Chemistry for Engineering Students
3rd Edition
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter5: Gases
Section: Chapter Questions
Problem 5.23PAE
Related questions
Question
7
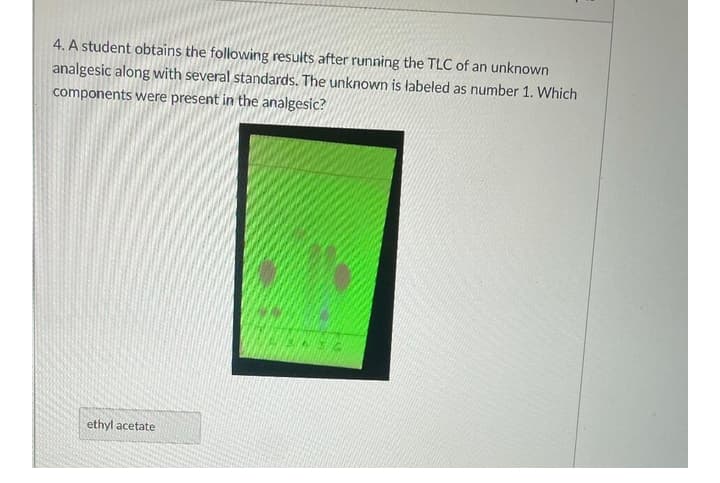
Transcribed Image Text:4. A student obtains the following results after running the TLC of an unknown
analgesic along with several standards. The unknown is labeled as number 1. Which
components were present in the analgesic?
ethyl acetate
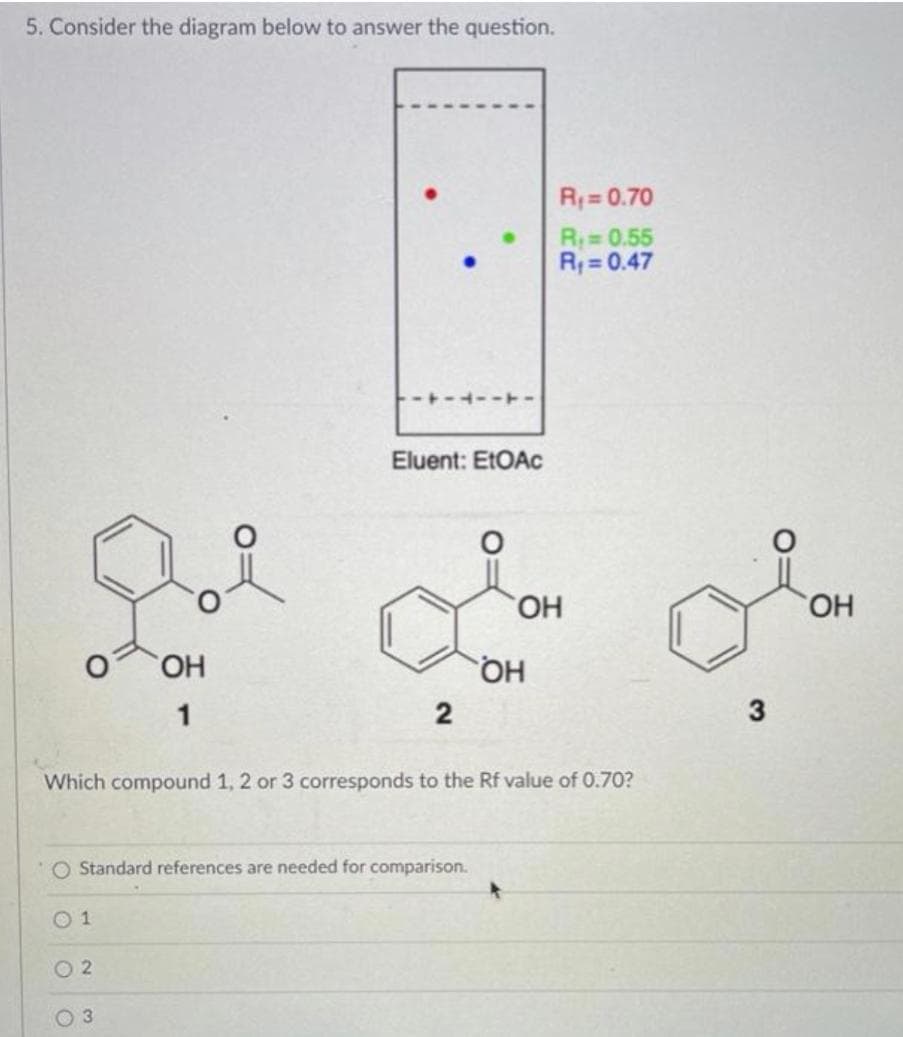
Transcribed Image Text:5. Consider the diagram below to answer the question.
R;= 0.70
R0.55
R; = 0.47
Eluent: EtOAC
HO,
HO.
HO.
HQ
1
2
3
Which compound 1, 2 or 3 corresponds to the Rf value of 0.70?
O Standard references are needed for comparison.
O 1
O 2
O 3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning