Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter5: Gases
Section: Chapter Questions
Problem 129E
Related questions
Question
Transcribed Image Text:Shelby Thomas - Dr. Warrick--lay x
- Shelby Thomas - Winesett-- Lay
resentation/d/1hH1ZcyareTSe8se5_uf9F3JaWOelMBbijF5vifp1Eks/edit#slide=id.ge87bdf15c6_0_71
O Present
2 Share
ett-- Layers of the Atmosphere Escape Room
Last edit was 3 minutes ago
mat Slide Arrange Tools Add-ons Help
T O- O -
Background
Layout-
Theme
Transition
1 2 3 4 S 6 7 8 9
Level 4
Q1
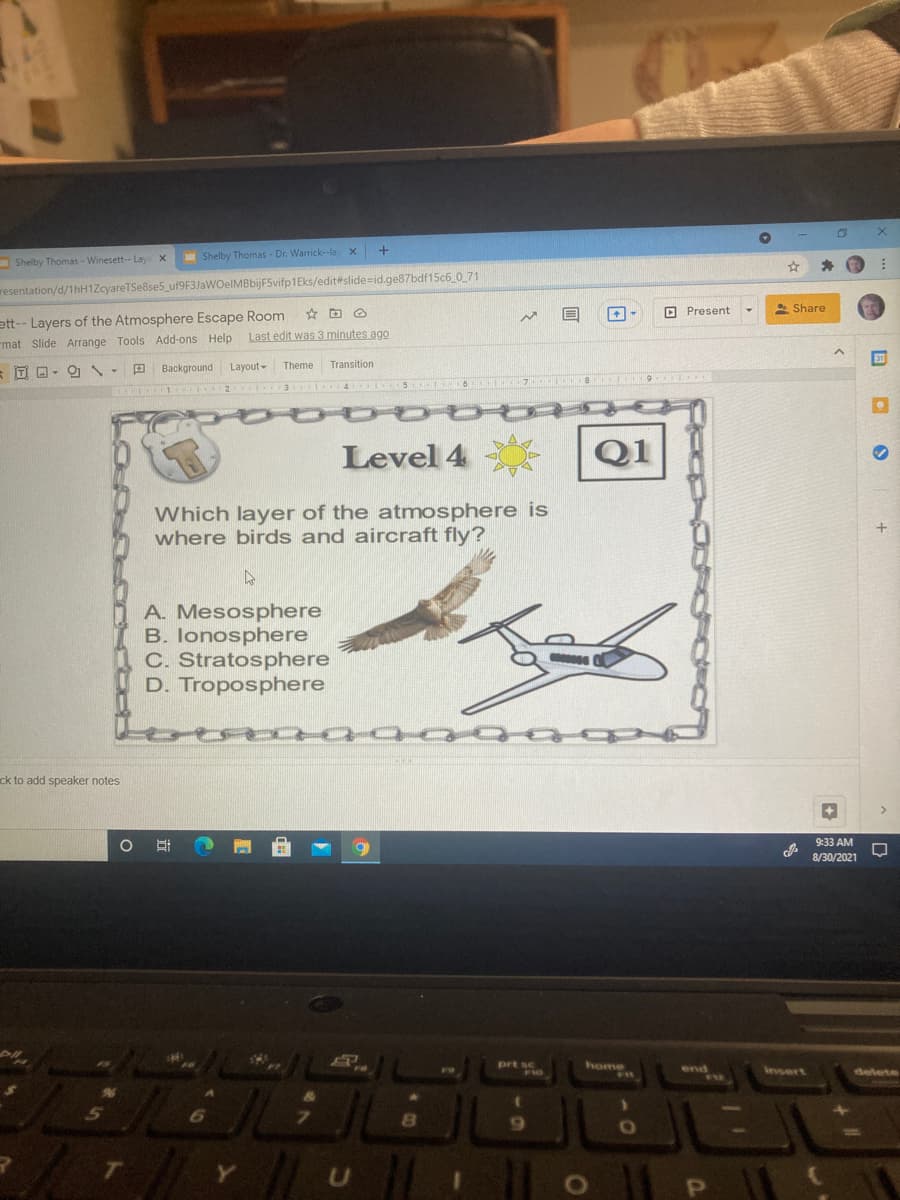
Which layer of the atmosphere is
where birds and aircraft fly?
A. Mesosphere
B. lonosphere
C. Stratosphere
D. Troposphere
ck to add speaker notes
9:33 AM
8/30/2021
prt se
home
end
insert
delete
Y
立
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
