Q: Which of the groups below is capable of only hydrophobic interactions? a b c d e
A: The answer is as follows:
Q: III. PRACTICE EXERCISE 3. Answer the following questions in the provided space provided: 1. Which is...
A: The bond longer or shorter based on the bond length which is inturn dependent on the strength of the...
Q: Categorize each alcohol below as primary, secondary or tertiary OH OH OH
A: Identify which type of alcohol is present--
Q: determine whether they are the same, diasteromers, enantomers
A:
Q: a. Calculate the number of moles of HCI(g) that must be added to 1.0 L of 1.1 M NaC,H3O2 to produce ...
A:
Q: Select the reactions below that are elementary reactions as written. Select all that apply. NO2(g) +...
A: Solution Given that 2 NO2(g) → NO3(g) + NO(g) Rate = K[NO2]² Hence option (C)...
Q: 3 mol % Bi(NO3)3•5H,O Co H2O, rt HO,
A: The reaction given is,
Q: What is the mass % of carbon in dimethylsulfoxide, C2H6SO, rounded to three significant figures?
A:
Q: Question 38 of 39 Submit > What is the correct IUPAC name for the compound shown here? 2,3-2-2,2,3-2...
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: 4. The following data were obtained in calibrating a calcium ion electrode for the determination of ...
A: a) Answer - the pCa vs potenttial data are plotted below as scatter point . Aline is drawn through t...
Q: line angle structural form of 5-cyclobutyl-2-cyclopropyl-6,8-diethyl-3,7,7-trimethyldecane
A:
Q: A 24.817 gram sample of water with specific heat of 4.184 J/g0C changes temperature from 25.00 0C to...
A: Given :- Mass of water = m = 24.817 g Initial temperature of water = Ti = 25.00 °C Final temperatu...
Q: 2. Name the following hydrocarbons using the IUPAC system: CH3(CH2)2CH3 СНЗСН2СI а. b. C. CH,CH=CHCC...
A: "To get remaining sub-parts solved please repost the complete question and mention the sub-parts to ...
Q: HBr a)
A: protonation of etheral oxygen loss of a group as alcohol leading to the formation of carbocation at...
Q: Write an equilibrium constant expression, Kc, for the following reactions: (CH3)2CO (l) ⇆ (CH3)2CO ...
A: Active mass of pure liquid component is equal to 1
Q: If 0.025 mol of COCl2 (g) is placed in a 1.0L container at 4000C, 16.0% of the COCl2 (g) is dissocia...
A:
Q: My professor said the answer choice was A.) please explain why in detail. Which of the following so...
A:
Q: n the presence of excess thiocyanate ion, SCN-, the following reaction is first order in iron(III) ...
A:
Q: a) (C2H5)2Be + Be(CH3)2 b) R2BH + R-CH=CH2 c) MgEt2 t-BUOH
A:
Q: Either sulfuric acid or bismuth(III) nitrate pentahydrate can catalyze this reaction. What is an adv...
A: Either sulfuric acid or bismuth(III) nitrate pentahydrate can catalyze this reaction.we have to desc...
Q: 2. Three immiscible liquids are added to a tall graduated cylinder. The three liquids are hexane (d ...
A: a.) To draw a diagram for layers of three liquids , we would use the fact that liquid having highes...
Q: If a solid AB2 is added to water, and at equilibrium the concentration of A2+ is 6.23 x 10, what is ...
A: Solubility product is the parameter to understand the amount of solubility of a salt.
Q: Let Us Apply Element, 1 Sea Water 6. Bronze 2. Titanium 7. Krypton 3. Rubidium 8. Sulfuric acid 4. G...
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: . Let’s say you did this experiment again and got to the drying /filtering out the drying agent step...
A: (a) During the working up of a reaction, In the step which involves the addition of the drying agent...
Q: Match Column A with Column B. Mobile phase is more polar than the stationary phase A. Reverse Phase ...
A: Explanation is on the image. 1)-‐---A 2)-----A 3)-----B 4)-----B
Q: Identify the reactant that is a Brønsted–Lowry acid and Brønsted–Lowry conjugate base in the reactio...
A:
Q: QUESTION 20 Match following waste with the proper waste container. Used capilary tube after m.p. det...
A: 20 . ) To match the wastes with correct container , we would check the type of waste produced in al...
Q: You are asked to identify an unknown salt. You weigh out 5.00 grams of the unknown, and place it in ...
A: Answer is as follows. :
Q: 1. the amount of N2 is decreased? 2. the heat of the system is increased? 3. an inert gas with press...
A: Le Chatelier's principle states that if a dynamic is disturbed by changing the conditions, the posit...
Q: エZ
A: Here we have to synthesize the given compound by suitable starting materials.
Q: Each row of the table below describes an aqueous solution at about 25°C . Complete the table. That...
A: pH is negative logarithm of hydrogen ions
Q: Atomic Atomic Mass symbol number Protons Neutrons Electrons Number
A: The missing elements details are given below
Q: Given the following reactions 2NO → N2 + O2 AH = -180 kJ 2NO + O2 → 2NO2 AH = -112 kJ
A: Solution Given that 2NO N2 + O2 ∆H = -180 kJ 2NO + O...
Q: To synthesize Tylenol, p-aminophenol will be acetylated using 1.4 equivalents of acetic anhydride. S...
A: Given: Mass of p-aminophenol = 2.1 g In order to synthesize, Tylenol, p-aminophenol will be acetylat...
Q: Calculate the molal concentration, molar concentration, and mole fraction of 6.25% solution of H3PO4...
A: Given, Mass percentage of H3PO4 = 6.25% Density of the solution = 1.06 g/mL Calculate the molal c...
Q: Which one of the following could be a product of the reaction shown below? OH H* catalyst ??? HO. OH...
A:
Q: What is the expected boiling point of a solution prepared by dissolving 4.28 g of sodium iodide (Nal...
A:
Q: Answer the following comprehensively based on what you have learned on the properties of liquids and...
A: 1. a) Answer - According to the question - Surface tension - Surface tension is the tendency of liqu...
Q: 1) Explain why when performing stoichiometric calculations, it is important to use a balanced equati...
A: A balanced equation has the same number of atoms on the both sides of the equation. It tells the exa...
Q: Write the balanced NET ionic equation for the reaction when aqueous aluminum nitrate and aqueous pot...
A:
Q: Using only sigma overlap parameters, a 5-coordinate dº complex TABLE 10.10 Angular Overlap Parameter...
A: In the given problem the preferential geometry of a 5-coordinated d6 complex is needed to determined...
Q: HI b)
A:
Q: Construct a (011) within a unit cell.
A: A lattice plane in a unit cell is those plane whose intersections with the lattice are periodic.
Q: 7. At room temperature cyclopentadiene reacts with itself to form dicyclopentadiene in a Diels-Alder...
A: The answer is given below
Q: Calculate the standard enthalpy of reaction at 333 K for A(s) + 2B(g) -> C(g) given that the standar...
A:
Q: Gaseous ammonia chemically reacts with oxygen (0,) gas to produce nitrogen monoxide gas and water va...
A:
Q: What is the mass % of carbon in dimethylsulfoxide, C2H6SO, rounded to three significant figures?
A: Since you have posted multiple questions as per guidelines we can answer only one per session . If y...
Q: The hydroxide ion concentration, [OH-], of an aqueous solution of 0.429 M methylamine (a weak base w...
A:
Q: Q2/B/A Compound (A) mass of (109.23) composed of 78.46% C, 8.46%H and 13%N mixed with 40.77gm O. fin...
A: Step 1 : Determination of mass Mass of mixture = 109.23 gm Carbon mass = 78.46 % of 109.23 ...
Q: he highest solubility in u
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

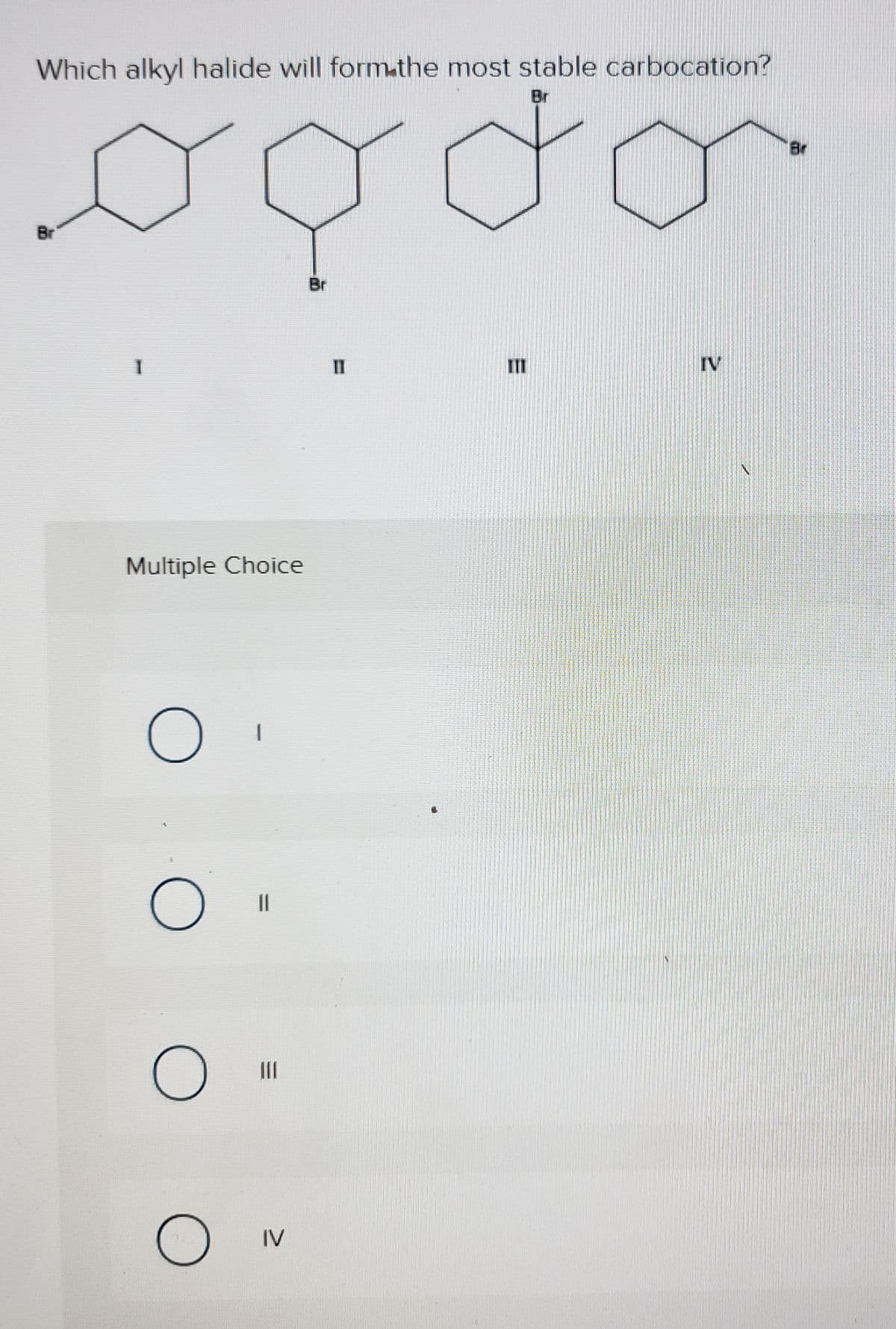
- Molecular halogen (X2) adds to alkenes to make vicinal dihalides. In the boxes, draw thecurved arrow notation for the reaction.(b) Consider the reaction of 1-bromobutane with a large excess of ammonia (NH3). Draw the reactants, the transition state, andthe products. Note that the initial product is the salt of an amine (RNH3+ Br - ), which is deprotonated by the excess ammonia to give the amine.Rank the following carbocations from most stable to least stable:
- 1. When you add H-Br in a terninal alkene, the product has the rule od Markovnikov? 2. The type pf intermetary thag forms when we add HgSO4/H2SO4/H2O in an alkyl is? Please solve my both questions.thank you.Which alkene choices and reaction conditions created this product?Which of the following alkenes (a-c) reacts faster with HI?
- What other alkene is also formed along with Y in Sample Problem 9.3? What alkenes would form from X if no carbocation rearrangement occurred?plz shw the steps include alkens and alkynesWhich of the following alkene addition reactions is regioselective?A) HalogenationB) Hydration C) Both A and BD) Neither A and B
- What steps are needed to convert Y to the alkene Z ?Consider a reaction where cis-but-2-ene is treated with sO followed by NaHSO/H, O. Draw the structure of one product that is formed in the reaction, including correct stereochemistry.What alkene is the major product formed from attached alkyl halide in an E1reaction?

