Q: Choose the reagent(s) that would be most likely to complete this reaction. I I I NaBr Br
A:
Q: 2. The gas pressure in a can is 2.5 atm at 25 °C. Assuming that the gas obeys the ideal-gas…
A: ♣ From Ideal Gas Law equation, PV = nRT…
Q: Determine the mass of 0.0146 mol KOH mass = ___ g Determine the mass of 10.2 mol ethane (C2H6)…
A:
Q: A sample of nitrogen inside a rigid, metal container at 20.0ºC is placed inside an oven whose…
A:
Q: What reagents are necessary for the following transformations? ⁰- - (c) CH₂Br one. H (b) (d) -OCH₂…
A: For transformation, one should has good knowledge of reagents. Such as for transformation from C=O…
Q: OH Br₂ H₂C H₂C CH₂. Br
A:
Q: What is the IUPAC name of this molecule? OH A/ 신
A:
Q: ▼ Part A The forensic technician at a crime scene has just prepared a luminol stock solution by…
A:
Q: How many grams of Kr are in a 1.83 L cylinder at 65.4 ∘C and 6.87 atm?
A: Ideal gas equation is the equation which obeys gas laws under all condition of temperature and…
Q: Show all the steps and labels for full credit. 8 Propose a structure for the following: C6H13Br. NMR…
A:
Q: ZI 1. NaH 2. CI
A:
Q: Why is it important to take both the polarity of the bonds and the VSEPR shape of the molecule into…
A: VSEPR theory is known as valence shell electron pair repulsion theory,which explains the molecules…
Q: What is the pH of a 0.00830 M solution of HCI?
A:
Q: 0 OH O || --CH-C H --0 NAD*
A: The Concept used here is from oxidation and reduction reactions Here we have... NAD+ which is an…
Q: A molecular material has two non-degenerate energy states, a ground (ground) state and an excited…
A: This is a simple problem of Boltzmann distribution where distribution of particle between two states…
Q: A chemist prepares a solution of copper(II) sulfate (CuSO4) by measuring out 15. g of copper(II)…
A:
Q: Write the formula of the conjugate acid of CH₂CO₂.
A: Answer:- This question is answered by using the simple concept of conjugate acid and conjugate base.…
Q: SiO₂ (s) + 2 C(s) → SIC(s) + CO(g) If 100.0 kg of sand reacts with 100.0 kg of C, and 55.0 kg of SiC…
A:
Q: Oxidation - Reduction Reactions Complete and balance the following half-reactions. In each case…
A: Oxidation: It involves loss of electrons Reduction: It involves gain of electrons
Q: Part A If you had excess chlorine, how many moles of of aluminum chloride could be produced from…
A: Aluminium (Al) react with chlorine gas (Cl2) to produce aluminum chloride (AlCl3) Given mass of Al…
Q: 0.2 ppm d- 2535.4 ppm .5543 grams of Na,CO, (MW-105.9 g/mole) are placed in a 500.0 mL volumetric…
A: Given that, 0.5543 g of Na2CO3 are placed in a 500.0 mL volumetric flask. The molar mass of Na2CO3…
Q: fod + H₂O-> a. Draw the structure of the tetrahedral intermediate INITIALLY FORMED in the reaction…
A: Acid anhydride reacts with water to give corresponding carboxylic acids. O-atom of a molecule of…
Q: What volume of carbon dioxide (in L) will 3.10 g of antacid made of calcium carbonate produce at…
A: Given, Mass of CaCO3 = 3.10 g. Temperature = 370C = (37 + 273) = 310 K. Pressure = 1.00 atm.
Q: What is the charge of the second product complex (not just the metal center) in the following…
A: Electron transfer (ET) occurs when an electron relocates from an atom or molecule to another such…
Q: Consider the reaction: PO ()+6H₂O(l)-> 4H,PO, (aq) Using standard thermodynamic data at 298 K,…
A:
Q: Data Table and Results (Part B - Determining Solution Concentration) Measurement Volume of Solution…
A: In the given question we have to calculate the following value, i.e mass of solution, mass of solute…
Q: Use the Born-Haber cycle and the data shown to calculate the lattice energy of SrO. Express your…
A: This question is based on calculation of lattice energy by using Born -Haber cycle. In this question…
Q: 1.6 A potassium permanganate solution with a concentration of 0.2834x10-5 exhibits a transmittance…
A: Beer's law states that the absorbance is directly proportional to the concentration of the absorbing…
Q: 1. Which among these would be the most stable carbocation? a. Allylic b. Vinylic c. Primary d.…
A: We have been asked two questions.In one question, we have been asked which is the most stable…
Q: 1) For the following sets of data 0.1065, 0.1991, 0.1016, and 0.1018. The Standard deviation is: -…
A: Standard deviation is a measure of how member of a group differs or deviates from the actual mean…
Q: Give the iupac name
A: Rule of IUPAC- 1) Choose the longest carbon chain as parent chain. 2) Numbering start from those…
Q: Rank the following substances in order of increasing boiling point: F2, Ne, He, Cl₂ He < Ne < F₂ <…
A: Boiling point of compounds depend on the bonds and attractive force present in the molecules.
Q: EXERSICE 1 Calculate: a) the thermal De Broglie wavelength and b) the transport contribution to the…
A: Given= Temp. ---> 4000k Atomic mass of Xe= 131.29 Side of cubic = 2 cm
Q: Draw the structure(s) of the major organic product(s) of the following reaction. NH₂ NaNO₂ aqueous…
A: Reactions are given below
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of…
A:
Q: Write the equilibrium expression: ((s) + C(s) + 2H2 (g) (H 4 (g)
A: For a reaction aA(g)+bB(g)⇔cC(g)+dD(g) pressure-based equilibrium constant can be written as…
Q: When the reaction below is balanced under acidic conditions, what is the coefficient in front of the…
A:
Q: What volume in (mL) would 0.0709 moles of gas held under 1.40 atm pressure and 461 K occupy?
A:
Q: Complete the balanced neutralization equation for the reaction below: H₂SO (aq) + Mg(OH)₂(aq) -
A:
Q: 3. Explain the regioselectivity of the nucleophilic attack of the chloronium ion below. Compare it…
A:
Q: Consider the mechanism. Step 1: Step 2: E Overall: A+ D B+E Determine the rate law for the overall…
A:
Q: QUESTION 1 The initial volume of a sample of argon gas is 766 mL at 124°C. If the pressure is held…
A: Initial volume = 766 ml Initial temperature = 124 °C New volume = 941 ml New temperature (in °C)…
Q: A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 2Cr³+ (aq) +…
A:
Q: Question 7: Draw the products formed when each alkene is treated with O3 followed by Zn, H₂O.
A: When alkene is treated with ozone and zn/H2O then ozonide is formed which breaks into aldehyde or…
Q: Write the formula for the conjugate base of HSO. formula for the conjugate base of HSO: HSO + H+…
A: The given problem is based on the concept of conjugate acid-base pair.
Q: fischer esterification: 5.4mL of isopentyl alcohol, molar mass 88.15g/mol: 8.5mL of acetic…
A: Given that the Isopentyl alcohol is the limiting reactant. Volume of isopentyl alcohol is 5.4 mL
Q: A jar holds 1.65 L of ideal nitrogen gas (N2, which is a diatomic gas) at -0.86 C and at pressure…
A:
Q: Choose the reagent(s) that would be most likely to complete this reaction. OsO4 (catalytic) NMO OH
A:
Q: A: unhindered carbon Route B: hindered carbon For a small lab which route is better for a Grignard…
A: Grignard reagent acts as nucleophile during conversion of aldehyde/ketone to alcohol. Grignard…
Q: Explain what scientific principles you used in making this prediction and how this supports your…
A: Highest melting point - CaO Melting point of the molecules is given below--> NaCl - 801°C CaO -…
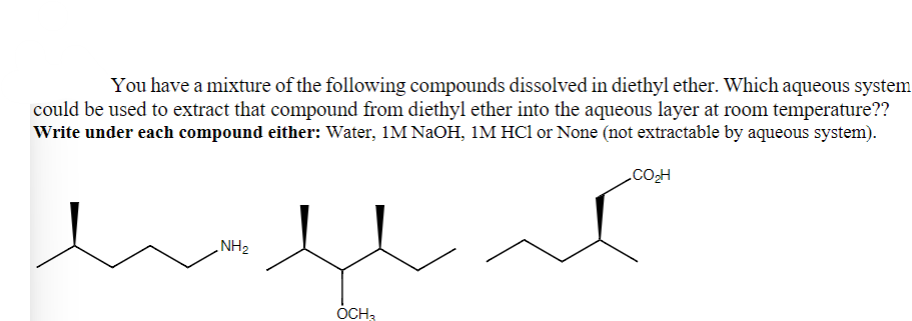
What type of question is this and what is it expecting me to do?
Step by step
Solved in 4 steps with 7 images

- You are employed as a coop student at the Drug and Alcohol Testing Association of Canada (DATAC) developing analytical tests for sports doping agents. You are asked to prepare a procedure for the extraction of methylphenidate, the active compound in Ritalin, from urine samples (consider them as simple aqueous layers, you do not need to consider other components!). The goal of the procedure is to extract the methylphenidate into an organic layer which will then be evaporated, and the residue will be tested for the drug.You find that methylphenidate is highly soluble in 2-methyltetrahydrofuran, a bio-renewable solvent. Draw the structure of 2-methyltetrahydrofuran and give two reasons why it is a good solvent choice for liquid-liquid extraction.You are employed as a coop student at the Drug and Alcohol Testing Association of Canada (DATAC) developing analytical tests for sports doping agents. You are asked to prepare a procedure for the extraction of methylphenidate, the active compound in Ritalin, from urine samples (consider them as simple aqueous layers, you do not need to consider other components!). The goal of the procedure is to extract the methylphenidate into an organic layer which will then be evaporated, and the residue will be tested for the drug.Your colleague is helping you develop the urine test. They suggest that the urine should be adjusted to a pH above 7 before extracting with the organic solvent. Why is this necessary? Support your explanation with a full arrow-pushing mechanism for the reaction that would occur if the pH was below 7. Include all formal charges, intermediates (if applicable) and products.A chemist is asked to separate the components of a mixture composed of 1.00 g each of the two compounds shown below. The chemist has access to the following bases: 10% aqueous NaHCO3 and 10% aqueous NaOH. Which base and how many mLs of the base (i.e., the minimum amount) are needed to completely separate these compounds from each other by liquid-liquid extraction?
- Naphthalene, C10H8 is insoluble in water but sublimes easily when heated. How could you separate a mixture of naphthalene and NaCl? Suggest two different methods. asapSuppose you have a mixture of copper sulfate (CuSO4) and azulene (C10H8, you may wish to know the structure of azulene). Both copper sulfate and azulene are beautiful deep blue cystalline solids. If you transferred the mixture to a separatory funnel containing both an aqueous and and organic solvents (1M HCl and dichloromethane), into which layer would the copper sulfate partition, aqueous or organic? Aqueous layer (1M HCl) Organic layer (dichloromethane) Copper sulfate would partition nearly equally into both the aqueous and organic layersIsopropanol is a good extraction solvent for plant pigments as it breaks down cell walls and is miscible with pigments. Can you be sure that you only extracted chlorophyll-a from your spinach leaf? If not, what else might have been extracted?
- Describe in words and by means of complete chemical equations, how a mixture of 1.25 g of benzoic acid tertbutylbenzene can be separated by applying the separation of 2 components by liquid-liquid extraction.Suppose you want to determine the eutectic melting point of a eutectic mixture of adipic acid (literature MP 153 degree C) and salicylic acid (literature MP 158 degree C) .What would you do and what would expect to observe? Be sure to include a very specific detailed plan that any inexperienced organic chemistry lab student could follow if they were to use your procedure.We want to extract terpenoids from an aqueous sample by continuous liquid-liquid extraction. Which of the following solvents is not suitable for this purpose? Why? Propanone (acetone), dichloromethane, heptane.
- 1. An organic student did an experiment extracting benzoic acid from a mixture of other organic substances. Other analyses of the obtained sample confirmed the presence of benzoic acid. The student also measured the melting point of the white solid, 117.4-121.6 oC. The literature mp of benzoic acid is 122.3oC. What can you conclude about the sample? Select one. Group of answer choices The sample of benzoic acid contains soluble impurities. The student obtained a good yield of benzoic acid. The sample of benzoic acid contains insoluble impurities. The student obtained pure benzoic acid from the extraction. The student obtained a poor yield of benzoic acid. 2. Which phenomenon are sometimes confused with melting during mp analysis? Select all that apply. Group of answer choices decomposition cracking shaking sagging/shrinking sweating solidifying 3. Study the bond types in the compound shown. Select the type(s) of intramolecular force(s) you predict the substance to exhibit. Select all…A student is given a solid mixture containing 2 polar components and 1 nonpolar component. The student is asked to separate the nonpolar component by solid-liquid extraction. Which of the following solvents is the most appropriate to use for the extraction? Group of answer choices methanol (CH3OH) H2O hexane (C6H6) ethanol (CH3CH2OH)A bloodhound, with an acute sense of smell, sits at one end of a long, unventilated corridor.At the other end of the corridor a small amount of an aerosol is released containing the volatile, fragrant compounds: acetaldehyde (C2H4O) jasmine lactone (C10H16O2) acetoin (C4H8O2) hexyl acetate (C8H16O2) 1-octen-3-one (C8H14O) Rank the compounds in the chronological order they are detected by the hound. 1-octen-3-one acetoin jasmine lactone hexyl acetate acetaldehyde



