When starting with completely un-dissociated acetic acid, is it accurate to assume that [HOAc]0 = [HOAc]eq? Why or why not? 2) A highly concentrated acetic acid solution contains 15.0 M acetic acid at equilibrium. What are the equilibrium concentrations of the hydronium and acetate ions in this solution?
When starting with completely un-dissociated acetic acid, is it accurate to assume that [HOAc]0 = [HOAc]eq? Why or why not? 2) A highly concentrated acetic acid solution contains 15.0 M acetic acid at equilibrium. What are the equilibrium concentrations of the hydronium and acetate ions in this solution?
Chapter10: Effect Of Electrolytes On Chemical Equilibria
Section: Chapter Questions
Problem 10.4QAP
Related questions
Question
1) When starting with completely un-dissociated acetic acid, is it accurate to assume that [HOAc]0 = [HOAc]eq? Why or why not?
2) A highly concentrated acetic acid solution contains 15.0 M acetic acid at equilibrium. What are the equilibrium concentrations of the hydronium and acetate ions in this solution?
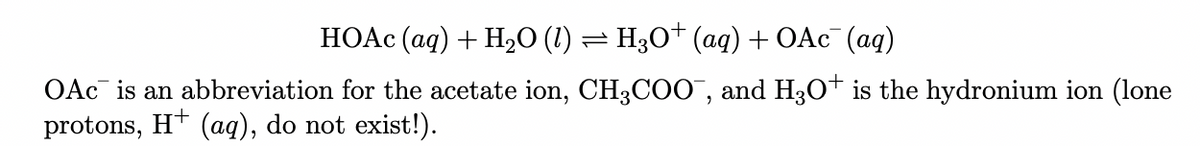
Transcribed Image Text:HOAC (ag) + H,O (1) = H3O† (aq) + OAC¯ (aq)
OAc is an abbreviation for the acetate ion, CH3COO", and H3o* is the hydronium ion (lone
protons, H* (aq), do not exist!).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning