Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: Hii there, since there are multiple questions posted. we are answering first question. if you need…
Q: What is the correct Ko expression for the following reaction? N2 (g) + 3 H2 (g) = 2 NH3 (g)
A: Given :- N2(g) + 3H2(g) <----> 2NH3(g) To write :- Its correct Kc expression
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A:
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A:
Q: Which of the below figures best represents what happens when MeINOs is added to water NO, Mg NO, Mg…
A: Given: Mg(NO3)2 is added in water.
Q: PART B DATA ANALYSIS (LEMON JUICE) Balance the following reaction: 2 NaOH (aq) + C6H3O7 (aq)–→ 2…
A: Balanced chemical reaction is the one in which number of atoms remain same in both the sides i.e.…
Q: 2. Write the balanced "net ionic" equations for the following reactions: HNO₂ (aq) KOH (aq) 7 HOX+ +…
A:
Q: What is the concentration of hydrochloric acid The data The volume of HCl 26 mL Mass of beaker…
A: Given mass of beaker is 51.9 g. Given mass of beaker and marble chip is 66.0 g Mass of weighing boat…
Q: (2c) _____ HC2H3O2 (aq) + ______ NaHCO3 (s) → ______ NaC2H3O2 (aq) + _____ H2O (l) + _____ CO2 (g)…
A: Given skeletal equations are shown below: (2c) _____ HC2H3O2 (aq) + ______ NaHCO3 (s) → ______…
Q: What would be the concentration of X 2- if a 10 mL of a 1 M solution of KOH was added to 50 mL of a…
A: The chemical reaction for the neutralization of a diprotic acid H2X with KOH is needed to be…
Q: ___ Na + ___ F2 → ___ NaF balance the equation
A: In a balanced equation no.of atoms in both side (reactants and products) should be equal. In a…
Q: What is the concentration of lithium ions in 0.170 M Li₂HPO₄?
A: Given that: Concentration of Li2HPO4 = 0.170M Li2HPO4 is strong electrolyte.
Q: How many moles of HNO3 are present if 0.172 mol of Ba(OH)2 neutralize the acid solution? was needed…
A:
Q: If 0.248 g of oxalic acid dihydrate requites 28.4 mL of NaOH for neutralization, what is the…
A: The given mass of oxalic acid dihydrate (C2H2O4. 2 H2O) = 0.248 g The molar mass of oxalic acid…
Q: The following molecular equation represents neutralization reaction of acid and base
A: Molecular equation: HBr(aq)+LiOH(aq) --> LiBr(aq) + H2O(l) Total Ionic equation: H +(aq)+ Br…
Q: Can I please get assistant with the following problem? Write a balanced complete ionic equation for…
A: We would write the ionic equation of compound A2B2 (s)
Q: Select the balanced net ionic equation for the reaction of HNO3(aq) with Ca(OH)2(aq). Select an…
A: Balance Chemical reaction for the reaction HNO3 (aq) with Ca(OH)2 (aq)
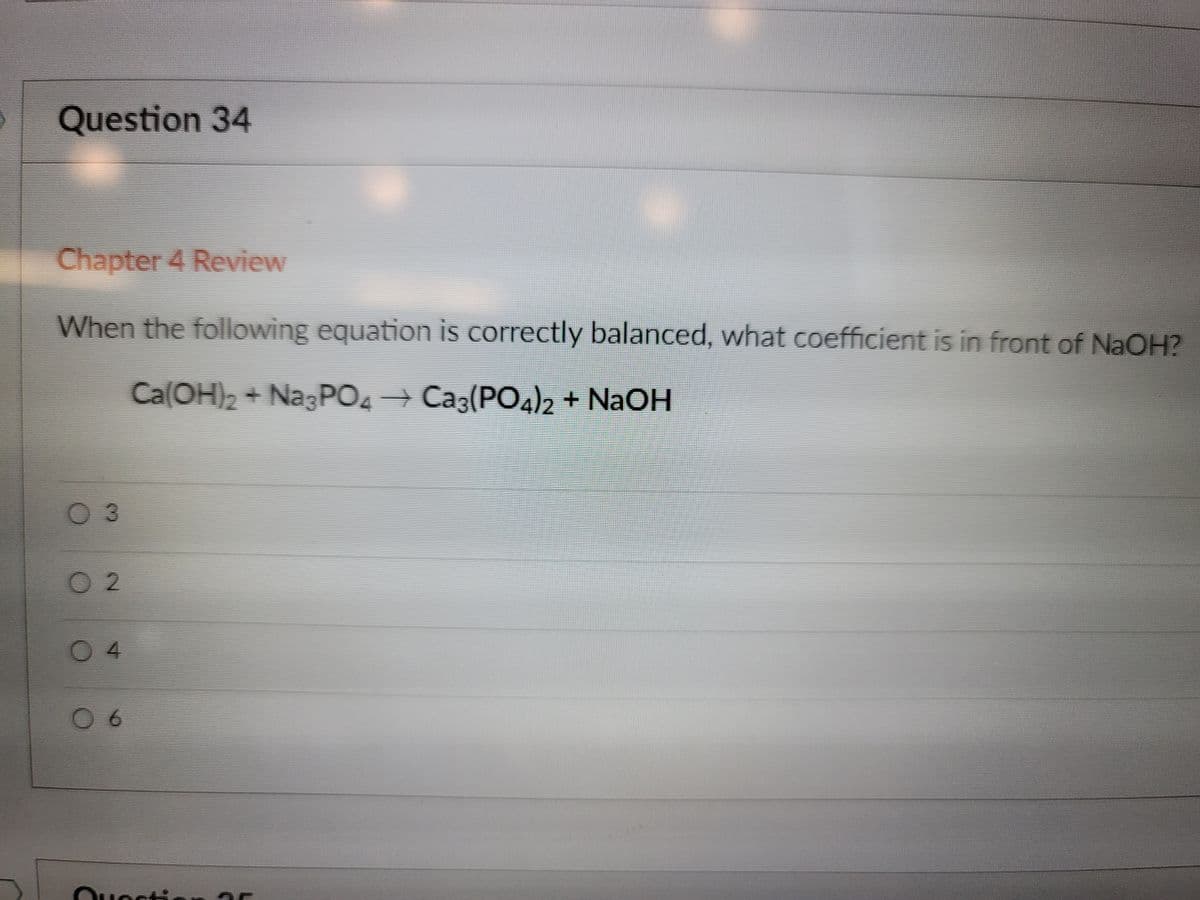
Q: n the following equation is balanced properly under acidic conditions, vhat are the coefficients of…
A:
Q: 1 CaCO3(s) + 2 HCl(aq) → 1 H2O(l) + 1 CO2(g) + 1 CaCl2(aq) (Use balanced equation above to…
A: Given :- 1 CaCO3(s) + 2 HCl(aq) → 1 H2O(l) + 1 CO2(g) + 1 CaCl2(aq) molarity of HCl = 0.100 M…
Q: Binary mixtures of equal moles of which of the following acid-base combinations will lead to a…
A:
Q: Determine the molarity of NaOH and KHP (C8H5KO4) react in a 1:1 ratio.
A:
Q: Aluminum sulfate, known as cake alum, has a remarkably wide range of uses, from dyeing leather and…
A: Given : We have to write the ionic equation for aluminum sulfate and NaOH.
Q: Erength. They collect the following data after the chemical reaction has stopped: Blue- Remained…
A: The question is based on the concept of chemical reactions. we have reacted acid with a base. both…
Q: F (aq) 4. HF(ag) + H200 > H30*aq) HF (aq) in the equation above is a(n) H3O" (aq) in the equation…
A:
Q: When the equation NaOH(aq) + CaC2(aq) -> Ca(OH)2(s) + NaCl(aq) is balanced using smallest whole…
A:
Q: Na+ and Cl- Complete ionic equation H+(aq) + OH-(aq) →H2O(1) spectator ions H+(aq) + Cl-(aq) +…
A: Write the balanced molecular equation Split electrolytes to their corresponding ions to achieve…
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A:
Q: 5.Consider the following reaction. 4 PH3(g) + 8 02(9g) → P4010(s) + 6 H20(g) If a container were to…
A: Limiting Reactant
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: Introduction : As per company norms we cannot answer more than one question per session as they…
Q: 1. ACID/BASE. Supply the missing reactants and products and balance eac equation. All are acid-base…
A: Balanced reaction are:
Q: A solution contains 5.0 meq of SO42− ion. Based on this information,
A: (a)
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The given reaction is, Ni2++Hg2+→NiO2+Hg The balanced reaction under acidic conditions is,…
Q: This is a question for a lab about the standardization of an acid and base. In the lab, we used the…
A: KHP+NaOH→NaKP+H2O
Q: 2 Li + 2 H20 -> ? LIOH + H2 When the equation is balanced, what coefficient is needed for LIOH?
A: Given, 2 Li + 2 H2O ------> ? LiOH + H2
Q: calculate the amount of salicylic acid needed if only 74.9 percent of salicylic acid is converted to…
A: Step : 1 Calculate the amount of salicylic acid needed if only 74.9 percent of salicylic…
Q: For the dissociation of iodic acid, HIO3(aq) + H20(1) =H30*(aq) + 103 (aq) If, at equilibrium,…
A: The dissociation of the iodic acid is given as: HIO3aq + H2Ol ⇌ H3O+aq + IO3-aq Given: The conc. of…
Q: Given the balanced equation: HBr(aq) + NaOH(aq) NaBr(aq) + H2O) and remembering that Molarity =…
A:
Q: Which reaction below is the balanced complete ionic eguation for occurs when aqueous solutions of…
A: Aqueous sulfuric acid reacts with base potassium hydroxide and neutralization reaction occurs. And…
Q: What is the mole ratio of NaOH to H2SO4 when the following equation is balanced: NaOH + H2SO4 → H2O…
A: Answer:
Q: Oxalic acid, H2C2O4, reacts with sodium hydroxide in a neutralization reaction. What is the balanced…
A: The reactants given are H2C2O4 and sodium hydroxide i.e NaOH. Hence the acid base neutralisation…
Q: What is the concentration of a H3PO4 solution if 17.5 mL of the solution is neutralized by 31.2 mL…
A:
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The reaction taking place is given as, => ClO2 + Cd2+ -------> ClO3- + Cd In the above…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The unbalanced redox reaction is: Pb2+(aq) + Cr3+(aq) → Pb(s) + Cr2O72-(aq) While balancing a redox…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: Water is present as reactant. With the coefficient of two. Three electrons are transport in this…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A:
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A: Chemical equations are balanced in order to follow the Law of conservation of mass. According to…
Q: H3PO4 + NaOH --> Na3PO4 + H2O A. Balance the equation B. If 36.0 ml of H3PO4 react exactly with…
A: The given chemical reaction is, H3PO4 + NaOH --> Na3PO4 + H2O
Q: given the following equation what are the spectator ions? (answer from the image) Na2SO4(aq) +…
A: complete ionic equation can be used to answer this question as:
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The reaction taking place is given as, => Fe2+ + I2 -------> Fe + IO3-
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Show work One ingredient in some antacids is Al(OH)3 which reacts with stomach acid, HCl . Complete and balance the equation for this neutralization reaction written below. __?__Al(OH)3 + __?_ HCl → __?____ + __?____Which below is the proper equation(s) for ionization of nitrous acid? a) HNO3 + H2O --> H3O+ + NO3- b) HNO3 + H2O --> H2NO3+ + OH- c) HNO2 + H2O --> H3O+ + NO2- d) HNO2 + H2O <--> H3O+ + NO2- e) H2NO3 + 2 H2O <--> 2 H3O+ + NO32- f) HNO3 + H2O <--> H2NO3+ + OH- g) HNO3 + H2O <--> H3O+ + NO3-Br2, bromine tested with chloride and iodide ions Halide salt Color change observed in the cyclohexane layer? Net Ionic Equation(s)/ No reaction NaCl NaI Comment on the ability of bromine to displace chlorine and iodine from their halide salts: What does this say about the relative reactivity of bromine?
- If you have a sample of an acid, which of the following best describes the concentration of [H3O+]? Question 27 options: 0.0 M 1.0 x 10-14 M > 1.0 x 10-7 M < 1.0 x 10-7 M 1.0 x 10-7 MMake correct choice in the following equation: NH₂ än OH an O No reaction COOH an L aqueous NaHCO3Question 29: Balance each of the following neutralization reactions Part A: HNO3(aq)+NaOH(s)→H2O(l)+NaNO3(aq) Part B: H2SO4(aq)+NH4OH(s)→H2O(l)+(NH4)2SO4(aq)
- What is the effect of H2SO4 on Epsom salts? Select all that apply. Select all that apply. a. Visible reaction b. No visible reaction c. Exothermic (heat-producing) d. Endothermic (heat-consuming)T1 Is chromium strong enough to remove nickel from nickel chloride? Is lead strong enough to remove nickel from nickel sulfate? Select one: a. None of these is correct b. Only chromium is strong enough c. Chromium is not strong enough but lead is d. Neither is strong enough e. Both are strong enoughQuestion 34 What is the net ionic equation for this reaction? Pb(NO3)2 (aq) + 2 KCl (aq) --> PbCl2 (s) + 2 KNO3 (aq) Group of answer choices 2 NO3- (aq) + 2 K+ (aq) --> 2 KNO3 (aq) None of these is correct Pb2+ (aq) + 2 Cl- (aq) --> PbCl2 (s) Pb2+ (aq) + 2 NO3- (aq) + 2 K+ (aq) + 2 Cl- (aq) --> PbCl2 (s) + 2 K+ (aq) + 2 NO3- (aq)
- NEED HELP PLEASE GRAVIMETRIC ANALYSIS (Exercises) Instructions: Answer each of the problems below by showing your solutions neatly and logically. You may use periodic table for atomic masses of elements. Copy and answer each problem and please box your final answers. 7. Orthophosphate (PO43-) is determined by weighing as ammonium phosphomolybdate, (NH4)PO4 • 12Mo03. Calculate the percent P in the sample and the percent P205 if 1.1762 g precipitate (ppt) were obtained from a 0.3711-g sample. NOTE: KINDLY WRITE THE SOLUTIONS ON A SEPARATE SHEET OF PAPERWhich response includes all of the following substances that have ∆Hf= 0, and no other substances? I. HCl(g) II. Na(s) III. HCl(aq) IV. F2(g) A. II b. I, II, and IV C. I, II, III, and IV D. II and IV E. I and IIWhich of the following reactions are redox reactions? Select all that apply. Group of answer choices None are redox reactions 11H+(aq) + 2MnO4–(aq) + 5Cl–(aq) --> 2Mn2+(aq) + 5HClO(aq) + 3H2O(l) CH3COOH(aq) + NaOH(aq) --> CH3COONa(aq) + H2O(l) 2Al(s) + 6H+(aq) --> 2Al3+(aq) + 3H2(g) 3AgNO3(aq) + FeCl3(aq) --> 3AgCl(s) + Fe(NO3)3(aq)

