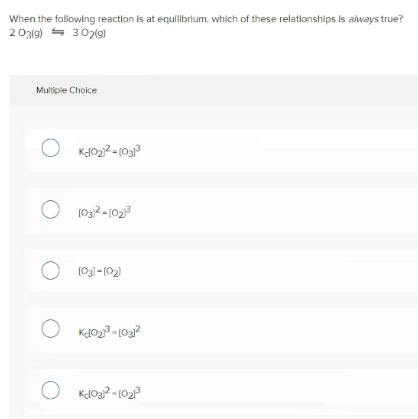
When the following reaction is at equilbrlum, which of these relationships is always true? 203lg) - 302(9) Multiple Choice O Kdo22 = [03 O 1032-1023 O 103l-(02) O Kdoa? - 102
Q: Write the balanced NET ionic equation for the reaction when Li,sO, and Ca(NO,), are mixed in aqueous…
A: We would solubility rules to predict the products formed when given compounds are mixed.
Q: How will the system respond to an increase in the amount of S? S(s) + 0,(g) = SO,(g) AH < 0 Select…
A:
Q: Which of the following can increase the observed potential in a galvanic cell? a. ohmic…
A: Correct option is D.
Q: CH3OH(g)⇌CO(g)+2H2(g)CH3OH(g)⇌CO(g)+2H2(g) (Note that…
A: we need to calculate ΔG for the given reaction
Q: 103 IR LUU 13C NMR ppm 209.13 178.49 50 42.35 32.95 29.87 18.64 4 D00 3000 1000 e000 1500 S00…
A: The proton and carbon NMR helps us to find the structure of any given unknown organic compound by…
Q: Question 18 Calculate the standard cell potential, E°cal, for the voltaic cell based on the…
A:
Q: 4 Find the nuclear binding energy of potassium-40 (atomic mass = 39.9632591 amu) in units of joules…
A:
Q: 25. A 25 mL sample of Ca(OH)2 was analyzed with a 0.10 M HCl standard. If 30 mL of the acid standard…
A: Given that: Molarity of acid(M1) = 0.10M Volume of acid (V1) = 30 ml Valume of sample with…
Q: True/False 4.184 joules equal 1 kilocalorie carbon atom can form 4 bonds 31. 32. 33. molar mass of…
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost…
Q: Question 11 Which of the following would be the correct equation for determining the number of moles…
A: Choose the correct expression for number of moles ---
Q: 12.0 g of sodium acetate is added to 125 mL of a 0.83 M acetic acid solution. What is the pH of this…
A: pH of buffer solution is calculated by using Henderson Hassel balch equation
Q: Calculate for the mechanical energy of an object that has a potential energy of 12 kJ and a kinetic…
A:
Q: Balance the reactions below using the change in oxidation number method. 1. РЬО, + HBr - PbBr, + O,…
A: Hii there, As there are multiple sub parts posted. we are answering first-three sub parts . If you…
Q: help with mechanism plz NH3 -он 2 CI 'N' S. H. NaOH Он
A: The reaction given is,
Q: The decomposition of antimony pentachloride (SbCl5) is described by the following equation:…
A: 1. Given :- SbCl5(g) ⇔ SbCl3(g) + Cl2(g) [SbCl5] = 3.557 M [SbCl3] = 1.181 M [Cl2] = 4.189 M…
Q: Part A MISSED THIS? Read Section 6.6 (Pages 228-235): Watch KCV66 What is the total pressure of the…
A: Given: partial pressure of N2 = 239 torr The partial pressure of O2 = 105 torr And partial pressure…
Q: If 46.3 mL of a 0.794 M Ca(OH)2 solution is required to neutralize a 40.0 mL sample of HCl, what is…
A: Given :- Molarity of Ca(OH)2 solution = 0.794 M Volume of Ca(OH)2 solution = 46.3 mL Volume of…
Q: *Given the following information, answer the questions 6 and 7 Acid pKa Acid NH3 HC=CH pKa CH:CH:OH…
A:
Q: Consider the following system at equilibrium where AH° = 87.9 kJ/mol, and K. = 1.20×10-2 , at 500 K.…
A: The equilibrium reaction given is, Given: 0.35 moles of PCl5 is removed from the equilibrium…
Q: You are synthesizing aspirin. You start with 3g of salicylic acid (mm 138g/mol). If you end with…
A: Given that the synthesis of aspirin from salicylic acid. Also, given that the amount of salicylic…
Q: A chemistry student weighs out 0.0690 g of hypobromous acid (HBrO) into a 250. mL volumetric flask…
A: Given: Mass of HBrO = 0.0690 g. And concentration of NaOH = 0.1700 M
Q: Which of the following aqueous solutions are good buffer systems? O 0.33 M ammonia + 0.39 M sodium…
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The…
Q: What is the charge on X in Na2[X(PO4)3]? Type your answer.
A: Charge on Na is +1 Charge on (PO4)3 is -3
Q: Sodium carbonate (MM = 105.988 g/mol) is a primary standard base that reacts with hydrochloric acid.…
A:
Q: A sample of oxygen produced in a laboratory experiment had a volume of 493 mL at a pressure of 575…
A:
Q: In the laboratory, a general chemistry student measured the pH of a 0.545 M aqueous solution of…
A: Given :- Concentration of acetic acid solution = 0.545 M pH of solution = 2.490 To calculate :-…
Q: A fertilizer advertised as 8-8-8 generally means that it contains 8% total nitrogen, 8% P2O5, and 8%…
A: Given: The fertilizer contains 8% of P2O5 and 8 % of K2O. Assuming 100 g of the fertilizer. Hence…
Q: Which of the following molecules has an assumed van't Hoff factor equal to 17 a. MgS0+ b. NaHS04…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: The concentration of hydrogen peroxide decreases from O. 1 mol/L to 0.05 mol/L in 900 s at 240C on…
A: a ) Half life is defined as the time required to reduce the concentration of a substance to half .…
Q: A (3.6500 g) of impure ammonium aluminium sulfate (NH4AI(SO4)2)) was treated with ammonia (NH3(aq))…
A: Given: Mass of sample = 3.6500 g. Mass of Al2O3 formed = 0.4935 g.
Q: For pH = 6.00, calculate the alpha one (a1) in an aqueous solution of citric acid. (Ka1 = 7.45 x…
A: PH of solution is 6. K1,k2,k3 are dissociation constant of citric acid.
Q: Part C A sample of neon initially has a volume of 2.50 L at 20. °C. What final temperature, in…
A: Charle's law
Q: As a result of alpha emission, the product nucleus is
A:
Q: Solar energy is: O the energy stored within the structural units of chemical substances. O the…
A: To define: Solar energy.
Q: the
A: According to the question, we need to determine which of the following electrochemical methods…
Q: Question 10 Based on this equation, how many moles of NaOH react with 1 mole of H2SO4? H2SO4 + 2NAOH…
A: Given : Moles of H2SO4 = 1 mole
Q: The weight of helium in a balloon can be estimated by calculating the volume of a sphere with the…
A: Formula used: Valume of sphare: 4/3πr3 Where r = radius of sphare Mass = volume* density Unit…
Q: Write a balanced half-reaction describing the oxidation of solid magnesium to aqueous magnesium…
A: Given: Oxidation of magnesium to magnesium cation.
Q: A chemist adds 0.30 L of a 2.04M sodium thiosulfate (Na, S,0,) solution to a reaction flask.…
A:
Q: 800 705 kJ 600 497 kJ PE (kJ) 400 200 175 kJ 100 Reaction Progress Created by E. Lee for Virtual…
A: Given: potential energy of activated complex without any catalyst = 705 KJ. And the potential energy…
Q: Lab para-iodo nitrobenzene If aniline was used instead of p-nitro aniline, comment on which of…
A:
Q: Which of the following parameters was held constant in the Styrofoam calorimetry experiment?…
A: Given: Styrofoam calorimetry experiment.
Q: You measure out 42 mL of 6.0 M HNO3 to make the 250.0 mL of dilute solution. What volume of…
A: Given, Vacid = 42 ml Vsolution = 250.0 ml Vwater = ?
Q: An ingredient of Tiger Balm salve is composed of C, H and O. A 0.1005 g sample of the ingredient is…
A: Given : Weight of sample = 0.1005 g Weight of CO2 produced = 0.2829 g Weight of H2O produced =…
Q: A (3.6500 g) of impure ammonium aluminium sulfate (NH4Al(SO4)2)) was treated with ammonia (NH3(aq))…
A: Given: Mass of sample = 3.6500 g. Mass of Al2O3 formed = 0.4935 g.
Q: What is the percent of CaCO3 in an antacid given that a tablet that weighed 1.3198 g reacted with…
A:
Q: A voltaic cell utilizes the following reaction: 4FE²+(aq) + O2(g)+4H*(aq)→4Fe3+(aq) + 2H2O(1)…
A: The balanced reaction taking place is given as, => Given: Eocell = 0.46 V [Fe2+ ] = 1.8 M, [Fe3+…
Q: 4.A(aq) + 3B(aq) = C(aq) + 8D(aq) AG355K =? The reaction above was run at 388K and allowed to come…
A: The reaction given is, => And the equilibrium concentration of species at 388 K are,
Q: NAOH, H20
A: Detail mechanistic pathway is given below to find out the product
Q: Matching the following plants with the heavy metals they absorb v Hydrangeas A. Chromium ]Water…
A: Hydrangeas's flower colors ultimately depend on the availability of aluminum ions and hence it…
Step by step
Solved in 2 steps with 1 images

- Calculate ΔrG° for the following reaction at 387 K. HCN(g) + 2H2(g) → CH3NH2(g) ΔrH°= -158.0kJmol-1; ΔrS°= -219.9JK-1 mol-1Consider this scenario and answer the following questions: Chlorine atoms resulting from decomposition of chlorofluoromethanes, such as CCl2F2, catalyze the decomposition of ozone in the atmosphere. One simplifiedmechanism for the decomposition is:O3 ⎯⎯⎯⎯⎯⎯⎯→ sunlight O2 + OO3 + Cl ⟶ O2 + ClOClO + O ⟶ Cl + O2(a) Explain why chlorine atoms are catalysts in the gas-phase transformation: 2O3 ⟶ 3O2(b) Nitric oxide is also involved in the decomposition of ozone by the mechanism:O3 ⎯⎯⎯⎯⎯⎯⎯→ sunlight O2 + OO3 + NO ⟶ NO2 + O2NO2 + O ⟶ NO + O2Is NO a catalyst for the decomposition? Explain your answer.Prof. Neiman and Prof. James were first to discovered in 1973 that chlorofluorocarbons (CFCs) were depleting the Earth’sozone layer when released into the atmosphere. Once they reach the stratosphere, Clis released from the CFCs molecules by interaction with UV light. Free Cl atoms areable to react with ozone in a catalytic cycle that converts O3into the more stable O2.It is estimated that a single Cl atom is able to react with∼100000 O3molecules.Although CFCs production was banned in 1996, there are still a substantial numberof motor vehicle air conditioners (MVACs) that use CFC-12 (CF2Cl2) as refrigerant.The average CFC-12 emission rate from operating MVACs has been estimated tobe 59.5 mg per hour per vehicle (Zhang et al.Environ. Sci. Technol. Lett.2017).How much chlorine, in kg, is added to the atmosphere in a year due to 100 millionMVACs using CFC-12 as refrigerant?
- The value of ΔS° for the catalytic hydrogenation of acetylene to ethane, C2H2 (g) + 2H2 (g) → C2H6 (g) is __________ J/K∙ mol. A) -76.0 B) +440.9 C) -232.5 D) +232.5 E) +28.7For the reaction 5Ce4+ + Mn 2+ + 4H2 O → 5Ce3+ + MnO4- + 8H+ , given E0 (Ce4+ /Ce3+ ) = 1.70V,E0 (MnO4- /Mn2+ ) = 1.507V, calculate ΔG0 for this reaction ?Write the expression for Kc for the following reactions. Ineach case indicate whether the reaction is homogeneousor heterogeneous.(a) 3 NO1g2 ∆ N2O1g2 + NO21g2(b) CH41g2 + 2 H2S1g2 ∆ CS21g2 + 4 H21g2(c) Ni1CO241g2 ∆ Ni1s2 + 4 CO1g2(d) HF1aq2 ∆ H+1aq2 + F-1aq2(e) 2 Ag1s2 + Zn2+1aq2 ∆ 2 Ag+1aq2 + Zn1s2(f) H2O1l2 ∆ H+1aq2 + OH-1aq2(g) 2 H2O1l2 ∆ 2 H+1aq2 + 2 OH-1aq2
- For the arrhenius equation what are the other deviations for the formula like the ones i have to solve for t1 and k1.The vapor-liquid reactive equilibria for ethyl lactate synthesis follows the expressionlog K = 7.893 – (2.4312 x 103)K/Tbetween 83.12 °C and 101.54 °C. What are the ΔH°rxn, ΔG°rxn, and ΔS°rxn at 85 °C?Ind. Eng. Chem. Res. 2008, 47, 5, 1453–1463What process of catalysis is 2SO2(g) + O2(g) → 2SO3(g) with V2O5 called?
- Which of the following is a concerted reaction with a ΔG < 0? (Hint: Exergonic reaction)One of the many remarkable enzymes in the human bodyis carbonic anhydrase, which catalyzes the interconversionof carbon dioxide and water with bicarbonate ionand protons. If it were not for this enzyme, the body couldnot rid itself rapidly enough of the CO2 accumulated bycell metabolism. The enzyme catalyzes the dehydration(release to air) of up to 107 CO2 molecules per second.Which components of this description correspond to theterms enzyme, substrate, and turnover number?Calculate the rate of methane oxidation by OH (in units molecules cm-3 s-1) using the k at T=298 and pressure = 1 atm = 2.4x1019 molecules cm-3. What type of reaction is this? Assume: [OH] = 1 x 106 molecule cm-3 at all altitudes CH4 mixing ratio = 1.8 ppm (parts per million) = 1.8 umol mol-1 at all altitudes Use the Arrhenius expression k=A e-(Ea/RT) where k is the rate constant, Ea is activation energy, R is the gas constant (in appropriate units), and T is temperature in Kelvin. Arrhenius parameters for the reaction of OH with CH4: -Pre-exponential factor: A= 2.45x10-12 cm3 molec-1 s-1 -Exponential factor: Ea/R = 1775 K

