Q: 6) How many isomers of C₂H, can you construct? Draw a dash structural representation of each…
A: Isomers are the different compounds having same molecular formula. There different physical and…
Q: No Plagiarism Please! Use the "My solution mode" simulation to complete Describe how you could…
A: Acids, bases and pH
Q: At 500 C, cyclopropane (CaHe) reamanges to propene (CH₂-CH=CH₂). The reaction is first order, and…
A: Given, The rate constant of the reaction (k) = 6.7×10-4 S-1 The initial concentration of C3H6 =…
Q: 10. The following structure is a combination of the aniline resonance structures. Place a 8 on the…
A: Given that, a combination of the aniline resonance structures is given below We have to place δ- on…
Q: In order to prepare a buffer of pH 4.60, you start with 500.0 mL of 0.100 M benzoic acid (C6H5COOH)…
A: 1 mole benzoic acid react with 1 mole NaOH. Henderson equation for buffer solution, pH = pKa + log…
Q: What is the effective mass yield for the reaction shown? The density of ethyl L-lactate is 1.03…
A: We are given an organic synthesis reaction and we have to calculate the effective mass yield of it.
Q: Draw structural formulas for the products of the reaction shown below. CH₂CH₂-O-CHCH3 + HBr CH3
A: Given : structure of reactant
Q: Match each of the elements on the left to an elemental form on the right. Xe Cu N C(fullerenes) Ge…
A:
Q: Under certain conditions, the substance ammonium chloride can be broken down to form ammonia and…
A:
Q: Ca(OH)2 (s) + H₂S(g) →→→ CaS(s) + 2H₂O(1) In the reaction above, calcium hydroxide and hydrogen…
A: As we know limiting reactant is the one that is completely reacts in the reaction and the excess…
Q: Br :Br: G Select to Add Arrows Br Select to Add Arrows Br :Br:G Select to Add Arrows
A: The concept used in this question is taken from basic organic chemistry. Here in this question we…
Q: The following initial rate data are for the reaction of UO₂+ with hydrogen ion in aqueous solution:…
A:
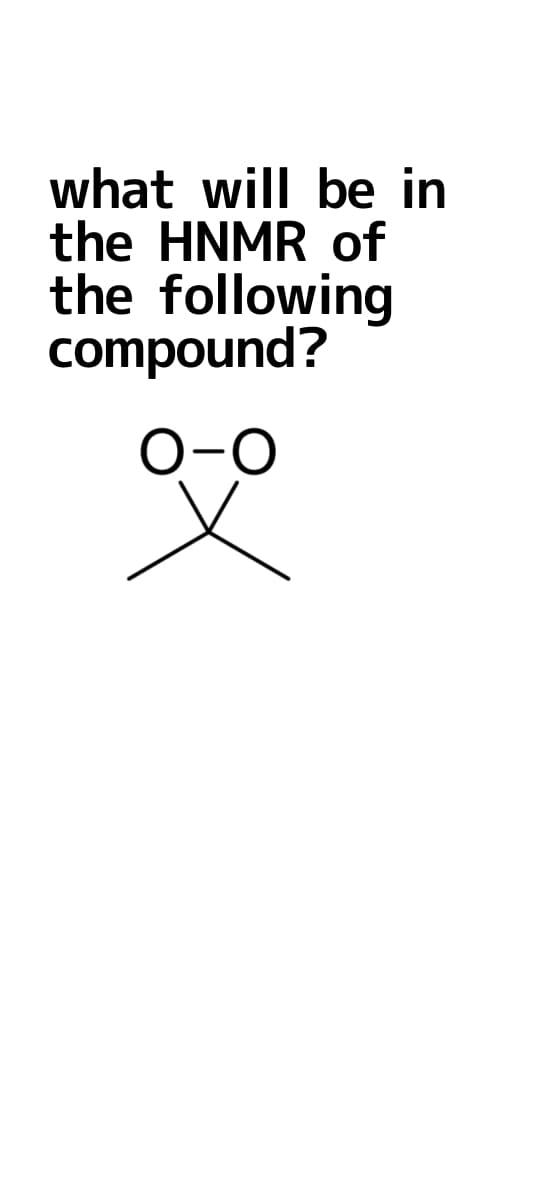
Q: Which molecule has two ¹H NMR signals and will produce the following NMR spectrum? 3 II
A: Given : 1H NMR signal
Q: At 25 °C and 775 Torr, carbon dioxide has a solubility of 0.0347 M in water. What is its solubility…
A: Given -> T = 25°C P1= 775 torr Solubility (S1) = 0.0347 M P2 = 1330 torr
Q: Please use two Friedel-Crafts acylation reactions to produce:
A: Friedel-Crafts acylation reaction: Benzene gives an aromatic electrophilic substitution reaction.…
Q: A mixture of 7.0 grams of nitrogen gas and 48.0 grams of helium has a pressure of 12.0 atm under…
A: we have to calculate the partial pressure of nitrogen
Q: H₂ H₂ (a) H₂C=C-C-C-CH3 1-pentene (b) (c) (d) H3C C C CH3 ΤΗ CH3 2-butene cyclobutene…
A: The structures of the given molecules are
Q: Which molecule has two ¹H NMR signals and will produce the following NMR spectrum? 1 10 9 8 7 6 5 A.…
A: -> Number of signal in 1HNMR is equal to total number of non equivalent hydrogen present in…
Q: A student performed three aqueous layer titrations in Part 1 and they reported the following…
A: To answer: Which determinations were the student's 'best two'? Calculate the percent difference for…
Q: O + NaH DMF > OH D
A: When an alcohol reacts with haloalkane in presence of a base then ether is formed
Q: A KClKCl solution containing 41 gg of KCl per 100.0 g of water is cooled from 70 ∘C to 0 ∘C. What…
A: A saturated solution is one in which a solvent has completely dissolved all of the solutes presents.…
Q: Hydrocarbon Reactions mayd •*-||||||| HHHHHHH HH CH₂ C10H22 (1)→ C2H6 (9) + C7H16 (1) + C(s)
A: 1.Cracking in chemistry refers to the process of breaking down large hydrocarbon molecules into…
Q: What protecting group might you use on the problematic compouind above to allow for a Grignard…
A: Protecting a functional group refers to the process of chemically modifying a reactive chemical…
Q: The following two structures would be considered isomers. Select one: True False Use the following…
A:
Q: There are three sets of sketches below, showing the same pure molecular compound (ammonia, molecular…
A: According to the question, The melting point of ammonia is given by = -77.7°C The boiling point of…
Q: Identify the functional groups by matching the given structures with the provided options. Write the…
A: In IUPAC nomenclature when a compound has more than 1 functional group then the order of precedence…
Q: A solution that contains 55.0g of ascorbic acid in 250. g of water freezes at 2.34*C. Calculate the…
A:
Q: Given the following IR data, what is the most likely identity of the compound? Wavenumber (cm¹)…
A: IR spectroscopy is an important tool for the determination of the functional group in the compound.…
Q: what is the purpose of adding concentrated sulfuric acid to a product after distillation? Write the…
A: Distillation is a process of separating mixtures based on their boiling points. The mixture is…
Q: Order the 3 standard phases of matter from least entropy to most entropy.
A: Any matter have 3 standard phases - solid, liquid and gas. Entropy is a measure of disorder in a…
Q: What is the atom in the image
A: The structure of atom is provided with nucleus and the electrons on the orbits. Counting of…
Q: alculate the molality of a solution formed by adding 2.60 g NH4 olality: to 19.1 g of water.
A:
Q: 17. As shown below, the amino acid histidine can exist in 4 different protonation states depending…
A:
Q: 1.34. A derivative with respect to the same variable can be taken more than once: (a) and is called…
A: The given terms are We have to find the expression of the following terms for the ideal gas…
Q: 8 TSHETKEIX 4800 001 TINSHETTINKE 8 1000 3000 1000 A B 2000 2008 MIYEHUNGER AVEMBER- 1100 1500 m…
A:
Q: Data for the following reaction are given in the table below. CO(g) + NO₂(g) → CO₂(g) + NO(g) Trial…
A: Suppose a reaction M +N –> O Then rate law equation of this reaction given as Rate = k[M]a[N]b…
Q: Iodine and bromine react to give iodine monobromide, IBr. I2(g) + Br2(g) → 21Br(g) What is the…
A:
Q: A canister containing 8g of helium gas (atomic mass 4.0U) was used to fill up party balloons. When…
A: Answer:- Firstly using the ideal gas equation, volume of the canister was calculated then using the…
Q: 4. Write the condensed formula of the following structures. H 3. H Н I N- hydroxylamine H-Q…
A:
Q: The percent yield of this reaction is 78.0%. 2Al(s) + 3 F2 (g) →→→ 2AlF3 (s) Determine the mass, in…
A:
Q: Consider the following reaction where Kc = 10.5 at 350 K: 2 CH₂Cl₂ (9) ⇒ CH4 (9) + CCl4 (9) A…
A: The expression for the reaction quotient for the given reaction is as follows: Q = CH4 CCl4CH2Cl22
Q: 3. 2. нс. нс- н.с. Н.С- ostwork CH Complete the statement: "The reason organic acids and bases can…
A: Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to…
Q: Write the electron configuration of the following atoms and identify what Family and period do they…
A: The electronic configuration and the class of the elements are given below
Q: D. Give the quantum numbers for the last electron of each element given in Part II following the…
A:
Q: Please modify this drug (tamoxifen) to make it water soluble and easy to be absorbed in stomach? mal…
A: In the given molecule we have to tell that ,how this compound can be easily soluble in water and…
Q: When 273.55 grams of water are produced, 1.5429 × 1025 particles of potassium hydroxide were…
A: Answer: This question is based on stochiometric calculation where by using stoichiometric…
Q: 1. 2. 3. A
A: Secondary orbital overlap is main reason to get endo product. In these three reaction, dienophiles…
Q: Dose ordered: docusate 40 mg Stock available: docusate sodium 20 mg/5 mL syrup What is the volume to…
A: The given data is Dose ordered: Tagamet (cimetidine docusate sodium 150 mg Stock available:…
Q: Please answer the following study question clearly and thoroughly: Explain Le Chateliers Principle…
A: •Question is regarding le Chatlier principle. •Here is the explanation of principle.
Q: 2. What quantity should be dispensed when 50 mg of amitryptyline has been prescribed two times per…
A: Dosage of medication
Step by step
Solved in 3 steps with 1 images

- Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The pKa of unsubstituted phenol, for example, is 9.89, while that of p-nitrophenol is 7.15. Draw resonance structures of the corresponding phenoxide anions and explain the data.please explain how to get thrree answers to thr following questions thanks. Need all threeCan u help me im a student w this problem.
- Really hoping for solutions since I’m having a hard time with this. Pls. skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. A kinetics experiment was performed for the formation of a novel compound, UNO, from its reactants, Chemicals G, R, A, D, and E. The first six runs (Runs 1-6) were all performed at 301 K, while the next two runs (Runs 7-8) at varying temperatures. These are tabulated in the table shown below. Table 1. Reaction rate for the reaction of the different chemicals at a given concentration at 301 K. Run [G], M [R], M [A], M [D], M [E], M Rate, M/s 1 0.80 0.01 0.20 0.50 2.00 0.180 2 0.80 0.02 0.20 0.50 0.02 3.54 x 10-3 3 0.80 0.01 0.20 0.50 0.02 1.80 x 10-3 4 0.80 0.01 0.60 0.50 0.02 1.80 x 10-3 5 1.20 0.01 0.20 0.50 0.02 4.02 x 10-3 6 0.80 0.01 0.20 0.15 0.02 1.95 x 10-2 a. Calculate the rate order of each chemical and overall rate order of the…Explain why ethene does not react with HX ( X=Cl , Br or I) under normal conditions.The following reactions are unlikely to provide the indicated product in high yield. What is wrong with each?
- Isoleucine, another of the twenty amino acids found in proteins, is metabolized by a pathway that includes the following step. Propose a mechanism.Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline and bromobenzene.predict the products: XCL2+ E3PO4 --->



