Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter6: Alkanes & Alkenes
Section: Chapter Questions
Problem 3E: Using your model of butane (CH3CH2CH2CH3) , complete the following graph of the anglebetween the two...
Related questions
Question
Which corresponds to the best answer?
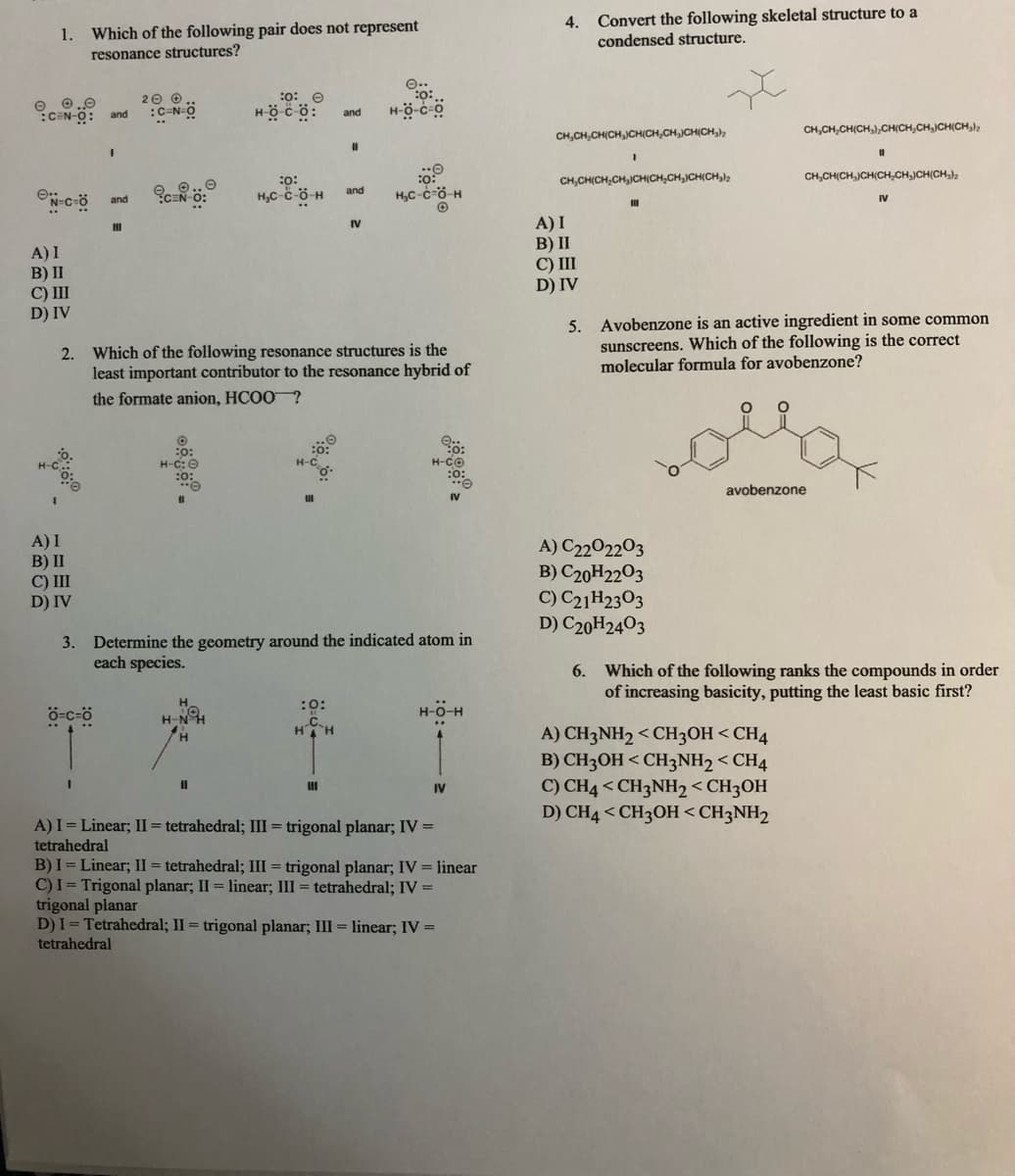
Transcribed Image Text:4.
Convert the following skeletal structure to a
1.
Which of the following pair does not represent
condensed structure.
resonance structures?
:0: 0
O 0.0
:CEN-O: and
20 0
:C-N-Ö
Höcö:
and
CH,CH,CH(CH,JCH(CH,CH,JCH(CH,)
CH,CH,CH(CH,),CH(CH,CH,JCH(CH,);
CH,CH(CH,CH,JCH(CH,CH,CH(CH,
CH,CH(CH JCH(CH,CH,)CH(CH,),
:o:
H,C-c-ö-H
and
H,C-c-ö-H
and
IV
A) I
B) II
C) III
D) IV
IV
A) I
B) II
C) III
D) IV
5. Avobenzone is an active ingredient in some common
sunscreens. Which of the following is the correct
molecular formula for avobenzone?
2. Which of the following resonance structures is the
least important contributor to the resonance hybrid of
the formate anion, HCOO?
:o:
4-C:0
H-CO
avobenzone
A) I
B) II
C) II
D) IV
A) C2202203
B) C20H2203
C) C21H2303
D) C20H2403
3. Determine the geometry around the indicated atom in
each species.
6. Which of the following ranks the compounds in order
of increasing basicity, putting the least basic first?
:0:
ö-c-ö
H-N
H-O-H
HH
A) CH3NH2 < CH3OH <CH4
B) CH3OH < CH3NH2 < CH4
C) CH4 < CH3NH2< CH3OH
D) CH4 < CH3OH<CH3NH2
IV
A) I = Linear; II = tetrahedral; III = trigonal planar; IV =
tetrahedral
B) I = Linear; II = tetrahedral; III = trigonal planar; IV = linear
C)I = Trigonal planar; II = linear; III = tetrahedral; IV =
trigonal planar
D)I= Tetrahedral; II = trigonal planar; III = linear; IV =
tetrahedral
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 4 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
