When 0.0700 moles of metal "X" is allowed to completely react with 200 mL of aqueous HCI, the temperature rises from 25.0 °C to 42.9 °C. Calculate AH in KILOJOULES per MOLE (kJ/mol) for the reaction written below, assuming that the heat capacity of the calorimeter is 776 J/°C, that the specific heat of the final solution is the same as that of pure water [4.18 J/(g°C)), and that the density of the solution is 1.00 g/ml. (note: The answer box only accepts numerical answers.do not include units of kJ/mol. Apply the correct sign to your answer, negative or positive.) X (s) + 2 HCI (aq) XCI2 (aq) + H2 (g) AHan- ??? kJ/mol
Thermochemistry
Thermochemistry can be considered as a branch of thermodynamics that deals with the connections between warmth, work, and various types of energy, formed because of different synthetic and actual cycles. Thermochemistry describes the energy changes that occur as a result of reactions or chemical changes in a substance.
Exergonic Reaction
The term exergonic is derived from the Greek word in which ‘ergon’ means work and exergonic means ‘work outside’. Exergonic reactions releases work energy. Exergonic reactions are different from exothermic reactions, the one that releases only heat energy during the course of the reaction. So, exothermic reaction is one type of exergonic reaction. Exergonic reaction releases work energy in different forms like heat, light or sound. For example, a glow stick releases light making that an exergonic reaction and not an exothermic reaction since no heat is released. Even endothermic reactions at very high temperature are exergonic.
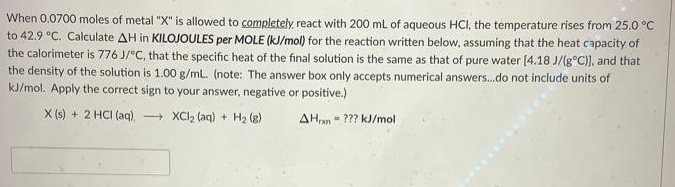
Trending now
This is a popular solution!
Step by step
Solved in 2 steps









