Which element will produce a new compound when added to a beaker containing an aqueous solution of copper (II) sulfate (CusO)? Reactivity Series for Metals Most Reactive Least Reactive K Ca Na Mg Al Zn Fe Ni Sn Pb H Cu Ag Au Pt A Fe Ag Cu None of the above.
Which element will produce a new compound when added to a beaker containing an aqueous solution of copper (II) sulfate (CusO)? Reactivity Series for Metals Most Reactive Least Reactive K Ca Na Mg Al Zn Fe Ni Sn Pb H Cu Ag Au Pt A Fe Ag Cu None of the above.
Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.23QAP
Related questions
Question
100%
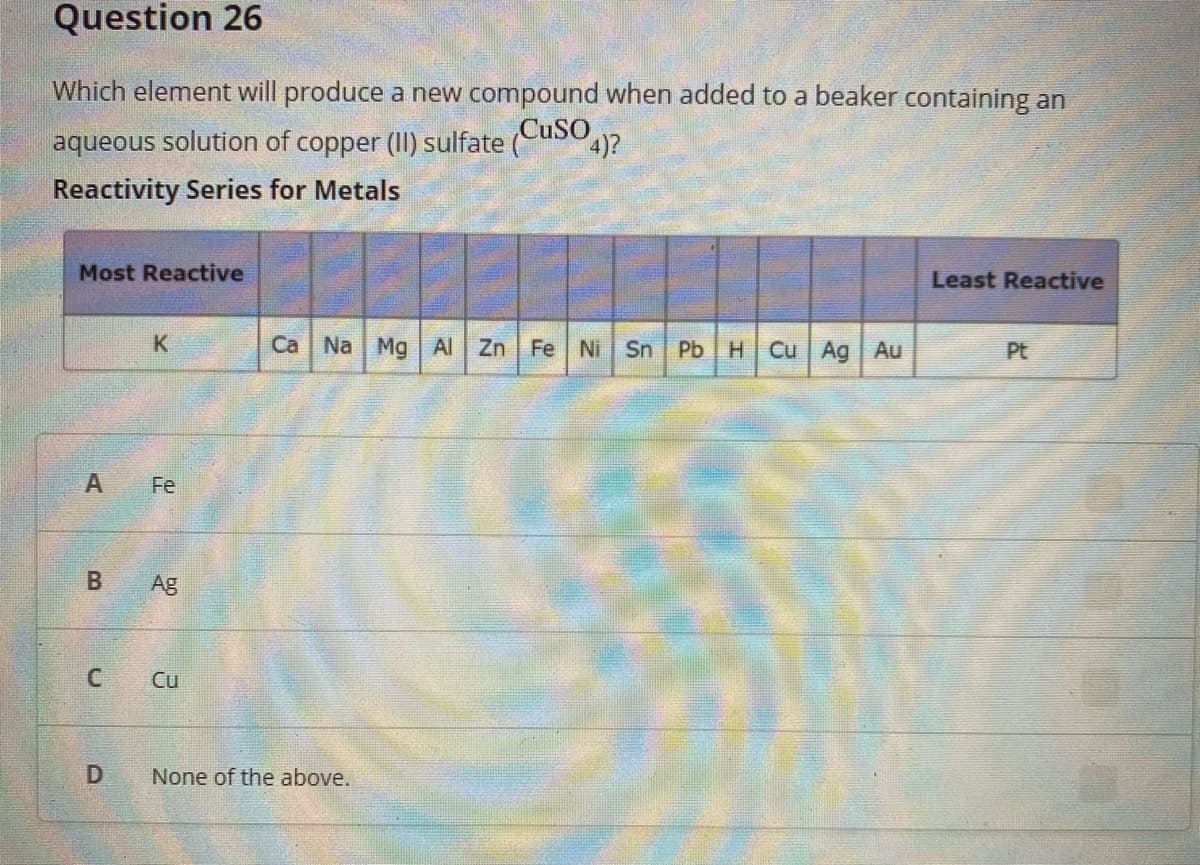
Transcribed Image Text:Question 26
Which element will produce a new compound when added to a beaker containing an
aqueous solution of copper (II) sulfate (CusO?
Reactivity Series for Metals
Most Reactive
Least Reactive
K
Ca Na Mg Al Zn Fe Ni Sn PbH Cu Ag Au
Pt
Fe
Ag
Cu
D
None of the above.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

