Q: What kind of intermolecular forces act between Note: If there is more than one type of intermo
A: Dipole - dipole interaction can be present among molecules which contains a permanent dipole. Only…
Q: The intermolecular forces in chloroform are than the intermolecular forces in methanol.
A: The graph is drawn between vapor pressure and temperature of different compounds (carbon disulfide,…
Q: What are the intermolecular forces found in Chlorophyll? What are the majority of intermolecular…
A: A green colored green pigment is present in the plants. This helps them in the process of…
Q: Identify the compound that ABLE to form hydrogen bonding with each other? но
A:
Q: What kind of intermolecular forces act between a hydrogen peroxide molecule and a water molecule
A: The strongest intermolecular force between a water molecule and hydrogen peroxide molecule is…
Q: What kind of intermolecular forces act between a dichloroethylene (CH,CCl,) molecule and a…
A: The type of interactions present in the molecules depends on the polarity of the molecule. The C-Cl…
Q: . How do intermolecular forces differ from bonds? What are the three major intermolecular forces for…
A: Topic-- 1.Difference between Intermolecular forces and chemical bonds 2. Discussion of…
Q: Which type of intermolecular attractive force operates between (a) all molecules; (b) polar…
A:
Q: What kind of intermolecular forces act between a hydrogen fluoride molecule and an ammonia molecule?…
A: The given molecules Hydrogen fluoride (HF) Ammonia (NH3) The type of intermolecular forces between…
Q: What kind of intermolecular forces act between a potassium cation and a hydrogen sulfide molecule?…
A: Intermolecular forces are defined as the forces of attraction or repulsion which is present between…
Q: What type(s) of intermolecular forces are expected
A: Intermolecular forces (IMF) are the forces which mediate interaction between molecules, including…
Q: What are the general types of intermolecular forces?
A: The general types of intermolecular forces has to be given.
Q: Consider the intermolecular forces present in a pure sample of each of the following compounds:…
A:
Q: What causes dipole interactions?
A: Dipole - It is basically the separation of charges within a molecule between two covalently bonded…
Q: What is the strongest type of intermolecular forces present in acetone (look up the structure)?…
A:
Q: Answer each of the following questions correctly (This is all about Intermolecular Forces of Liquids…
A: The chemical formula of uric acid is C5H4N4O3, a heterocyclic compound containing hydrogen, carbon,…
Q: What kind of intermolecular forces act between a bromine (Br,) molecule and a tetrachloroethylene…
A: We will estimate the polarity of molecules given and then predict the intermolecular forces present.
Q: What type of intermolecular forces are present in this compound?
A:
Q: How Intermolecular Forces can be Exhibited ?
A: Hydrogen bonding: Hydrogen bond is formed when an electromotive element like F,O and N is combine…
Q: What kind of intermolecular forces act between a hydrogen chloride molecule and a nitrogen…
A: The compounds given are hydrogen chloride i.e HCl and nitrogen trichloride i.e NCl3. Since in HCl,…
Q: How is the volatility of a substance related to the intermolecular forces present within the…
A: intermolecular forces are held elements or atoms together in a molecule. types of intermolecular…
Q: What are intermolecular forces and how they are different than true bonding?
A: The intermolecular forces are explained as the force of attraction or repulsion between the…
Q: What kind of intermolecular forces act between a bromide anion and a hydrogen bromide molecule?…
A:
Q: n one to two sentences describe an experiment that would show that intramolecular forces…
A: Intramolecular force of attraction is the attraction between atoms within the single-molecule but in…
Q: What type of intermolecular forces are present in argon gas, solid, and liquid?
A:
Q: What kind of intermolecular forces act between a hydrogen iodide molecule and a dichloroethylene…
A: Dipole-dipole interactions one pole of the dipolar bond is hydrogen. Since hydrogen is highly…
Q: For the following molecules, list them from low boiling point to high boiling point, and list the…
A: SiH4 PH3 CH3OH
Q: what substance has the stronger intermolecular forces at room temperature ? water or mercury
A: We know water has strong hydrogen bonding and mercury has metallic bonds. Also mercury is liquid at…
Q: What kind of intermolecular forces act between a nitrosyl chloride (NOCI) molecule and a hydrogen…
A: The dipole dipole interaction is a type of intermolecular attraction i.e. the attraction between the…
Q: What kind of intermolecular forces act between a hydrogen chloride molecule and a chlorine…
A: Hydrogen chloride and chlorine monofluoride both are polar molecules.
Q: What kind of intermolecular forces act between two hydrogen chloride molecules? Note: If there is…
A: Intermolecular forces are forces which are responsible for the physical and chemical properties of…
Q: What types of intermolecular forces are exhibited by each compound?
A:
Q: What kind of intermolecular forces act between a hydrogen chloride molecule and a hydrogen sulfide…
A: Intermolecular forces are the forces that hold the atoms together within a molecule . This forces…
Q: What kind of intermolecular forces act between two ammonia molecules? Note: If there is more than…
A: The strength of intermolecular forces decides the bulk properties of substances. London dispersion…
Q: Which type of intermolecular attractive force operates between all molecules polar molecules the…
A: Answer:- This question is answered by using the simple concept of intermolecular forces which act…
Q: What type(s) of intermolecular forces are expected between SCl, molecules?
A: Intermolecular forces are the forces of attraction or repulsion which act between neighboring…
Q: b) Draw a diagram below to showing the strongest intermolecular forces present between molecules of…
A:
Q: What kind of intermolecular forces act between a dichlorine monoxide molecule and a chloroform…
A:
Q: what would our world look like if dispersion forces were the strongest intermolecular forces
A: We know that there are following type of intermolecular forces - (1) Dispersion or London…
Q: What do we mean when we say that a liquid is volatile? Do volatile liquids have large or small vapor…
A:
Q: Identify the intermolecular forces for CH4, H2S, and H2O. Which molecule has the lowest boiling…
A:
Q: Determine the kinds of intermolecular forces that are present in element or compound. I2
A: Inter molecular forces hold molecules or atoms together in liquid or solid. The strength of the…
Q: a compound’s boiling point is a rough index of the strength of its intermolecular forces. True…
A: Boiling point of a compound depends on the intermolecular force. Greater is the intermolecular…
Q: What is viscosity? For a liquid, it's viscosity decreases as temperature increases. Explain it in…
A: Viscosity is a property of fluids such as liquids and gases. SI unit of viscosity is poiseiulle…
Q: What types of intermolecular attractive forces can be found between the molecules with the following…
A:
Q: What intermolecular forces are present in CH3OH?
A: Hydrogen bonding.
Q: Identifying the intermolecular forces between atoms, ions and molecules. What kind of…
A: We know that, Molecular formula for carbon tetrachloride is CCl4 Carbon tetrachloride is non polar…
Q: What types of intermolecular forces exist and how do they determine a compound’s boiling point and…
A: Types of Intermolecular Forces : There are Three types of intermolecular forces are, 1. London…
Q: What happens to the intermolecular forces of attraction inside an ice when it melts?
A: As we know , the change in state of any substance is due to the change in average kinetic energy of…
Q: What kind of intermolecular forces act between an ammonia molecule and a zinc cation? Note: If there…
A: Intermolecular forces are forces of attraction or repulsion acts between neighbouring atoms. Types…
What types of intermolecular forces are exhibited by each compound?
*this question is from my book
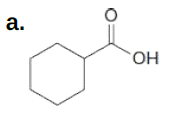
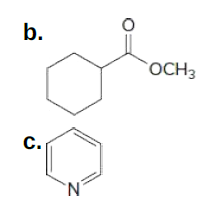
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- But the correct answer is pH 8.81 for part bConsider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)what species could be represented by HB+?

