Q: What are the problems encountered if tablets are under or excessively lubricated?
A:
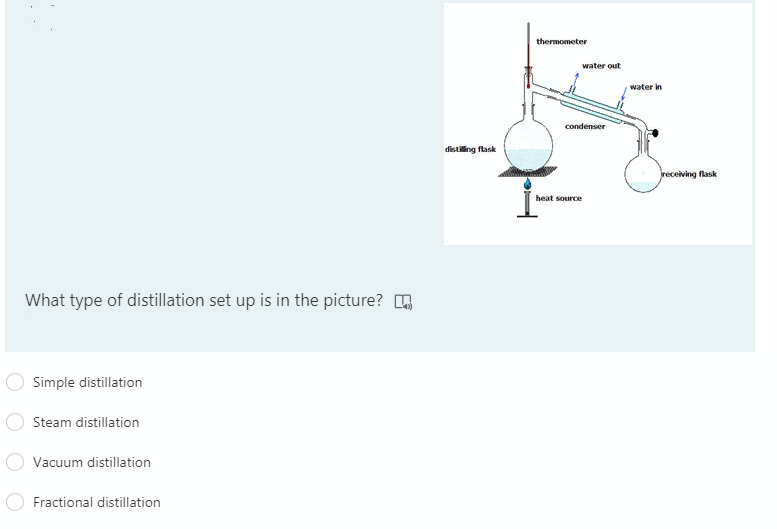
Q: Define Distillation?
A: Various separation techniques are used for the purification of the substance. Purification of the…
Q: you use hand soap (which helps to form micelles). You notice that after washing your hands are no…
A: Soap has cleaning properties . As soap has both polar and non-polar parts.
Q: How were the first soaps made? Who patented liquid soap?
A: The History of soaps is not very clear there are traces of using different materials in a different…
Q: Explain how soap cleans away dirt
A: Soaps are prepared by sponification reaction which includes the raising of temperature of fatty…
Q: B) How the softening of tap water is carried out?
A: Hardness of water is due to the bicarbonate, chloride and sulphates of Mg2+ and Ca2+. This water is…
Q: Difference of toothpastes and tooth powders. What are the basic components of toothpaste? A
A:
Q: What are emulsions? What are their different types? Give one example of each type.
A: When two or more constituents are mixed, it may result in the formation of unlike products. One of…
Q: Write in details about types of gel in pharmacy
A: A gel is a form of semi-solid or solid structure made up of at least two elements that are…
Q: Explain how soaps and detergents work with diagrams or drawings.
A:
Q: What should you do when working with chemicals:? Never return unused chemicals to their original…
A: Various types of chemicals are used in chemistry. These chemicals help in the preparation of various…
Q: Why is soap soluble in water if the starting materials are not?
A: The question is based on the concept of soaps. we have to explain the reasoning for its solubility…
Q: Give three ingredients of personal care products that consumers should avoid. Why?
A: There are many chemical ingredients in some personal care products which affect our skin harmfully…
Q: Describe any visual differences between the hydrated sample and the dried, anhydrous form.
A:
Q: What is the purpose of adding cold alcohol into the banana liquid? What reagent is used in research…
A: The cold alchol added helps to precipitate (DNA undissolves when comes in contact with cold alchol)…
Q: Simple Distillation Cold water out Simple distillation separates 2 with different boiling points.…
A: In this question, we have to fill in the blank given for simple distillation method.
Q: What is an emulsifier? Why do ice cream manufacturers put them in the mix? Explain using concepts…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Which diagram best represents a liquid–liquid emulsionsuch as milk? The colored balls represent…
A: Emulsion is a fine dispersion of droplets of one liquid into another liquid in which it is not…
Q: Which of the following is a suspension? salt water Soda pop (e.g. Sprite, Coke, or Pepsi) Gasoline…
A: Suspension :- When a solute is insoluble in a solvent then it forms a heterogeneous mixture . The…
Q: What type of Cosmetic Product is the below. Describe it in any manner and please identify the…
A: "Since there are multiple sub-parts in this question and it is not mentioned that which one has to…
Q: Complete the Venn diagram below. Write down the similarities (at the middle) and differences (side)…
A: The answer is given below
Q: Mixture 2: Cyclohexane and toluene Boiling point, Boiling point Type of Distillation you will use
A: The boiling point of the following mixture of compounds. Encircle which would boil first. Finally,…
Q: Indicate the most convenient type of distillation to distill each of the following and provide an…
A: A liquid that cannot be distilled of atmospheric pressure is distilled by vacuum distillation.…
Q: unsaturated solution
A: Solubility is the maximum amount of solute which can be dissolved in specific amount of solvent and…
Q: What happened with the balloon when the menthol candy was mixed with the soft drinks?
A: As the menthol candy sinks into the soft drink bottle, the candy creates more carbon dioxide…
Q: artificial sweeteners good or bad?
A: Artificial sweetners are sugar substitutes. It is sweet in taste
Q: How much water is required to raise the moisture content of 100 kg of a material from 30% to 75%
A:
Q: Draw what one molecule of ethanol would look like when dissolved. Show 1 molecule of ethanol and 6…
A:
Q: Sodium formate is a white solid. Draw an atomic/molecular level picture to represent solid sodium…
A: 1)
Q: What do you mean by Solvent Effects ?
A: A solvent is a substance that dissolves a solute, resulting in a solution. A solvent is usually a…
Q: Discuss the cleaning process of soaps and detergents. What is biodegradability as applied to soaps…
A: The cleansing action of soaps and detergents follows the same principle. Soaps and detergents…
Q: Write the appropriate distillation method in the blank of the statement below. i) Separation of…
A: Distillation is the separation technique where different components of a mixture are separated based…
Q: What are oil in water emulsions ? Give examples from daily life
A: Oil in water emulsion means,
Q: Characteristics of a substance that can be separated from a mixture through steam distillation
A: Steam distillation is a distillation or separation process in which steam is generated by distilling…
Q: Why do lactomer stitches dissolve within 2 to 3 weeks following surgery?
A: Lactomer stitches dissolve within 2 to 3 weeks because they are absorbed within the body by…
Q: Purpose of the boiling stones during the distillation process is: a. avoid the formation of bubbles…
A: Small pieces of black porous rock usually made of silicon carbide which are added to solution or…
Q: In a test tube, dissolve 10 drops of methanol in 3 ml. of water. Oxidize a copper wire spiral by…
A:
Q: What should you know about household cleaning products?
A:
Q: What are emulsions? What are their different types? Give an example of each type.
A: The colloidal solutions are the mixtures in which the substances are evenly suspended in a liquid…
Q: define pharmacy
A:
Q: 3 ways to separate water to a liquid mixture and its step by step procedure
A: 1. Fractional distillation is a method for separating a liquid from a mixture of two or more…
Q: Bad2b0-1be8-11e8-8a14-e5ed980bc25f/session cional Distillation Fractional distillation separates a…
A: Fractional distillation ia used to separate the complex mixture of miscible liquids.
Q: True or False? Soaps produce no suds in hard water
A: The solution is given below -
Q: What is the observable result in the test for glycerol? Give the equation involved in the test for…
A: Glycerol has three hydroxyl group attached to the carbon. Glycerol is used as sweetner in food…
Q: What is the boiling point of the antifreeze described?
A: The boiling point of the antifreeze has to be described.
Q: the relative dangers of wrong and unapproved
A: Overdose consumption of alcohol leads to severe effects.
Q: Explain what would happen in a distillation if a student did not add any boiling stones to the…
A: If u will not add boiling stone in your distillation flask the solution starts bumping resulting…
Step by step
Solved in 2 steps with 1 images

- Write the appropriate distillation method in the blank of the statement below. i) Separation of components with similar boiling points is required a ...(a)... distillation.ii) Isolation of water-insoluble components often with high boiling points from plant materials is required a ...(b)... distillation.iii) Separation of components with very different boiling points is required a ...(c)... distillation.iv) Separation of components with high boiling points is required a ...(d)... distillation.Please answer the question at the bottom. Fractional distillation Measure 55 mL of 40% ethanol Place 50 mL of ethanol in your distilling flask taking care that no liquid enters the side arm. Place the remaining 5 mL of ethanol in a test tube and set aside for procedure 2. Add a few marble or porcelain chips to the distilling flask to prevent bumping. Bumping is a term given to irregular boiling Assemble the rest of the apparatus for a simple distillation set-up. You may ask assistance from your instructor Cover the top of the distilling flask with a cork fitted with a thermometer. Make sure that the tip of the thermometer bulb is just below the side arm of the distillation flask Check all connections for tightness Heat the contents of the flask to boiling. Adjust the flame when the sample is already boiling. Make sure that it does not boil to dryness. Note the temperature of the first drop. Collect 60 drops of distillate per test tube. You will need 5 test tubes, thus five…Define Distillation?
- Compare and contrast simple and steam distillation in terms of the type of mixtures separated by each technique.Purpose of the boiling stones during the distillation process is: a. avoid the formation of bubbles b. prevent overheating of the solution c.help the liquid heat up faster d. help separate the components of the mixtureWhen should you NOT use simple distillation? Two liquids with boiling points less than 30 °C apart. The liquid has no more than 10% liquid contaminants. One of the liquids is non-volatile. Two liquids with boiling points more than 30 °C apart.
- Are each of the following things good or bad in a distillation? (a) The distillation stops at one point, and the temperature drops. (b) The material in the distilling flask boils violently and some sloshes up and into the condenser (c) You forget to run water through the condenser. (d) Distillate drops out of the condenser at the rate of about one drop per second.A sample of crude oil undergoes fractional distillation with _____ being collected at the bottom of the distillation tower, and _____ at the top. a. Diesel and jet fuel; gasoline b. Gasoline; kerosene c. Methane and ethylene; diesel and jet fuel d. High molecular weight fractions; methane and ethylene e. Kerosene; high molecular weight fractionsA natural product (MW = 150) distills with steam at a boiling temperature of 99°C at atmospheric pressure. The vapor pressure of water at 99°C is 733 mmHg. Calculate the weight of natural product that co-distills with each gram (1 g) of water at 99°C.
- Chemicals AmountBenzophenone 0.523 g (2.87*10^-3mol)Sodium borohydride 0.12 g (3.2*10^-3 mol)Benzhydrol 0.357 g (1.94*10^-3mol)Observed melting point 65-66 degree C Theroretical yield of Benzophenone ________ Percent yield _______________ Theoretical melting point __________--Please answer both questions.1. Arrange the following steps based on the order they occur in a distillation process:I. The vapor condenses with the aid of flowing cold water in the condenserII. The liquid is collected in the receiving flask, removing the first 1 mL of the distillateIII. The mixture is heated in the distilling flask while simultaneously monitoring the temperatureIV. The more volatile component evaporates and passes through the three-way connector2. The components of an 80-mL rubbing alcohol sample were separated using distillation and the resulting percent recovery of isopropyl alcohol is 14.70%. Determine the volume, in mL, of isopropyl alcohol present in the sample. a. 11.86 mL b. 11.76 mL c. 11.23 mL d. 12.45 mLWhich of the following pairs of liquids could be separated by simple distillation? Why? a. Ethyl acetate (b.p. 77°C) & Phenol (b.p. 181.7°C) b. Cyclohexane (b.p. 80.7°C) & Toluene (b.p. 111C) C. Hexane (b.p. 69.10C) & Ethylbenzene (b.p. 136°C)



