Which is the correct sequence, from left to right, to separate the following ions from each other, and from the solution? T/I CATIONS High Solubility (aq) Low Solubility (s) Cl, Br, I Halides Most Ag+, Pb²+, Tl+, Hg²+, Hg, Cu Co+2/Cr+3/Sr+2 / Sn+4 S²/SO4²/CT/CO3² Nb +5 / Sn+2/Pb+2 / Mg*2 Cat2/Sn+2/Ag*/Pb+² Mg +2 / Fe+3 / Ba*2/Pb+2 Be +2 / Sn+2/Pb+2/Nb+5 Be+2/Pb+2 / Ag+/Ca+² Solubility Table Group 1, Nhì, Group 2 Most ANIONS OH Group 1, NH₂, Sr², Ba²+, TL- Most SO4²- Most Ag+, Pb²+, Ca²+, Ba²+, Sr²., Ra² CO₂², PO₂³, SO3²- C₂H₂O₂ NO3 Group 1, Most All NH₂ Most All Group 1 compounds, including acids, and all ammonium compounds are assumed to have high solubility in water. Ag+ None
Which is the correct sequence, from left to right, to separate the following ions from each other, and from the solution? T/I CATIONS High Solubility (aq) Low Solubility (s) Cl, Br, I Halides Most Ag+, Pb²+, Tl+, Hg²+, Hg, Cu Co+2/Cr+3/Sr+2 / Sn+4 S²/SO4²/CT/CO3² Nb +5 / Sn+2/Pb+2 / Mg*2 Cat2/Sn+2/Ag*/Pb+² Mg +2 / Fe+3 / Ba*2/Pb+2 Be +2 / Sn+2/Pb+2/Nb+5 Be+2/Pb+2 / Ag+/Ca+² Solubility Table Group 1, Nhì, Group 2 Most ANIONS OH Group 1, NH₂, Sr², Ba²+, TL- Most SO4²- Most Ag+, Pb²+, Ca²+, Ba²+, Sr²., Ra² CO₂², PO₂³, SO3²- C₂H₂O₂ NO3 Group 1, Most All NH₂ Most All Group 1 compounds, including acids, and all ammonium compounds are assumed to have high solubility in water. Ag+ None
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter15: Equilibria Of Other Reaction Classes
Section: Chapter Questions
Problem 5E: What additional information do we need to answer the following question: How is the equilibrium of...
Related questions
Question
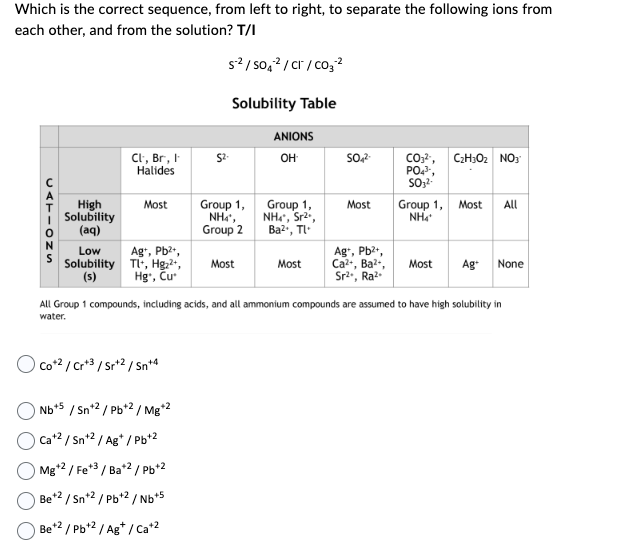
Transcribed Image Text:Which is the correct sequence, from left to right, to separate the following ions from
each other, and from the solution? T/I
CATIONS
High
Solubility
(aq)
Low
Solubility
(s)
Cl, Br, I
Halides
Most
Ag+, Pb²+,
Tl+, Hg₂²+,
Hg, Cu
Co+2 / Cr+3/Sr+2 / Sn+4
S²/SO4²/CT/CO₂ ²
Nb+5 / Sn+2/Pb+2 / Mg+2
Ca+² / Sn+² / Ag*/Pb+2
Mg+2 / Fe+3 / Ba+2/Pb+2
Be +2 / Sn+2/Pb+2/Nb+5
Be+2 / Pb+² / Ag+ / Ca+2
Solubility Table
Group 1,
Nhì,
Group 2
Most
ANIONS
OH
Group 1,
NH4*, Sr²*,
Ba²+, Tl
Most
SO4²-
Most
Ag+, Pb²+,
Ca²+, Ba²+,
Sr²., Ra².
CO₂²¹, C₂H302 NO3
PO4³-,
SO3²-
Group 1,
NH4*
Most All
All Group 1 compounds, including acids, and all ammonium compounds are assumed to have high solubility in
water.
Most Ag+ None
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
