Q: on the ions they produce when dissolved in water, classify the following salts as acidic, basic or…
A: In order to categorize the salts as either acidic, alkaline, or neutral depending on the ions they…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. OH HO OH Na2Cr2O7…
A: Na2Cr2O7 is a strong oxidising agent. Na2Cr2O7 in presence of acetone and sulphuric acid, oxidises…
Q: Please don't provide handwritten solution ....
A: Answer: IUPAC name of the given compound is Hexan-1-olExplanation:Step 1: While writing the IUPAC…
Q: 34.6 mL sample of a 0.459 M aqueous nitrous acid solution is titrated with a 0.261 M aqueous barium…
A: Given that, volume of nitrous acid(HNO2) = 34.6 mLmolarity nitrous acid(HNO2) = 0.459 MMolarity of…
Q: 8. Draw a reaction coordinate diagram depictin Indicate the locations of any starting materials,…
A: The reaction follows the following path:
Q: Consider the cylcohexane, shown left as a line structure. Which of the following statements is…
A: The most stable conformer of a cyclohexane derivative is the conformer having least 1,3-dixial…
Q: Telluric acid, H6TeO6 is a diprotic acid with Ka₁ = 2.1×10-8 and Ka2 = 1.0x10-11, Determine the pH…
A: The objective of this question is to calculate the pH of a 0.35e-2 mol/L aqueous solution of…
Q: Plese don't provide hand writtin solution....
A: The required answer is given below Explanation:Step 1: Step 2: Step 3: Step 4:
Q: Based on the activity series, which one of the reactions below will occur? Select one: O a. 3Hg (1)…
A: The objective of the question is to determine which of the given reactions will occur based on the…
Q: A sample of solid titanium is heated with an electrical coil. If 107 joules of energy are added to a…
A: mass of titanium = 11.4 gFinal temperature(Tf) = 38.0oCenergy added(q) = 107 Jspecific heat capacity…
Q: 5. Propose a synthesis of each of the following compounds using the indicated starting material. You…
A:
Q: Name the alkene. Be sure to indicate stereochemistry and use hyphens (-) not endashes (–). H2C—CH3 H…
A: Find the highest functional group containing the longest carbon chain.Replace the end of the parent…
Q: What is the predicted product of the reaction shown? excess CH3CH2SH Raney H2SO4 Ni A) B) OH ☎ D) E)
A: The objective is to determine the product formed by the given reaction. Ketone on reaction with…
Q: Identify the expected major product of the following reaction. OH H3O* H₂O III O IV OV OH Хон ☆ OH…
A: The given reactant is an alkene since it has a carbon-carbon double bond. Now, when an alkene reacts…
Q: OH I+ not main H₂O, EtOH lennul D H
A: An aromatic electrophilic substitution is a reaction in which an electrophile replaces the proton of…
Q: Ringlemann scale is used to analyze O a. Carbon monoxide Ob. Nitrogen dioxide O c. hydrocarbons O d.…
A: The objective of the question is to identify the substance that the Ringlemann scale is used to…
Q: Macmillan Learning A solution contains 0.0350 M Co2 + and 0.0250 M Ni2+. What is the concentration…
A: The question is based on the concept of solubility product principle. It states that when a weak…
Q: Please don't provide hnad writtin solution....
A: The reaction is an example of intra-molecular aldol condensation reaction which is shown in the…
Q: 1. Select the compound with the largest HOMO-LUMO energy gap. 2. Select if the compound in part 1…
A: In the given question we have to determine which of the given compounds has the largest HOMO-LUMO…
Q: Be sure to answer all parts. A voltaic cell consists of an Mn/Mn²+ half-cell and a Pb/Pb²+…
A: The cell potential The concentration of the ion, We have to calculate the concentration.
Q: With the aid of a phase diagram, explain what is meant by a supercritical fluid. Give examples of…
A: The objective of the question is to understand the concept of a supercritical fluid, its…
Q: The elementary reaction below is 2nd order with respect to NO2. The rate constant at 501 K is 7.93 ×…
A: t1/2 = t1/2 - Half life k - Rate constant[A0] - Initial concentration of reactant
Q: Table 7.2.Calculated and observed pH of ammonia during neutralization Solution 50mL 0.10M NH3 A)…
A: The objective of the question is to understand the changes in pH during the neutralization of…
Q: Draw the mechanism for the initiaion of the polymerization of the monomer n-butyl acrylate that is…
A: In the given question we have to draw the mechanism for the initiation of the polymerization of the…
Q: and CH3CH2CH2CH3 O structural isomers O conformational isomers O not related Submit Request Answer…
A: Molecules having the same molecular formula but different structural formula are known as…
Q: How many milliliters of 8.50\times 10-2 MNaOH are required to titrate each of the following…
A: Given Information:Determine the volume of required.
Q: The following is a key step in the synthesis of morphine involving the conversion of A to B. Draw a…
A: Morphine is an organic compound with the molecular formula of and commonly employed as a medication…
Q: The mono bromination of the following compound would be expected to give... NO2 NO2
A: The monobromination of benzene ring takes place in the presence of Lewis acid like FeBr3. This type…
Q: Many household cleaning products contain oxalic acid, H2C2O4, a diprotic acid with the following…
A: Given ,
Q: A 8.00 L tank at 11.8 °C is filled with 7.85 g of carbon dioxide gas and 3.51 g of boron trifluoride…
A: Volume = 8.00 Lmass of CO2 = 7.85 gmolar mass of CO2 = 44.01 g/molmass of BF3 = 3.51 gmolar mass of…
Q: Write the concentration equilibrium constant expression for this reaction. 3+ 312(s)+2 Cr³+…
A: The equilibrium constant for a particular reaction is the ratio of the concentration of the aqueous…
Q: Provide the product of this reaction ? Look at the starting sugar: Should it be an alpha-sugar or…
A: The product is condensation between two reactants. The starting material is beta-sugar Explanation:…
Q: Wolff-Kishner reduction of 7-methyl-2-cycloheptenone produces 3-methylcycloheptene, not…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Which of the following statements is right about the below compound? O The above compound is not…
A:
Q: Part: 0/2 Part 1 of 2 Rank the three compounds in order of increasing heat of hydrogenation. The…
A: Heat of hydrogenation ΔHhydro of an alkene is the standard enthalpy of catalytic hydrogenation of an…
Q: Provide the structure of the ozonolysis product of the most stable alkene from Part 1 shown below.…
A: This is ozonolysis reaction, In this reaction C=C bond breaks and forms corresponding carbonyl…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the alcohol side product.…
A: DIBAL- H = diisobutylaluminium hydride is a well known reducing agent.It reduces esters into…
Q: 4. ) What are the aldol condensation products of this reaction? Why might this not be a very useful…
A: We have been provided with reactants in which both have alpha hydrogen and have tendency to form…
Q: 5. Using the periodic table, classify each of the following elements as a metal or a nonmetal, and…
A: a)The atomic number of Co is 27Electronic configuration of cobalt is Co: 1s22s22p63s23p64s23d7It can…
Q: At 560 K, AG = -18.1 kJ/mol for the reaction A (g) + 2 B (g) → 3 C (g). If the partial pressures of…
A: Given,
Q: Order the following alkene in order of increasing stability: (1) (11) (111) (IV) O III, I, IV, II O…
A: We have to choose the correct option.
Q: (b) An aqueous solution of propanone (acetone, 1) of total volume 0.120 mL was analysed by…
A: b-i) the mass of propanone (1) contained in the original aqueous solution is approximately 1.05 mg.…
Q: At 25 °C, the relationship involving acids and bases is, [H3O+][OH-] = 1.00 × 10-14 True False
A: At 250C for pure water,[H3O+] = 1.0 × 10-7 M[OH-] = 1.0 × 10-7 M
Q: Please don't provide handwritten solution ....
A: The mass will land at 1.56 meters from the ramp's end.Explanation:Step 1:We may utilize the…
Q: Fill in necessary products reactants or reagants of these reactions. Please note the existence of…
A: please see the detailed explanation below.Explanation:
Q: OCH3 H H CHO
A: Prediction of the correct IUPAC names is : Rules for IUPAC naming :-Select the longest chain which…
Q: Please don't provide hand writtin solution....
A: Equilibrium molarity CO2of will be 0.45743 MExplanation:Volume is 500mL or 0.5LMolarity of…
Q: Should a G-M detector be used to measure the exposure rate in the vicinity of a storage safe for…
A: A Geiger-Muller (G-M) detector is a common type of radiation detector that detects the presence and…
Q: T 5.5 75 4.85 5.0 7.5 7.0 4.86 4.5 2.04 4.0 C10H12O2 3.5 3.0 (c) 7.0 6.5 6.0 0.97 2.05 C10H12O2 5.5…
A: 1 ) NMR spectrum of C10H12O2 :At 4.25 ppm, a 2H signal is observed which is influenced by an…
Q: A scientist measures the standard enthalpy change for the following reaction to be - 1141.6 kJ:…
A: The standard enthalpy change (ΔH) during a given reaction can be calculated from the enthalpy of the…
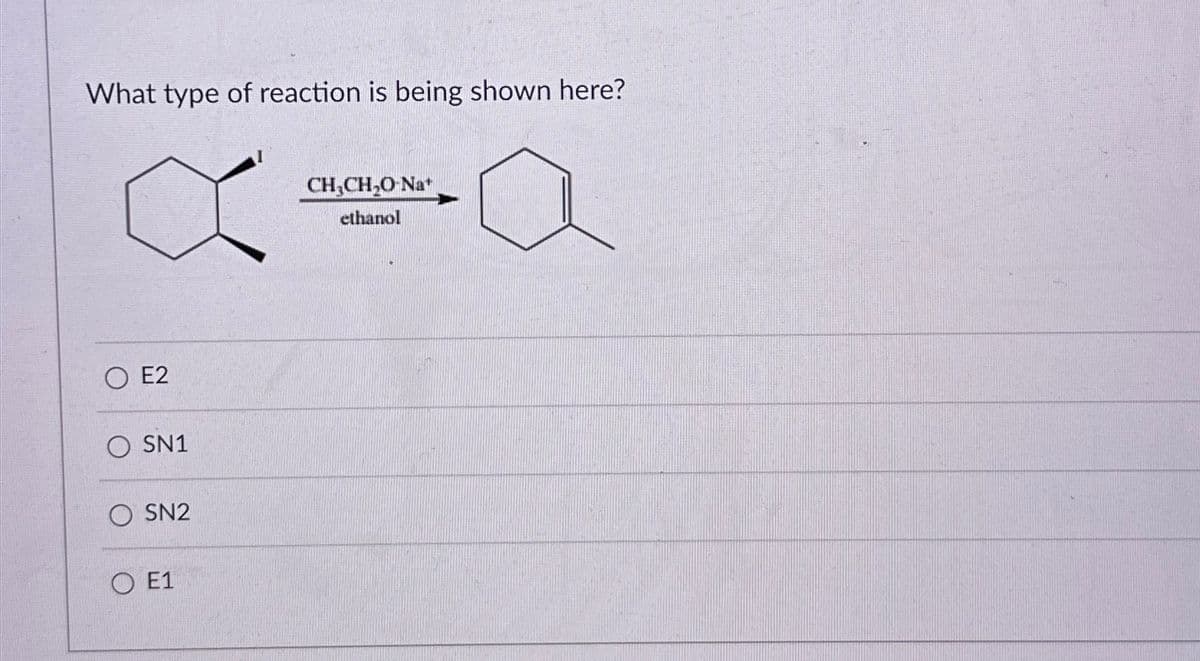
What type of reaction is being shown here? E2 SN1 SN2 E1
Step by step
Solved in 3 steps with 2 images

- The -N(CH3)3+ group is considered a .... Ortho/Para Directing Activator Ortho/Para Directing Deactivator Meta Directing DeactivatorCyclohexanone do not produce isomeric oximes but cyclohexane does produces 2 stereoisomeric oximes. Explain whyWhich of the following will have the strongest IMFs? CH3OH with H2O CH4 with CH2Cl2 HCl with CH2Cl2 CH3OH with CH2Cl2
- Indicate the product of each reaction belowselect the most appropriate reagent(s) to effect the change. K2Cr2O7, H+ H2, Pd 1. Disiamylborane, 2. HO–, H2O, H2O2 NaOCl H2SO4, HgSO4Consider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:

