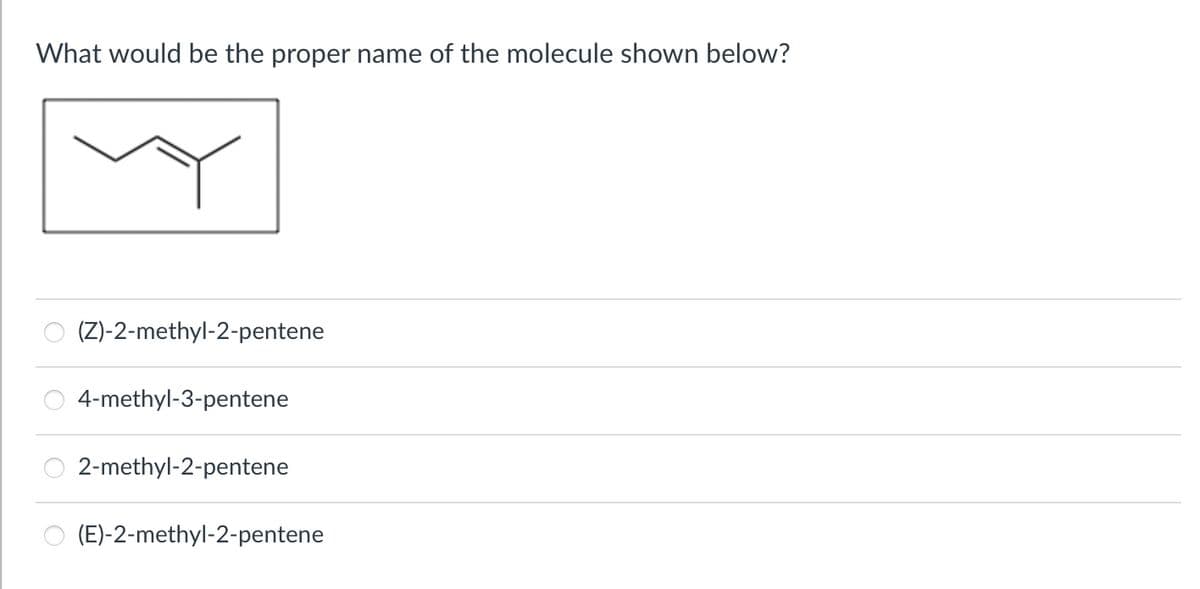
What would be the proper name of the molecule shown below? (Z)-2-methyl-2-pentene 4-methyl-3-pentene 2-methyl-2-pentene (E)-2-methyl-2-pentene
Q: Give the reducible representation of the trigonal planar with D3h point group
A: 4. Reduce the set of characters to a linear combination of the character sets of the point group.
Q: Use the References to access impo Classify each of the following as a strong acid or a weak acid.…
A: Strong Acid: An acid which completely dissociates in aqueous solution is called strong acid. e.g.…
Q: What are the Ka values of acetic acid and hydrochloric acid?
A: The objective of the question is to find the Ka values of acetic acid and hydrochloric acid. The Ka…
Q: Draw the two major products obtained in the reaction shown. Br2 (one equivalent) CH2Cl2
A: Given compound is brominated in the presence of CH2Cl2
Q: Complete the Mechanism with all intermediate steps (hint: what can be done can be undone; what do…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: can this be written on paper please
A: ∆Hrxn=−1276 kJ/molThe reaction is exothermic.Explanation:Since is negative so the reaction is…
Q: 4. For each of the following, determine if either a (a) hydride shift or (b) methyl shift has…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: 1. Complete the following reactions by providing any missing components. Provide all products formed…
A: Given are organic reactions. Reaction a is oxidation reaction. Reaction b is nitration reaction…
Q: Br E2 Br E2
A: E2 elimination is a concerted mechanism. In this process the base removes the proton from the alkyl…
Q: Problem 29 To determine the symmetry of the normal modes of methane, an analysis of the…
A: Γreducible = A1 + E + 2T2 Explanation:Step 1: Reducible representationA representation of a group…
Q: 1) Provide an arow-pushing mechanism for the reaction shown below. محمد py OH
A: In the given question we need to find the product with appropriate mechanism when this given…
Q: Example 6 An imine is a molecule that contains a carbon – nitrogen double bond. Describe the bonding…
A: See the answer in the explanation fieldExplanation:
Q: How many products do you expect when (2R, 3R)-dichlorocyclohexanone is reacted in the presence of…
A: The objective of the question is to determine the number of products that can be formed when (2R,…
Q: Draw the first step in the mechanism for the acid-catalyzed hydrochlorination of the alkene in the…
A: Alkenes are unsaturated organic compounds that have a double bond.They are reactive due to the…
Q: OH
A:
Q: You have an aliquot of 10 mL of 0.1097 mol/L phosphoric acid. The ka values for phosphoric acid are…
A: Volume of phosphoric acid = 10 mLMolarity of phosphoric acid sample = 0.1097 mol/LVolume of NaOH…
Q: Which of the following reagents can be used to convert the compound below into a carboxylic acid?…
A: The conversion of an alkyne to a carboxylic acid is a significant transformation in organic…
Q: Name each of the acids listed in the table below. chemical formula HCIO name HI ☐ H₂SO3 ☐ HCIO 4 ☐
A: For the binary acids, the name starts with the hydro. Then write the name of the anion with the…
Q: Name the alkenes below. Use ONLY E/Z designators to indicate stereochemistry. CH=CH2 H3C H3C CH3…
A: To determine the IUPAC name of the given alkene.
Q: Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist…
A:
Q: Snow White was brought to the ER after being found unconscious in the forest. She remained…
A: The objective of the question is to determine the acid-base condition of Snow White based on her…
Q: Provide an acceptable name for the following compound.
A: In the naming of the primary amines, the alkyl group is named as a substituent(prefix+yl), and the…
Q: .Draw Lewis Dot diagrams for the PF3 b) SiCl4 SeH2 d) Sl₂ e) SiSez
A: We have to draw the Lewis dot structure
Q: Arrange these substances in order of increasing boiling point: H2 H₂O LICI H₂S OH₂<H₂O<LiCl < H₂S…
A: The objective of the question is to arrange the given substances H2, H2O, LiCl, H2S in order of…
Q: Can the alkyl halide in the drawing area be made in one step from an alcohol, without losing…
A: The objective of this question is to draw the starting material and reagents used for the product…
Q: Provide the structure of the major organic product in the reaction below. NH2 FCI
A: N-Acylation is the reaction in which the acyl group (-COCH3) is introduced into nitrogen of the…
Q: Create 3-methyl-1-butanamine using the four different methods below. a. Gabriel synthesis b. Using…
A: Given are amine synthesis reaction. Given amine is, 3-methyl-1-butanamine.We can synthesize this…
Q: In the complex ion [Fe(C,O4)3], iron is in the +3 oxidation state. What is the d electron count of…
A: The electronic configuration of Fe (Iron) in its neutral state is: 1s2 2s2 2p6 3s2 3p6 3d6 4s2 Total…
Q: The chemistry tests of choice for differentiating hemoglobinopathies and thalassemias from iron…
A: The objective of the question is to identify the most appropriate chemistry tests for…
Q: HIN 1. CH3I (excess) 2. Ag₂O 3. A
A: Hofmann elimination is a chemical reaction that produces alkenes and tertiary amines from quaternary…
Q: Assuming no phase transition, what is the change in temperature (°C) if 310.0 mL ethanol (molar mass…
A: The change in temperature for the ethanol is = 4.51 °CExplanation:Step 1: Calculation of moles of…
Q: Give the systematic (IUPAC) names for these molecules. IUPAC name: 요 -OCCH2CH3 CH3OCCH2CH2CHCH3…
A: Given :Compound (I) :Compound (II) : Objective : Determine the IUPAC anme of the compound given.
Q: Draw the reaction of the Grignard reagent interacting water and the product produced. bromobenzene…
A: The objective of this question is to draw the complete reaction with mechanism for the reaction…
Q: Assume 99% of the starting aldehyde in Model 4 is in the keto form. Explain how most of the propanal…
A: Given:99% of the starting material aldehyde is in the keto form.Explain how Propanal converted to…
Q: Draw a scheme showing the 4 possible stereoisomers of 2,3-dibromo-3-phenylpropanoic acid,
A: Given is organic compound. The given compound has two streocenters.A stereocenter is carbon that has…
Q: i need the answer quickly
A: The given reaction is the reaction between alkene and alkyne with an excess of HBr. This is an…
Q: Design an experiment to measure the standard potential of a Silver Describe your electrochemical…
A: Summary:Experiment to Measure the Standard Potential of Silver:Setup:Obtain a standard hydrogen…
Q: In the process of heme degradation, _________________ bind(s) iron and _________________ bind(s)…
A: The objective of the question is to identify the correct substances that bind iron and alpha and…
Q: 1. MgCl & ether solvent 2. H₂O & H3O+
A: We have to predict the product.
Q: Draw a structural formula for the alkene you would use to prepare the alcohol shown by…
A: Alkene gives an electrophilic addition reaction with borane. Alkene reacts with BH3 and then NaOH…
Q: What is the major organic product obtained from the following reaction? (CH3)₂CuLi O a.…
A: The objective of the question is to predict the product formed in the following reaction given.
Q: 4. Please complete the table below. What is the functional group name? What is the IR stretching…
A: The objective of the question is to identify the properties of the functional group NH, including…
Q: Box or circle final answers for each row.
A: Energy of electron in the ground state =100zJ Energy of electron in the first excited state =250zJ…
Q: Rank the following dienes in order of increasing stability (least to most). =
A: Given are organic compounds.Given compounds are dienes.Compound I is conjugated diene.Compound II is…
Q: Draw a step-wise mechanism for the following substitution reaction
A: Given reaction is substitution reaction. A given reaction will follow Sn1 mechanism because…
Q: Draw a Lewis structure for SO2 in which all atoms obey the octet rule. Show formal charges. Do not…
A: Answer below this question Explanation:
Q: Which one of the following statements describes partially compensated metabolic acidosis?…
A: The objective of the question is to identify the correct statement that describes the condition of…
Q: Draw the mechanism for the following reactions (organic chemistry) e. LOH H₂O, HCI (cal) d. HO HO H…
A: Given,The reactions are:
Q: Calculate [H3O+] for a 0.45 M solution. SED THIS? Read Section 17.9. You can click on the Review…
A: Given data:Calculate pH and H3O+ concentration.The pH value is useful to determine whether the…
Q: A solution of household bleach contains 5.25% sodium hypochlorite, NaOCl, by mass. Assuming that the…
A: The objective of the question is to calculate the volume of bleach needed to dilute the solution in…
Trending now
This is a popular solution!
Step by step
Solved in 1 steps with 1 images

- Draw a Newman projection, similar to Figure 3-25, down the C1¬C6 bond in the equatorial conformation of methylcyclohexane. Show that the equatorial methyl group is also anti to C5.Step 1c: Draw the structure(s) resulting from the curved arrow mechanism of step 1. Include formal charges and all hydrogen atoms, but do not include lone pairs.Alkyl halides: find the molecular formula and the name of the molecule. Condensed Formula Molecular Formula Name CH3-CH2-CH2-CH2-I C4H9I Iodobutane CH3-CH-CH2-CH2-Br | Br ? ? CH3-CH-CH2-CH-CH2-Br | | F Br ? ?
- Complete the Newman projection below to draw the most stable conformation of 3-methylpentane, looking down the C2-C3 bond. Identify each substituent using "H" for hydrogen and "Me" for methyl. a= b= c= d=Draw the most and least stable Newman projections for the following molecules ( focus on C2 and C3). a. 2-chloro-2- fluoropentane b. 2,2-dimethylbutane C. 2-chloro-2-methylpentane d. 1,2-dibromoethaneWhat is the formula for (S)-2- bromopentane using a) Fisher projection b) Wedges and broken lines c) upload the model in each one of the projections
- how can i rank these isomers from lowest bp to highest please? Trans-pent-2-ene Cis-pent-2-ene 3-methylbut-1-ene 2-methylbut-1-ene 2-methylbut-2-ene Cyclopentane Methylcyclobutane Ethylcyclopropane 1,2-dimethylcyclopropane 1,1-dimethylcyclopropane (don't answer only, need explaination)how can i rank these isomers from lowest bp to highest please? Trans-pent-2-ene Cis-pent-2-ene 3-methylbut-1-ene 2-methylbut-1-ene 2-methylbut-2-ene Cyclopentane Methylcyclobutane Ethylcyclopropane 1,2-dimethylcyclopropane 1,1-dimethylcyclopropaneDraw 2,2 - dichloro - 3 - methylpentane . Fill in all H’s .



