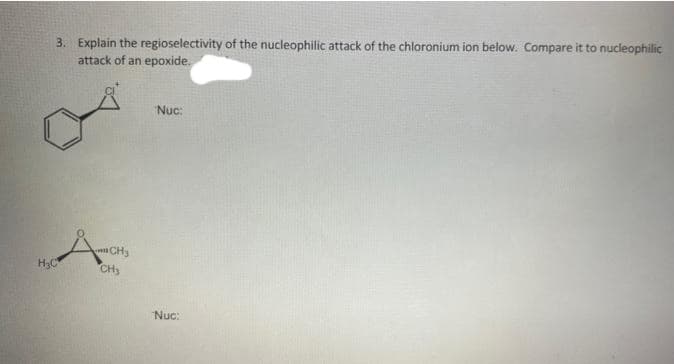
3. Explain the regioselectivity of the nucleophilic attack of the chloronium ion below. Compare it to nucleophilic attack of an epoxide. H₂C Десня CH₂ Nuc: Nuc:
Q: 7) Consider the following reaction: liter mol-sec 2 HI(g) → H₂(g) + I2 (g) Initially, a flask…
A: Initial concentration of HI= 0.670 M Equilibrium concentration of HI= 0.125 M Equilibrium constant…
Q: Part B A gaseous system undergoes a change in temperature and volume. What is the entropy change for…
A:
Q: Calculate the pH of a 5.8 × 10-8 M HCl solution. Report your answer to the hundredths place. pH =…
A: We have to calculate the pH of the 5.8×10-8 M of HCl solution. Also, we have to calculate the…
Q: Oxidation - Reduction Reactions Complete and balance the following half-reactions. In each case…
A: Oxidation: It involves loss of electrons Reduction: It involves gain of electrons
Q: How many stereoisomers are possible for this molecule? 08 16 02
A: Chiral carbon is a carbon atom which are attached with four different groups
Q: Under constant-volume conditions of heat of combustion of glucose,C6H₁2O6, 1s 15.57 KJ/g. A 2.500-g…
A: Given, Heat of combustion of glucose = 15.57kJg mass of glucose = 2.5 g
Q: The vapor pressure of liquid aluminum is 400. mm Hg at 2.59x10³ K. Assuming that AHvap for Al (296…
A: we have to calculate the vapor pressure of aluminium using Clausius Clapeyron equation
Q: Which compound has the lowest melting point? O NaCl O CaO O KBr O BaO
A:
Q: Complete the following nuclear equations and identify X in each case: b) ?1940→ ?−10+? d)…
A: These are nuclear reactions based on total atomic number and masses on both reactant and product…
Q: Provide the reagents that would accomplish the reaction below. R O 1. NaBH CN, CH₂OD 2. HO, H₂O O 1…
A:
Q: 11. sodium phosphide 12. diphosphorus pentoxide 13. potassium nitride 14. sodium oxide 15.…
A: -> Ionic compound is formed due to attraction of cation and anion . -> Ionic bond is formed…
Q: Obtain the electron configuration of the element with 21 electrons using pauli's exclusion principle
A: First, we need to check the filling of electrons in the orbital and then we need to draw its box…
Q: cis-1,2-dimethylcyclopentane and trans-1,3-dimethylcyclopentane are geometric isomer (circle one) to…
A: We know in order to be isomers, their molecular formula must be the same. There are various types of…
Q: 16. silver nitride 17. cesium fluoride 18. chlorine dioxide 19. iron (III) oxide 20. cobalt (II)…
A: Ionic compound are those compounds which are formed between metal and non -metals when metal…
Q: Which substance is the oxidizing agent in this reaction? Express your answer as a chemical formula.…
A: Oxidizing agent: Molecule or atom which oxidize other and get reduced.. Reducing agent: Molecule or…
Q: What kind of intermolecular forces act between two carbon dioxide molecules? List all intermolecular…
A: we have to determine the intermolecular forces present in CO2
Q: 16.26 Indicate whether each of the following statements describes primary, secondary, tertiary, or…
A: Primary protein structure:- The basic peptide structure between the -NH2 and -COOH groups of two…
Q: Substance 48f (2/11101)| M₂O(s) -10.90 M(s) 0 0₂(g) 0 Consider the decomposition of a metal oxide to…
A:
Q: What do CO, ethylene (C2H4), H2, and NO+ ligands all have in common? Group of answer choices they…
A: Ligand: An ion or molecule, which donates a pair of electrons to the central metal atom or ion to…
Q: A scientist wants to make a solution of tribasic sodium phosphate, Na3PO4, for a laboratory…
A: Volume = 675 ml Concentration of Na+ ion = 0.700M
Q: Which of the following would be the strongest acid? 4-Methoxybenzoic acid 4-lodobenzoic acid O…
A:
Q: Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic…
A: •Polar molecules : i) have polar bond. ii) have asymmetric structure. •Non - polar molecules :…
Q: 20 Write the overall molecular equation for the reaction of hydroiodic acid (HI) and potassium…
A: Hydroiodide acid represent as HI Pottasium hydroxide KI
Q: 5. A total synthesis of the marine natural product involved an intramolecular SN2- type process of…
A: SN2 reaction involves inversion in configuration because the nucleophile attacks from the back side…
Q: Predict the product(s) for each reaction below. In each case, draw the resonance forms of the…
A: The electron donating group on the benzene ring directs incoming electrophile to the ortho/para…
Q: a) Predict the products and write a balanced equation, including all physical symbols. b) Write the…
A:
Q: If ice homogeneously nucleates at -37.5°C, calculate the critical radius given values of -3.1 x 108…
A: Given, Ice homogeneously nucleates at -37.5oC
Q: A voltaic cell is constructed in which the following cell reaction occurs. The half-cell…
A: Given , Reaction : 3Cu2+(aq) +2Cr(s) →3Cu(s) +2Cr3+(aq)
Q: How many molecules (not moles) of NH3 are produced from 5.14×10−4 g of H2?
A:
Q: QUESTION 5 A sample of nitrogen at 20.0 °C has a volume of 1442 mL at a pressure of 850 mmHg. To…
A: Given -> Temperature (T1) = 20.0°C = 20.0 + 273 = 293 K Volume (V1) = 1442 ml Pressure (P1) =…
Q: Problem 10.110 Sodium oxide, Na₂O, reacts with water to give NaOH. Write a balanced equation for the…
A: We will first write the balanced reaction. And using that we will find moles of NaOH formed and from…
Q: The carbon from the heartwood of a giant sequoia tree gives 11 ?614 counts per minute per gram of…
A:
Q: Why does HF have the lowest melting point compare to CH4, MgCl2, and c(s, diamond./
A: The hydrogen fluoride have lowest melting point among all because the bond between hydrogen and…
Q: The 3rd Nitrogen from the left has 3 sigma bonds and a lone pair. Isn't that sp3?
A: For hybridization we check the electron groups. If there are 4 electron group on N ( 3 Sigma bond…
Q: Consider the following reaction: 2 CH3OH(g) → 2 CH4(g) + O2(g) ΔH = +252.8 kJ Is this…
A: Given -> 2CH3OH(g) ---> 2CH4(g) + O2(g) ∆H= +252.8 KJ ------(1) Weight of CH3OH = 24.0 gm
Q: What is the 1 letter abbreviation for the tripeptide
A: Given tripeptide - serylglycylalanine
Q: Draw the product of the reaction shown below. Use wedge and dash bonds to indicate relative…
A: Here we are required to predict the product of the reaction when the given alkene undergo…
Q: 0 LOH 1. LiAIH4 2. H2O PCC
A: LiAlH4 is a strong reducing agent .It reduces aldehyde ,ketone , carboxylic acid into alcohol. PCC…
Q: Identify the compound in each pair that has the higher melting and boiling points. Explain why each…
A:
Q: water-soluble collagens insoluble in water peptide chains arranged in Answer Bank often function as…
A: Fibrous protein is insoluble in water whereas globular protein is soluble in water.
Q: 0 OH O || --CH-C H --0 NAD*
A: The Concept used here is from oxidation and reduction reactions Here we have... NAD+ which is an…
Q: Balance this nuclear reaction by supplying the missing nucleus. Replace each question mark with an…
A:
Q: How much heat (in kJ) is needed to convert 10.0 grams of H₂O(l) at 99.97°C to H₂O(g) at 99.97°C?…
A:
Q: Use the observations about each chemical reaction in the table below to decide the sign (positive or…
A: The sign of enthalpy change for an endothermic reaction is positive and that for an exothermic…
Q: 2. Dibuje el tautómero ceto/enol faltante e identifique el tautómero más estable. os & OH 1
A:
Q: A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 2Cr³+ (aq) +…
A:
Q: Calculate the volume of a balloon that could be filled at 1.00 atm with the helium in a 30.2 L gas…
A: Boyle ' s law - At constant temperature volume of a given mass of a gas inversely proportional to…
Q: If propanal is mixed with an oxidizing agent, then what is the IUPAC name of the product? A
A: When propanal is treated with an oxidising agent then the propanal is oxidised to form the propanoic…
Q: Redox titrations are used to determine the amounts of oxidizing and reducing agents in solution. For…
A: Given, Volume of 1.68 (M) of KMnO4 required for titration = 14.3 mL = 0.0143 L
Q: In an experiment, 27.5 g of metal was heated to 98.0°C and then quickly transferred to 150.0 g of…
A: we have to determine the specific heat of the metal
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- rank these from least to most reactive in nucleophilic acyl substitution with a nucleophile I)CH3COOC2H5 II) CH3COO-Na+ III)CH3COCl IV) CH3CONH2Why is this considered an Elimination E2 reaction? C₃H₈O (l) → C3H6 (g) + H2O (l) propan-1-ol → propene + waterRank in terms of reactivity towards the nucleophile. 1=least 4=most
- What is the best reagent for carrying out the reaction shown below?a,b-Unsaturated carbonyl compounds are susceptible to either 1,2 addition or 1,4 (Michael)addition. Give examples of nucleophiles that would favor each case and explain yourreasoningWhat are the relative rates of the following nucleophilic substitution reaction below.
- Illustrate the resonance effect of the methoxy group -OCH3, on the structure of the benzene ring. Draw all the oissuvke resonance forms of methoxybenzene, including the hybrid Based on the structures, explain how the presence of the -OCH3 group affects: (i) the reactivity of the benzene ring towards electrophilic attack (ii) the orientation or point of attack of an incoming electrophilic reagent on the benzene ring.Arrange these compounds in increasing order of reactivity in SN2 nucleophilic substitution?Rank the primary, secondary, tertiary, and aryl halides in terms of reactivity with the nucleophile. Which type of halide react faster by an SN2 reaction? Explain
- Trimethylphosphine is a stronger nucleophile than triphenylphosphine, but it is rarely usedto make ylides. Why is trimethylphosphine unsuitable for making most phosphorus ylides?Alkyl diazonium salts (shown below) are considered "super" leaving groups; a consequence of this is that they tend to be contact explosives What quailities make alkyl diazonium salts such excellent leaving groups?Enolates are formed by deprotonation of an α-carbon hydrogen. Answer the following questions about enolate formation. In the molecule shown, select the α-carbon hydrogen that would be removed to form an enolate when NaOEt is used as a base. Draw the thermodynamic enolate that results for the molecule in Part 1. Draw only the enolate resonance form that includes a formal charge on the α carbon. Be sure to indicate that formal charge as well as any lone pair of electrons in your answer.

