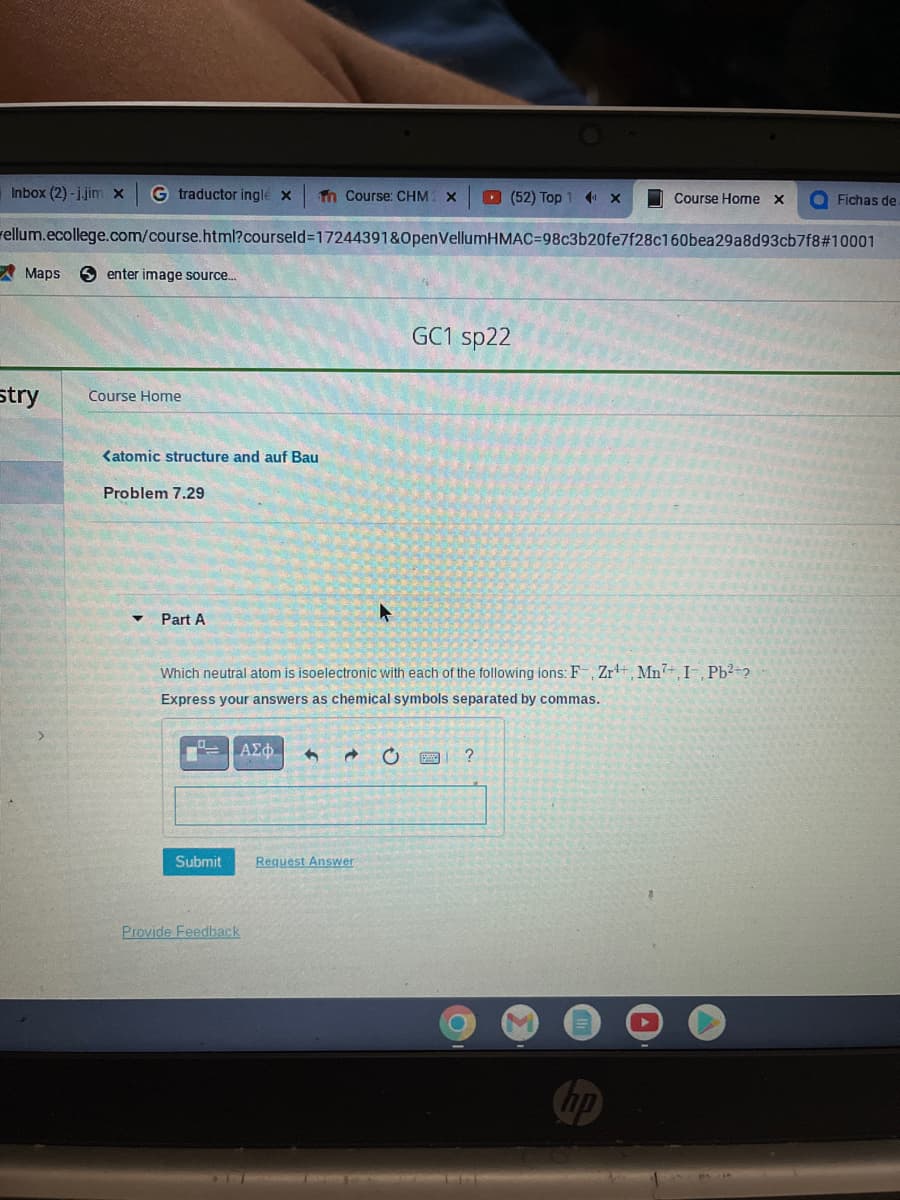
Which neutral atom is isoelectronic with each of the following ions: F, Zr, Mn, I, Pb²+? Express your answers as chemical symbols separated by commas.
Which neutral atom is isoelectronic with each of the following ions: F, Zr, Mn, I, Pb²+? Express your answers as chemical symbols separated by commas.
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.16QAP
Related questions
Question
100%
Transcribed Image Text:Inbox (2)-j.jim x G traductor inglé x in Course: CHM X
(52) Top 14 X
Course Home x
Fichas de.
ellum.ecollege.com/course.html?courseld=17244391&OpenVellumHMAC-98c3b20fe7f28c160bea29a8d93cb7f8#10001
Maps
enter image source.....
GC1 sp22
Course Home
<atomic structure and auf Bau
Problem 7.29
Part A
Which neutral atom is isoelectronic with each of the following ions: F-, Zr¹+, Mn7+, I-, Pb²+?
Express your answers as chemical symbols separated by commas.
ΑΣΦΗ 5 a G
?
Request Answer
stry
Submit
Provide Feedback
O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



