Which hydrogen atom(s) in the following molecule is acidic? (That is, which hydrogen atom(s) will disassociate in water?) ОН 3. Н 2. H. H C no acidic hydrogens O 2 only O 1 and 3 O 3 only O 1,2, and 3 O 2 and 3 O 1 and 2 O 1 only Submit Request Answer
Which hydrogen atom(s) in the following molecule is acidic? (That is, which hydrogen atom(s) will disassociate in water?) ОН 3. Н 2. H. H C no acidic hydrogens O 2 only O 1 and 3 O 3 only O 1,2, and 3 O 2 and 3 O 1 and 2 O 1 only Submit Request Answer
Comprehensive Medical Assisting: Administrative and Clinical Competencies (MindTap Course List)
6th Edition
ISBN:9781305964792
Author:Wilburta Q. Lindh, Carol D. Tamparo, Barbara M. Dahl, Julie Morris, Cindy Correa
Publisher:Wilburta Q. Lindh, Carol D. Tamparo, Barbara M. Dahl, Julie Morris, Cindy Correa
Chapter22: The Patient History And Documentation
Section: Chapter Questions
Problem 10CR
Related questions
Question
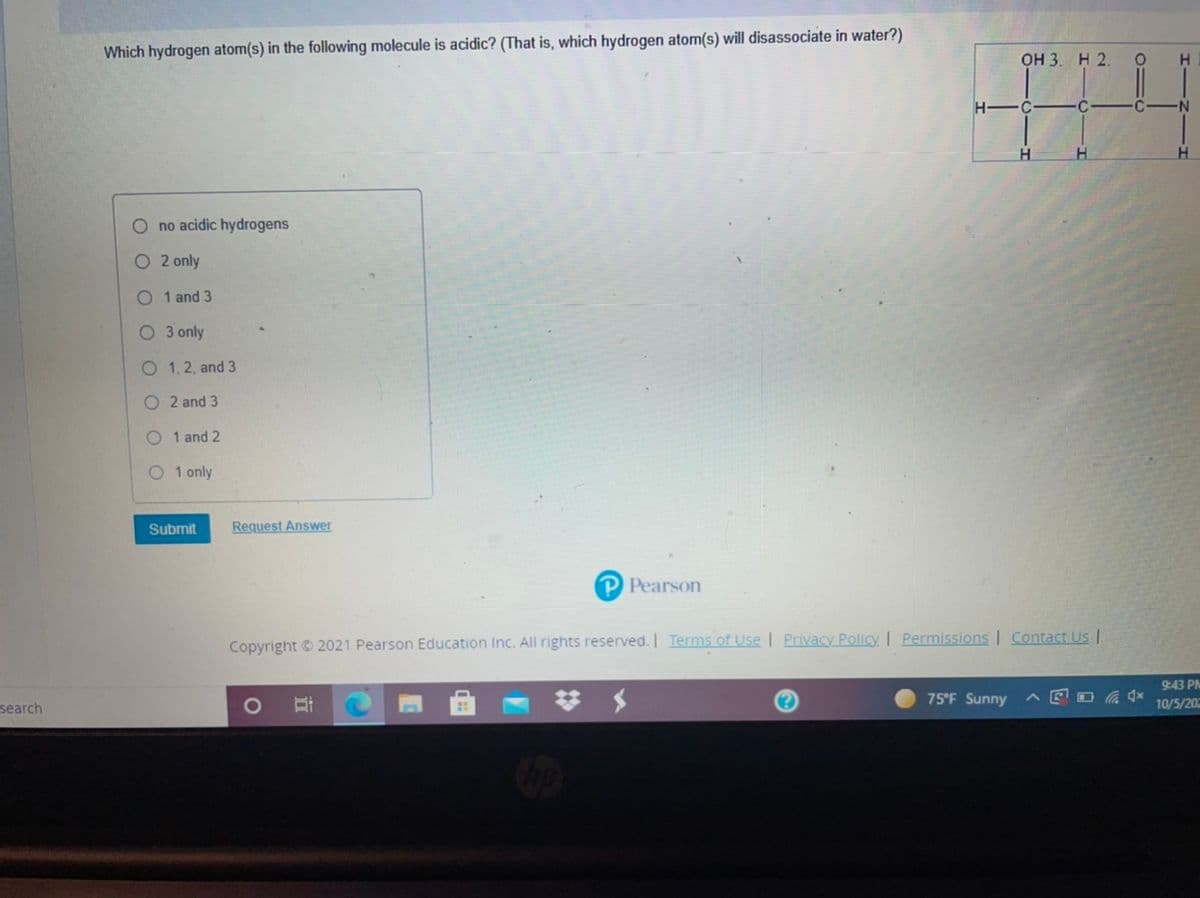
Transcribed Image Text:Which hydrogen atom(s) in the following molecule is acidic? (That is, which hydrogen atom(s) will disassociate in water?)
оН 3. Н 2.
N-
H
O no acidic hydrogens
O 2 only
O 1 and 3
O 3 only
O 1, 2, and 3
O 2 and 3
O 1 and 2
O 1 only
Submit
Request Answer
P Pearson
Copyright © 2021 Pearson Education Inc. All rights reserved. I Terms of Use | Privacy Policy | Permissions | Contact Us I
9:43 PM
75°F Sunny
search
10/5/20
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Comprehensive Medical Assisting: Administrative a…
Nursing
ISBN:
9781305964792
Author:
Wilburta Q. Lindh, Carol D. Tamparo, Barbara M. Dahl, Julie Morris, Cindy Correa
Publisher:
Cengage Learning


Comprehensive Medical Assisting: Administrative a…
Nursing
ISBN:
9781305964792
Author:
Wilburta Q. Lindh, Carol D. Tamparo, Barbara M. Dahl, Julie Morris, Cindy Correa
Publisher:
Cengage Learning
