Categorize the following reactions: 3. N, (g) + 3H, (g) = 2NH; (g) 4. С.Н,О, (аq) -> 2C,Н.О (аq) +2С0.(g) 5. Ca (s) + Zn(NO,), (aq) --> Ca(NO,), (aq) + Zn (s) 3 2
Categorize the following reactions: 3. N, (g) + 3H, (g) = 2NH; (g) 4. С.Н,О, (аq) -> 2C,Н.О (аq) +2С0.(g) 5. Ca (s) + Zn(NO,), (aq) --> Ca(NO,), (aq) + Zn (s) 3 2
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 29Q: When jump-starting a car with a dead battery, the ground jumper should be attached to a remote part...
Related questions
Question
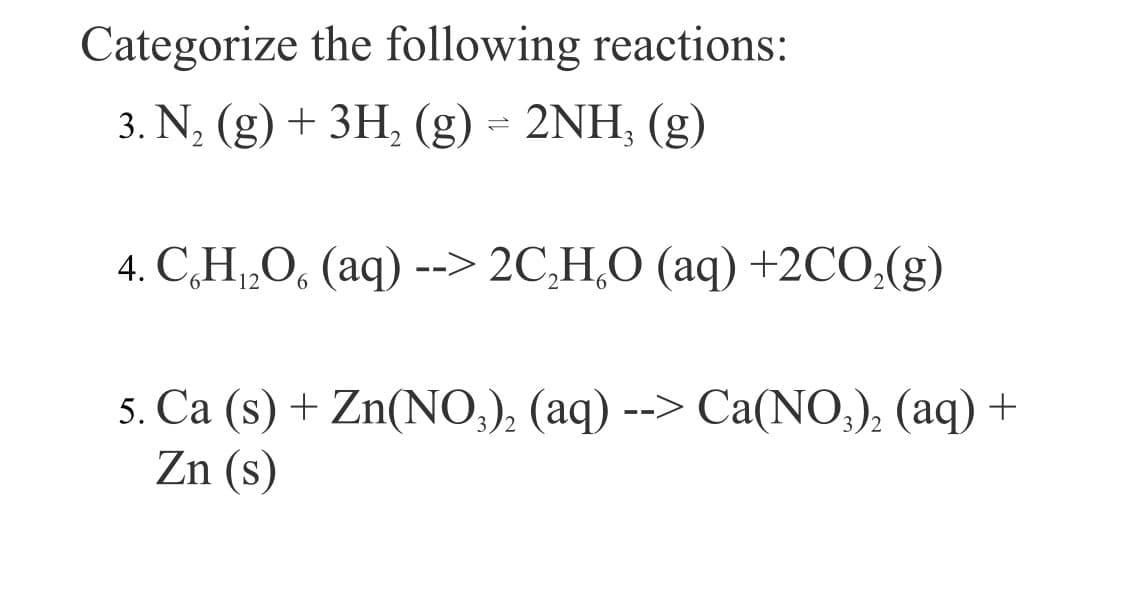
Transcribed Image Text:Categorize the following reactions:
3. N, (g) + 3H, (g) = 2NH, (g)
4. C,H„O, (aq) --> 2C,H,O (aq) +2CO,(g)
5. Ca (s) + Zn(NO,), (aq) --> Ca(NO;), (aq) +
Zn (s)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
