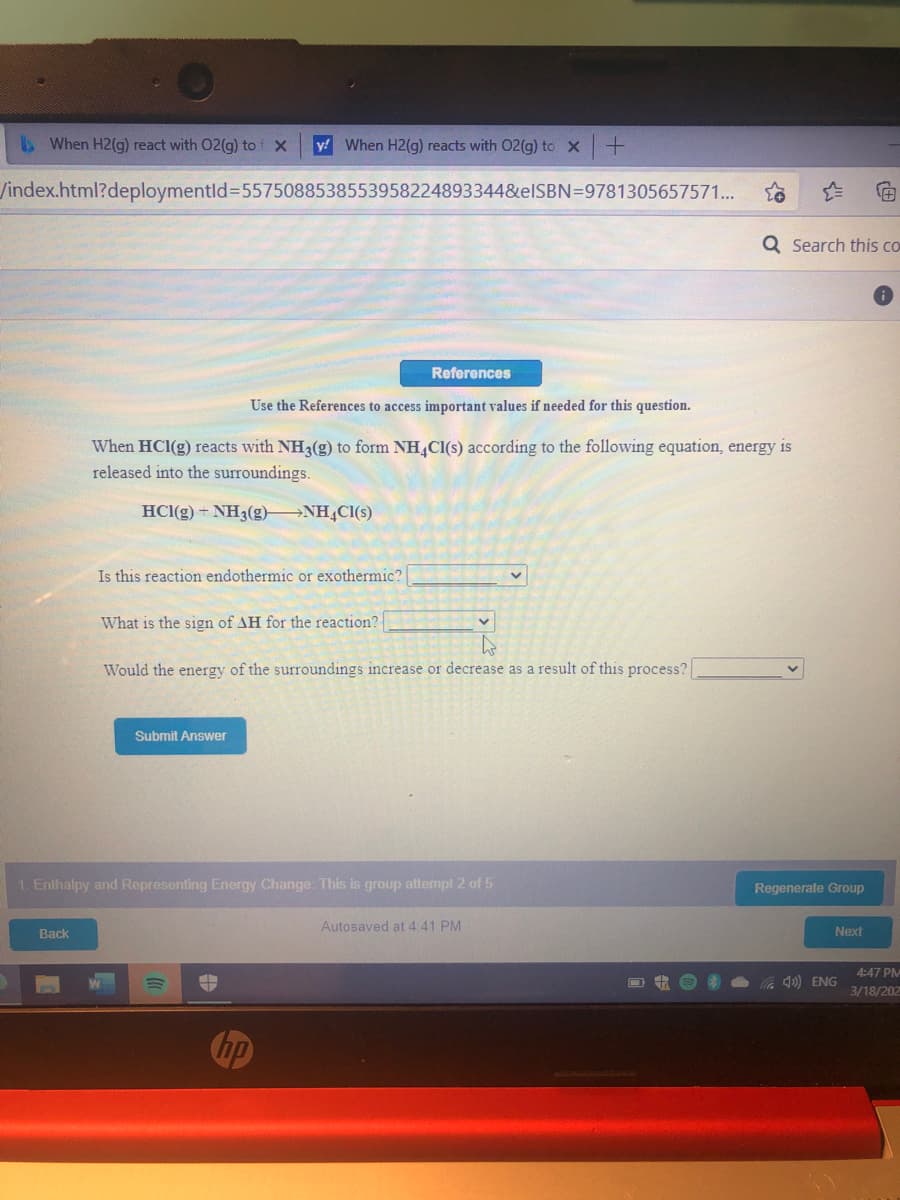
When HCI(g) reacts with NH3(g) to form NH,Cl(s) according to the following equation, energy is released into the surroundings. HCI(g) + NH3(g)- →NH,CI(s) 4. Is this reaction endothermic or exothermic? What is the sign of AH for the reaction? Would the energy of the surroundings increase or decrease as a result of this process? Submit Answer alny and Representing Enermy Change Thic is qrnup altenmnt 2 of 5
When HCI(g) reacts with NH3(g) to form NH,Cl(s) according to the following equation, energy is released into the surroundings. HCI(g) + NH3(g)- →NH,CI(s) 4. Is this reaction endothermic or exothermic? What is the sign of AH for the reaction? Would the energy of the surroundings increase or decrease as a result of this process? Submit Answer alny and Representing Enermy Change Thic is qrnup altenmnt 2 of 5
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
ChapterA1: Evaluation Of Analytical Data
Section: Chapter Questions
Problem A1.22QAP
Related questions
Question
Transcribed Image Text:bWhen H2(g) react with 02(g) to f X
y! When H2(g) reacts with 02(g) to X
/index.html?deploymentld%3D5575088538553958224893344&elSBN=9781305657571...
Q Search this co
References
Use the References to access important values if needed for this question.
When HCI(g) reacts with NH3(g) to form NH,Cl(s) according to the following equation, energy is
released into the surroundings.
HCl(g) + NH3(g) NH,Cl(s)
Is this reaction endothermic or exothermic?
What is the sign of AH for the reaction?
Would the energy of the surroundings increase or decrease as a result of this process?
Submit Answer
1. Enthalpy and Representing Energy Change: This is group attempt2 of 5
Regenerate Group
Autosaved at 4:41 PM
Back
Next
4:47 PM
) ENG
3/18/202
Chp
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT