funnel in this process.
- Please help me with the a proposed Williamson ether sysnthesisi for 3-ethoxyhexane. Thank you !hrydrocarbon tested in bromination rxn room temp. (time to react) elevated temp. (time to react) irradiation (hv) time to reacr toluene 2.10min 26.5 sec 33sec ethylbenzene 49 sec 4 sec 20.9 sec isoproprylbenzene 54 sec 20.3 sec 9.8 sec t-butylbenzene - (no reaction) 27.3 sec 1 min cyclohezane - (no reaction) 20 sec 2.2 min methylcyclohezane - (no reaction) 41 sec 1.22 min Does the order of reactivity you found based on the lab results matches the theory?Explain which compound is the most reactive and why it is the most reactive. Supportyou answer with the relevant structures. use the lab data above*Write out the reaction of Bromination of E-stilbene using chemdraw (ACS 1996 format). Include all reagents, products, solvents, reaction temperature and yield. Reagents used include glacial acetic acid, E-stilbene, and pyridinium perbromide, otherwise known as PHPB. Glacial acetic acid: 10mL Stilbene: 0.5g Perbromide: 1.0g Product yield (Precipitate) weight: 0.63g

