When inputting these rate constants below, use "NO" for NO, "02" for O2, "N202" for N₂O2, "N204" for N2O4, "k_1" for k₁, "k_1rev" for k-1, "k_2" for k2, "k_2rev" for k-2, and "k_3" for ką. Without making any approximations, what is the rate of production of NO2?
When inputting these rate constants below, use "NO" for NO, "02" for O2, "N202" for N₂O2, "N204" for N2O4, "k_1" for k₁, "k_1rev" for k-1, "k_2" for k2, "k_2rev" for k-2, and "k_3" for ką. Without making any approximations, what is the rate of production of NO2?
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter14: Chemical Kinetics: The Rates Of Chemical Reactions
Section14.7: Reaction Mechanisms
Problem 2.3ACP
Related questions
Question
PLEASE COMPLETE QUESTION ANSWER THAN RATING HEPLFUL

Transcribed Image Text:This problem will consider the following reaction mechanism:
NO + NO
k-1
N₂02
02 + N₂02 N₂O4
k_2
k3
N2O4NO2 + NO2
When inputting these rate constants below, use "NO" for NO, "02" for O2, "N202" for N₂O2, "N204" for
N2O4, "k_1" for k₁, "k_1rev" for k-1, "k_2" for k2, "k_2rev" for k_2, and "k_3" for k3.
Without making any approximations, what is the rate of production of NO2?
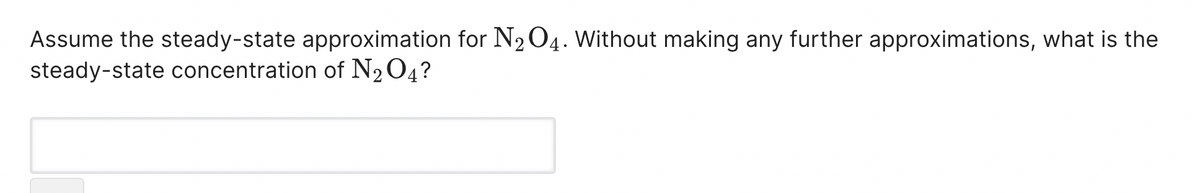
Transcribed Image Text:Assume the steady-state approximation for N₂O4. Without making any further approximations, what is the
steady-state concentration of N₂O4?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning