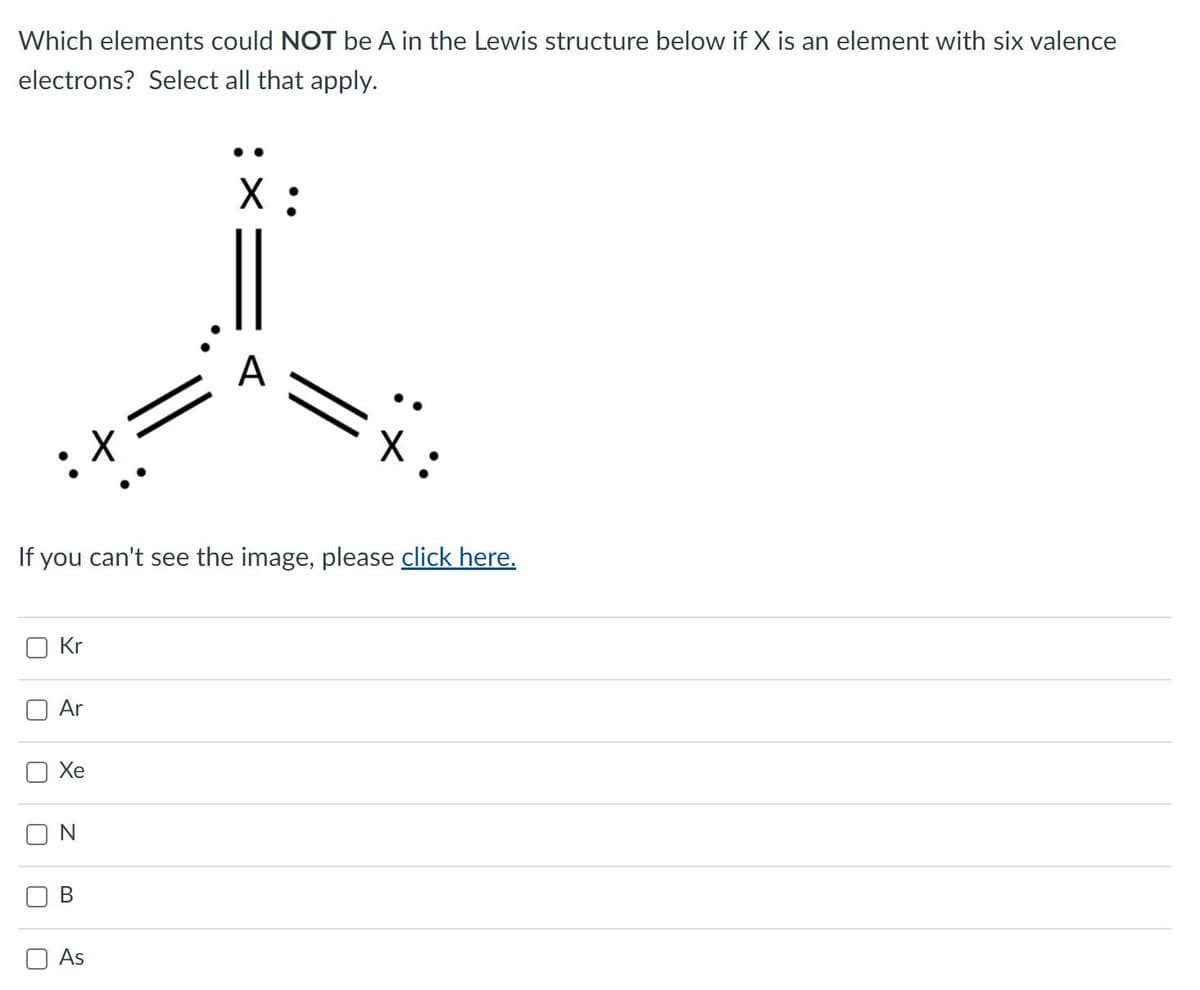
Which elements could NOT be A in the Lewis structure below if X is an element with six valence electrons? Select all that apply. X : A If you can't see the image, please click here. Kr Ar Хе O N В As
Q: Draw a Lewis structure for H2S Fill in the following information as well: 1. Formula Process Work…
A: The electronic structures of the elements, especially the electron pairings, are reflected in the…
Q: Question attached
A: Lewis Structure is a representation of the valence shell electrons in a molecule. These Lewis…
Q: Which of the following species are not a resonance forms of the anionic species in the box? HO A C…
A: Resonance structure are those which involves delocalization of charge and unsaturation over the…
Q: Draw a Lewis structure for CI0, in which Cl is the center atom with 10 valence electrons. Based on…
A: Resonance structure of ClO3- is given by,
Q: Is the proposed Lewis structure reasonable? proj V Lewis structure Yes. O No, it has the wrong…
A: Following are the appropriate Lewis structures of the given compounds.
Q: Consider an ion with the symbol Z- and the electronic configuration 1s22s22p63s23p6. a. What is the…
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for…
Q: Can you explain why these Lewis Structures are wrong? I was told I was correct that BrF5 has 42…
A: The nature of the bonding pattern associated with the molecule and the presence of non-bonding…
Q: NF3 CS2 Substance H,O2 H. Lewis structures apters in the text. 主
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: a) Draw the Lewis structure of H3PO4. b) In H3PO4, one P-O bond has a different bond length than the…
A: There are three polar and one slight polar bond present in the molecule of H3PO4. The polar bonds…
Q: Which of the molecules and polyatomic ions cannot be adequately described using a single Lewis…
A: Resonance structures have different Lewis structures that means one Lewis structure can not…
Q: Draw the lewis structure of the following ionic compounds: a. Sodium Permanganate b. Beryllium…
A:
Q: Xenon is a noble gas, but it forms a number of stable compounds. We examined XeF4 earlier. What are…
A: Lewis structures of XeF2 and XeF6 :
Q: Draw the Lewis structure(s) for the molecule with the chemical formula C,HN where the N is connected…
A: In lewis structure each atom try to fill octets rule. Two double bonds are present in the lewis…
Q: A В Original C D
A:
Q: A) 24 B) 20 25 D) 28 E) 21 ice ele SO, and then draw the corresponding Lewis structure (by following…
A: The number of valence electrons in SO3 is
Q: A Lewis structure with placeholder central atom is shown below. If the charge of the molecule is -1,…
A: Given information:The Lewis structure of unknown molecule is represented as follows:
Q: For the molecule, Sel4: 1) Draw the Lewis structure then state the molecular arrangement and shape.…
A: Solution -
Q: (A) Write the chemical names for the following chemical compounds. a) Table sugar b) Dry ice c) (B)…
A: Given information, (A) Chemical names (B) Bond formation by Lewis diagram
Q: Question 3
A: The electronic configuration of sulfur and hydrogen atom is,S = 1s22s22p63s23p4H = 1s1Sulfur has 4…
Q: You are given the below hypothetical Structure. The electronegativity values of the atoms are given…
A:
Q: -> i app.101edu.co I Apps G M Gmail D YouTube O Maps a AMAZON A Translate A Gflights S USCIS b…
A: In a Lewis structure the partial positive and partial negetive chage over atoms forming a bond…
Q: N - ö = F: I. Incorrect number of valence electrons II. Incorrect central atom III. Octet rule not…
A: Lewis structures also known as Lewis dot diagrams or electron dot structure are the diagrams that…
Q: A student proposes the following Lewis structure for the carbon dioxide (CO2) molecule. 0 = :0:
A: We have to find formal charges on O,C,O in CO2 molecules
Q: 2. The skeletal structure of acetic acid shown below is correct, but some of the bonds are wrong.…
A: The Lewis structure is the simplified structure of a molecule representing its valance electrons.
Q: Z is a fictitious element . If Z is an element of group , the molecule ZH 2 has how many lone pairs
A: Given: Z is a fictitious elements. Molecule formed is hydride of Z. To find: Lone pairs on the…
Q: 88. Determine the Bond Character of the following compounds. In the first blank place the…
A: The difference in the electronegativity between the two bonded atoms can determine whether the bond…
Q: Decide whether the Lewis structure proposed for each molecule is reasonable or not. molecule BeH₂ OF…
A:
Q: Lewis Structure Formula a. Al*3 and O -2 b. Sn*2 and I c. Au *3 and F
A:
Q: Draw the Lewis structure of the following substances a) ammonia (NH3) b) ammonium ion (NH4*) c)…
A: Lewis structure is the representation of a chemical element or a compound that shows the bonding…
Q: proposed Lewis structure Is the proposed Lewis structure reasonable? Yes. No, it has the wrong…
A: The solution is given below -
Q: Which of the species listed has a Lewis structure with a resonance structure? Select all that apply.…
A:
Q: An unknown molecular compound has the following Lewis structure. Which of the following elements…
A: In the given Lewis structure, -C=X part shows that X is capable of forming two bonds with C.
Q: Which atom in the C-F bond has a partial negative charge (δ⁻)?
A: Please find your solution below : When a more electronegative atom is attached to a less…
Q: Which of the following atoms can have an expanded valence shell? Select all that can. F Хе Br ON
A: Expanded octet: For any element to complete the octet, it must have 8 valance electrons. But in some…
Q: formula for the compound that forms between the two elements listed. Express your answer as a…
A: According to Lewis theory metal loses one or more than one electron and form cation, whereas…
Q: Which of the following atoms could not accommodate an expanded octet (i.e., more than eight valence…
A:
Q: Which elements could be X in the Lewis structure below? Select all that apply. 3- :0 If you can't…
A: 1- C and N both are wrong these can not be X element because both elements can not contains…
Q: 2) Which is not included on the em specturm? A) UV b) radio waves c) sound waves d) infrared 3)…
A: 2) Electromagnetic waves are also known as EM waves that are formed when an electric field comes in…
Q: Write the LEWIS DOT STRUCTURE for the following: A. Sodium, Na B. Aluminum, Al C. Chlorine
A:
Q: Which of the following atoms could not accommodate an expanded octet (i.e., more than eight valence…
A: Which of the following atoms could not accomodate an expanded octet ?
Q: Find the molecule that does not follow octet principle. O PH3 O BF3 O PC|3 O CF4
A: According to the octet rule, atoms or elements combined together either by sharing electrons or by…
Q: . Given the following atom: (1) NCl3. (2) OCS, C is the central atom a. Write the Lewis structure.…
A: Lewis structure of : NCl3 , OCS Resonating structure of : CO32-
Q: Write the Lewis dot structure for lonic compound lons symbols Ratio of ions Lewis symbol Aluminum…
A: Hi, since you have posted multiple questions, we will answer the very first question for you. Kindly…
Q: 1. Draw the Lewis structure to the following compounds. For each compound, state whether the bonding…
A: The main types of chemical bonds are ionic bond , covalent ,hydrogen and metallic bonds . A bond…
Q: 7. Identify the element Xwhich forms a polyatomic ion with the following Lewis Structure? [:CI-X-Cl:…
A: Lewis’s structure: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a…
Q: Which of the following elements are capable of expanding their octet? Select all that apply. C. S.…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Can someone just confirm my answer, did I draw the Lewis structure correct with line pairs.
A: No, the Lewis structure made by you is incorrect as the lone pairs of I are missing. Please find…
Q: 1. A) Predict formulas for all compounds that you might reasonably expect to form between each of…
A: Valence electrons in Se is 6 Valence electrons in O is 6 Valence electrons in Sn is 4 Valence…
Q: How many total valence electrons would you need to account for when drawing a Lewis Structure for…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Draw Lewis diagrams for the two resonance forms of thenitrite ion, NO2-. In what range do you expect the nitrogen–oxygen bond length to fall? (Hint: Use Table 3.6.)1a) Create a drawing of the lewis structure of the ion ?3 2+, show your work (show all your work, total electron count, etc). b) How many total nodes are present in a 4d orbital? How many are radial and how many are angular?Draw the Lewis structures for all seven diatomic elements (H2, N2, O2,F2, Cl2,Br2, I2) . Even though Br and I are not in the first 3 periods it is useful to consider them here.
- Which of the following Lewis Dot Structures is definitely incorrect? Group of answer choices C A B D EI need help ASAP Website for first simulation: https://www.learner.org/wp-content/interactive/periodic/bondin The second/last simulation: Open the following website: https://javalab.org/en/ion_model_en/ As seen in the table, compounds containing an integer ratio of elements depend on how many cations combine with how many anions to form a stable compound. For example, in table 1, to form a NaCl compound, first Na ionizes from the Na+ cation which is having +1 positive charge, which will lose 1 electron, it is having +1 charge, this electron then goes to Cl and it will change to Cl- anion by gaining the electron, so here 1:1 ratio charge which means 1 Na+ combines with 1 Cl-1 to form NaCl. so here integer ratio is 1:1 for this sodium chloride compound. As listed in table 2, Mg(OH)2, the integer ratio is 1:2 which means, Mg is a neutral atom that loses 2 electrons and forms an Mg+2 cation, which combines with OH which gains 1 electron to form OH- anion, So here Mg2+ can combine with 2…Draw the correc (best) Lewis structure (LS) for the ionic compound K2SeO3 (on looose leaf paper--NOT TO BE SUBMITTED) and then answer the following questions.(NOTE: Use the cardinal numbers 0, 1, 2, 3, and so on for any quantity required)a) What charge does the K ion have in this compound? (give size & sign of charge: e.g., 1+, 2-, etc.) b) What charge does the SeO3 ion have in this compound? (give size & sign of charge: e.g., 1+, 2-, etc.) c) How many lines, if any, attach each K ion to the SeO3 ion? d) How many valence electrons does the SeO3 ion (alone) have? e) How many single bonds does the LS of the SeO3 ion have? f) What is the number of lone electron pairs in the LS of the SeO3 ion?
- The formal charge of oxygen in HO− is _____. The number of valence electron of O in HO− is _____. The number of nonbonding electrons of O in HO− is _____. Thenumber of bonding electrons of O in HO− is _____. What are the answers for the Blanks A, B, C, D? Write the answers in a numerical form, not with letters, without any space.Consider the following ion: BrO3−. a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3− and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3−? What is its molecular shape? e) Does BrO3− have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−? Briefly explain your answer. g) Which of the following molecules would you expect to have the lowest vapour pressure? Briefly explain your choice. h) What is the molecular formula for Compound C? What is the empirical formula for Compound C? Please andwer f, g and h the image is for g and hConsider the following ion: BrO3−. a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3− and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3−? What is its molecular shape? e) Does BrO3− have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−? Briefly explain your answer. g) Which of the following molecules would you expect to have the lowest vapour pressure? Briefly explain your choice. h) What is the molecular formula for Compound C? What is the empirical formula for Compound C? please answer c, d and e
- Consider the following ion: BrO3−. a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3− and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3−? What is its molecular shape? e) Does BrO3− have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−? Briefly explain your answer. g) Which of the following molecules would you expect to have the lowest vapour pressure? Briefly explain your choice. (IMAGE WITH POSSIBILITIES) h) What is the molecular formula for Compound C? What is the empirical formula for Compound C?Consider the following ion: BrO3−. a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3− and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3−? What is its molecular shape? e) Does BrO3− have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−? Briefly explain your answer. h) What is the molecular formula for Compound C? What is the empirical formula for Compound C?Consider the following ion: BrO3 − . a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3 − and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3 − ? What is its molecular shape? e) Does BrO3 − have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3 − to have longer or shorter bonds than BrO3 − ? Briefly explain your answer. g) Which of the following molecules would you expect to have the lowest vapour pressure? Briefly explain your choice. h) What is the molecular formula for Compound C? What is the empirical formula for Compound C?

