Q: An azeotrope is, a mixture in which the liquid and vapor phases are at equilibrium with each other…
A: The given statement is- An azeotrope is, a mixture in which the liquid and vapour phases are at…
Q: 1) Describe a simple chemical test to differentiate the following pairs of compounds. Write the…
A: #1: (i): 2-propanone and propanoic acid: 2-propanone(CH3COCH3) has terminal -COCH3 group that gives…
Q: (i) Draw the cyclic beta form of structure II. (i) Draw the cyclic alpha form of structure II.
A:
Q: Calculate the change in energy (in Joules) when an electron transition from n=4 to n=2 in a hydrogen…
A: The electronic energy levels in an atom are quantized and when an electron undergoes a transition…
Q: The balanced chemical equation for the combustion of propane gas is: C3H8 (g) + 5 O2 (g) --> 3 CO2…
A:
Q: The engineer is extruding a polymer with the following characteristics: Specific volume, V = 10-6…
A: Polymer extrusion is a method where molten polymer is converted into components of fixed cross…
Q: Consider the following half-reactions: Half-reaction Ag+ (aq) + e Co2+ (aq) + 2e™ Cr3+ (aq) + 3e E°…
A: Here we are required to arrange the species on increasing order of strength of oxidizing agent
Q: Gallium hydroxide is a very sparingly soluble ionic salt. Given the molar solubility of gallium…
A:
Q: 3 What is the biggest difference between the Lewis and the AO-model in terms of the localisation of…
A: Localized electrons are the bonding electrons whereas the delocalized electrons are the non-bonding…
Q: 1) Determine the absolute configuration of the following compounds according to the (E)- (Z)-system…
A: E/Z stereoisomerism is shown by alkenes where each of the C of the -C=C- is attached to two…
Q: The mean free path of a gas at a temperature T1 and a pressure P1 is 7x10-5 cm. At these temperature…
A: Mean free path (λ) = 7×10-5 cm Number of molecules per unit volume (N) = 2×1018 molecules/cm3 We…
Q: Please send me the question in 30 minutes it's very urgent plz
A: According to henry's law: Henry's Law constant(Kh) for Helium in the blood = 4.44 x 10-4 M atm-1.…
Q: A sample of nickel silver contains 18.0% nickel by mass. What mass of nickel silver must be melted…
A: Given: Sample of Nickel Silver contains: 18% Nickel by mass.
Q: Consider the reactant below at 25°C. Describe the possible product to complete each scenario and…
A: Here we are given a reactant 4A which is in liquid stage below 25° C. Entropy : Entropy is the…
Q: An 80:20 mixture by mass of N, and O, is placed in a 5.00 L cylinder at a total pressure of 30.0 kPa…
A:
Q: Gallium hydroxide is a very sparingly soluble ionic salt. Given the molar solubility of gallium…
A: The solubility of a compound can be explained as the maximum amount of the compound that can be…
Q: Calcium hydroxide will precipitate from solution by the following equilibrium: 2+ Ca²+ (aq) +…
A:
Q: O Cu'laq) OH,Oilae) O Br O 10₂ (aq) Standard Electrode Potentials at 25 °C Reduction Half-Reaction…
A:
Q: 1) Provide the major organic product(s) in the reactions below. H₂CO CN + NC A
A: Since, In Diels-Alder reaction, Diene react with dienophile to form adduct as product. So,
Q: a) What terms can arise from the (He)2s22p13d1 excited configuration of carbon? Terms: b) What…
A:
Q: he balanced chemical equation for the combustion of propane gas is: C3H8 (g) + 5 O2 (g) --> 3 CO2…
A: The standard free energy (G) and the Gibbs free energy change (∆G°) can be related by the equation…
Q: Chemistry For the gas phase decomposition of dichloroethane, CH3CHC12 -> CH2 = CHCI + HCI the rate…
A:
Q: An analytical chemist has been given the task of precipitating lead cation (Pb2+) out of solution…
A: Given, Molar concentration of Pb2+ ion = [Pb2+] = 0.424 M Solubility product constant of PbI2 =…
Q: The oxidation of trichloroethane results in the following reaction equation: 4 CaH,C|a() + 11 0, (g)…
A:
Q: Why should solid diazonium salts never be separated from their mother liquor by filtration?
A: Why solid diazonium salts never be separated from their mother liquor by filtration - Let's discuss.
Q: Propoxide (CH3CH2CH2O- ) is a larger molecule than ethoxide (CH3CH2O- ), yet they are equally basic.…
A: Propoxide (CH3CH2CH2O- ) is a larger molecule than ethoxide (CH3CH2O- ), yet they are equally basic…
Q: 5. Write forward and reverse reactions for the following: CH4 (g) + 2H2S (g) (g) ==== CS2 (g) + 4H2…
A:
Q: Would you expect F₂ to be soluble in CBr4 or not? FULLY explain your answer. Edit Format Table
A:
Q: Enter the IUPAC name for the compound shown below:
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 5) Which of the following type of protons are chemically equivalent? A. homotopic only B.…
A: To solve this problem we have to know about chemical equivalence of homotopic , diastereotopic and…
Q: number of significant figures. To avoid rounding errors, do not round your answers until the very…
A:
Q: For each of the following transition metal complex ions (a) [FeBr2(OH)(NO3)]¹- (b) [RhF3(SCN)3]4-…
A:
Q: A 1.00 L buffer solution is 0.050 M in HC7H5O2 and 0.200 M in LiC7H5O2. Calculate the pH of the…
A: Given: A 1.00 L buffer solution is 0.050 M in HC7H5O2 and 0.200 M in LiC7H5O2 The Ka for HC7H5O2 =…
Q: Solve for x, where M is molar and s is seconds. x = = (6.1 × 10³ M-²s¯¹)(0.25 M)³ Enter the answer.…
A: Please find the attachment.
Q: Select all that is true for cycloheptatriene and the ions it can form. The 6 pi electrons in…
A:
Q: For the enzyme urease acting on urea, KM = 2.0 x 10-3 M, and k2 = 2.5 x 104 s-¹ at 298 K and pH 7.5.…
A:
Q: A voltaic cell is constructed from a standard H+H₂ half cell (Eº red = 0.000V) and a standard I₂|I…
A: A voltic cell is made up from a standard H+|H2 half cell and a standard I2|I- half cell .Here, we…
Q: Q2: Choose the correct answer for each of the following: () 1 - In the infrared spectrum of a…
A: Since, you have asked multiple questions, we solve the first question for you. If you want any…
Q: Consider an aqueous solution of acetic acid: CH3COOH (aq) H2O (l) CH3COO (aq) H2O (aq). If…
A: acetic acid is a weak acid and it undergoes partial dissociationto give acetate and H+ ions. The…
Q: 2. Ni(P(0-C6H4Me)3)2Cl2 is strongly paramagnetic, while Ni(PMe3)2Cl₂ is diamagnetic see the…
A: PR3 ligands are known to exhibit a weak π-acid character when forming an organometallic compound.…
Q: Which of the following compounds will react least rapidly when treated with CH3CH2Cl and AICI3?…
A:
Q: The K₂ of a weak monoprotic acid is 1.74 x 10-5. What is the pH of a 0.0939 M solution of this acid?…
A:
Q: Consider the reaction CO +BH3 → H3B-CO If the temperature of the reaction is increased would you…
A: This is a basics in inorganic chemistry. Here we have to use the concept of HSAB Concept.
Q: Rank the solubilities (from least to most soluble) of BaCO3, CdS, NICO3, AgCN, and SnS and explain…
A:
Q: A solution is prepared by adding Cu2+, S2-, Na+, NO3-, and PO43- ions. Identify each of the…
A:
Q: Consider the element Osmium (No. of Electrons = 76) a. Write the electronic configuration…
A:
Q: Which is the correct half reaction for the oxidation of S₂03² to S406²?
A:
Q: The major product that would result from the reaction scheme shown below is? ОА. OB. COOH CH3 H3C -…
A:
Q: A sample of methane of mass 45.00 g initially occupies 13.11 L at 310.0 K. Calculate the work done…
A: here we are required to find the work done when a gas expands isothermally.
Q: .) Compound A simultaneously undergoes a pseudo first order reaction and a second-order reaction, so…
A:
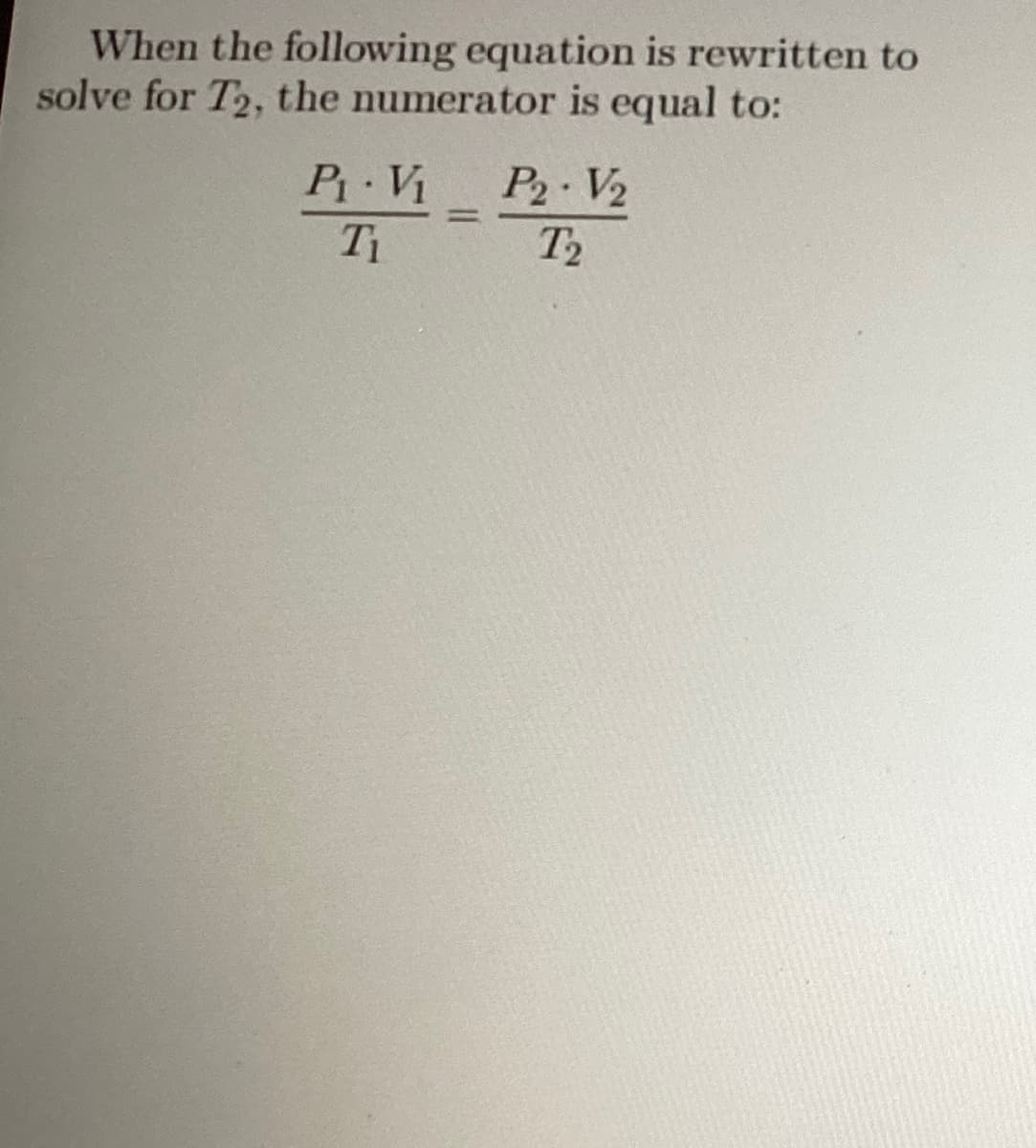
Step by step
Solved in 2 steps with 1 images

- Is this correct or does the true value need the same sig figs as the estimated value?Which of the following has the smallest value of IE1? O S Se TeThis is not correct, if you don't use sig figs until the end and get 1.89882 = 1.898 it is NOT correct, how do I do this problem, because this isn't correct.
- 2Cr(s) + 3Cu2+(aq) ⟶ 2Cr3+(aq) + 3Cu(s) Calculate the EcellThe results of experimental measurements on %SO3 in a sample repeated five times were the following: 24.24, 24.36, 24.91, 24.20, 24.10 Verify whether the third value, which seems to be high compared to the others, should be considered as an outlier. Use the Grubbs Test at 95% confidence level.it's not 1.33 either
- With respect to the rules of sig figs explained in the first image, is 8.96 g/cm3 the correct answer or would it need to be two sig figs instead?Suppose two different states (with no degeneracy so that g1=g2=1) have energies E2=2x10-22 J and E1=0.5x10-22 J. At what temperature will N2 have a population so that the rato of N2/N1=100? Suppose now that the degeneracies are g1=3, g2=2. Is it possible to find a temperature where the population in N2 is larger than N1?Chemistry The units for molar absorptivity are L/cm Mol Cm Mol/L Mol/L cm Unitless
