Which elements can commonly form more than one type of cation? (consult table 2.11) 01. Hydrogen O 2. Helium O 3. Lithium 0 4. Beryllium O 5. Boron O 6. Carbon 07. Nitrogen 0 8. Oxygen O 9. Fluorine O 10. Neon O 11. Sodium O 12. Magnesium O 13. Aluminum O 14. Silicon O 15. Phosphorus O 16. Sulfur O 17. Chlorine O 18. Argon O 19. Potassium O 20. Calcium O 21. Scandium O 22. Titanium O 23. Vanadium O 24. Chromium O 25. Manganese O 26. Iron O 27. Cobalt O 28. Nickel O 29. Copper O 30. Zinc O 31. Gallium O 32. Germanium O 33. Arsenic O 34. Selenium O 35. Bromine O 36. Krypton O 37. Rubidium O 38. Strontium O 39. Yttrium O 40. Zirconium
Which elements can commonly form more than one type of cation? (consult table 2.11) 01. Hydrogen O 2. Helium O 3. Lithium 0 4. Beryllium O 5. Boron O 6. Carbon 07. Nitrogen 0 8. Oxygen O 9. Fluorine O 10. Neon O 11. Sodium O 12. Magnesium O 13. Aluminum O 14. Silicon O 15. Phosphorus O 16. Sulfur O 17. Chlorine O 18. Argon O 19. Potassium O 20. Calcium O 21. Scandium O 22. Titanium O 23. Vanadium O 24. Chromium O 25. Manganese O 26. Iron O 27. Cobalt O 28. Nickel O 29. Copper O 30. Zinc O 31. Gallium O 32. Germanium O 33. Arsenic O 34. Selenium O 35. Bromine O 36. Krypton O 37. Rubidium O 38. Strontium O 39. Yttrium O 40. Zirconium
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter5: Nomenclature
Section: Chapter Questions
Problem 10CR: Are most elements found in nature in the elemental or the combined form? Why? Name several elements...
Related questions
Question
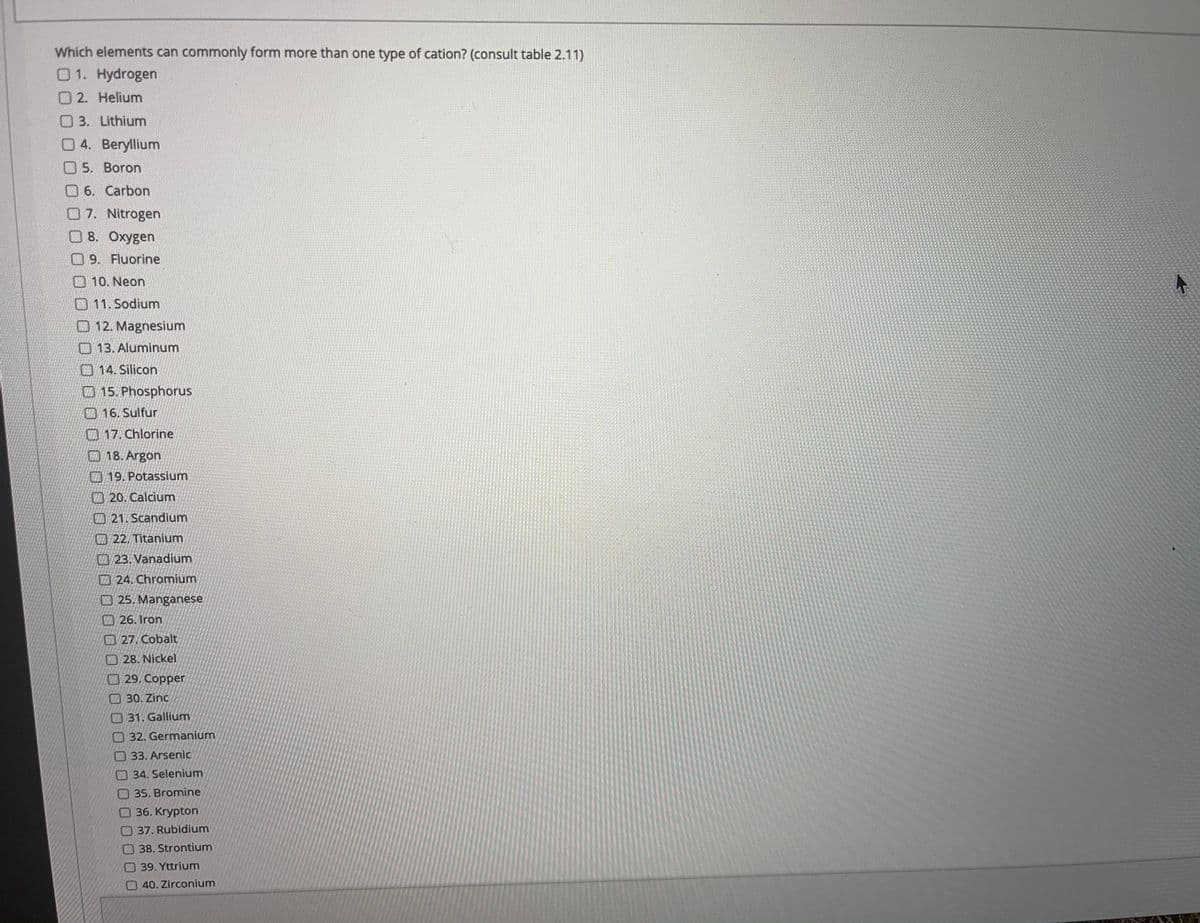
Transcribed Image Text:Which elements can commonly form more than one type of cation? (consult table 2.11)
01. Hydrogen
O2. Helium
O 3. Lithium
04. Beryllium
5. Boron
O 6. Carbon
07. Nitrogen
8. Oxygen
09. Fluorine
O 10. Neon
11. Sodium
O 12. Magnesium
O 13. Aluminum
14. Silicon
15. Phosphorus
16. Sulfur
17. Chlorine
18. Argon
VE19. Potassium
20. Calcium
21. Scandium
A22. Titanium
23. Vanadium
24. Chromium
O 25. Manganese
26. Iron
27. Cobalt
7 28. Nickel
29. Copper
O 30. Zinc
O 31. Gallium
32. Germanium
33. Arsenic
O34. Selenium
O 35. Bromine
O36. Krypton
37. Rubidium
38. Strontium
39. Yttrium
O40. Zirconium
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning