Q: ATR works by measuring the changes that occur in an internally reflected IR beam when the beam comes…
A: We have to tell the given statement is either true or false.
Q: How many milliliters of diluent should be added to a 2.00 mL if aliquot taken from a mother liquor…
A: In case of dilution of any electrolyte, the number of moles (or millimoles) of the electrolyte will…
Q: Buffer preparation: A 6.90 g sample of sodium nitrite (NANO2, 68.995) was dissolved in about 100. mL…
A: Given, When 6.90 g sample of NaNO2 was dissolved in 100.0 mL of water and it is added to 500. mL of…
Q: hich element es each
A: Electronic configuration of element is the complete adress of electron that is electron is present…
Q: After a monosaccharide has undergone mutarotation, which of the following statements is always true…
A: First we have to discuss mutarotation. Mutarotation : The change in optical rotation of an…
Q: Convert 4.53 x 10 C to uL.
A: Given:-
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: Here we to explain solubility of different compunds in water with change of pH.
Q: A 48.5 g sample of cis-butenoic acid (C4H6O2; pKa-4.44) is dissolved in 350.0 mL of water. What is…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: The reaction of boron and chlorine can be expressed as the following chemical equation: 2 B + 3 Cl2…
A: Here we use the limiting reagent and excess reagent concept. Please read the answer carefully. In…
Q: (OH)2(aq)+2HCl(aq)CaCl2(aq)+H2O(l) An aqueous solution of Ca(OH)2with a concentration of 0.209 M…
A: Molarity of Ca(OH)2 = 0.209 M Volume of HCl = 25.00 ml Volume of Ca(OH)2= 15.93 ml
Q: A 22.3 mL sample of 0.344 M ethylamine, C2H5NH2, is titrated with 0.364 M perchloric acid. After…
A: Given, Volume of C2H5NH2 = 22.3 mL Molarity of C2H5NH2 = 0.344 M Molarity of HClO4 = 0.364 M…
Q: What is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka =6.8 x 10-4) and 0.200 mol of NaF to…
A: We have to predict the pH of the solution.
Q: In which solutions the molar solubility of NICO3 will be greater than in pure water? A: 0.010 M…
A:
Q: Show the mechanism for the reaction conducted at -5 "C in CCI, cyclohexene + bromine…
A: ->When alkene reacts with dihalogen first of all cyclohalonium ion formed. -> then opening of…
Q: s all melt at temperatures lower than their salts. ローロ B 21 (1) → 1, (3) + 2e ? Mg*(1) + 21¯(4) →…
A: As we know, in electrolytic cell , Oxidation takes place at anode and reduction takes place at…
Q: [Review Topice) Draw the major product(s) of the following reactions including stereochemistry when…
A:
Q: Acid reflux occurs when acid escapes from the __________. lungs liver muscles small…
A: Acid reflux occurs when acid escapes from the stomach. This occurs when stomach acid and other…
Q: ऊज्क १ बेलाकोक मेल्ये पलयेnळ नe ananium कल्फणले ले प्रनाजर जाव्वे जा कडडवार कि व श्वायाचे वचव षeeaसत…
A:
Q: Calculate the pH of a solution in which one normal adult dose of aspirin (670 mg ) is dissolved in…
A:
Q: equilibrium will _________. decrease the amount of product increase the amount of reactants…
A: Equilibrium is affected by temperature. For endothermic reaction, by increasing temperature…
Q: Draw the major organic product formed in the reaction. conc. H* A. OH
A:
Q: Which of the following statement(s) is/are true? (Select all that apply.) ASuniy = ASsys + ASsurr O…
A: The second law of thermodynamics predicts the spontaneous nature of a process. According to this…
Q: Select an element on the periodic table to see its electron configuration. Spectroscopic (spdf)…
A: Electrons are filled in a different atomic orbital in an atom. According to the Aufbau rule, the…
Q: Pärt B If the normal physiological concentration of HCO3 is 24 mM, what is the pH of blood if Pco,…
A: We have find out the pH of blood if PCO2 drops to 28 mmHg.
Q: Draw the starting structure that would lead to the following two products under these conditions.…
A: OsO4 react with alkene gives diol.
Q: 50.0 g of tin(II) fluoride reacts with 50.0 g of sodium phosphate. Calculate the grams of tin(II)…
A: Weight of tin(2)fluoride =50 gram Weight of sodium phosphate = 50.0 gm
Q: 1) What is the pH at the equivalence point in the titration of a 29.1 mL sample of a 0.450 M aqueous…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 23. Fill in missing reagents and products in the following multi-step synthesis. 1) Br 2) reagent/s2…
A: We have find out reagents and products in multi step synthesis.
Q: If the vaccine effectiveness towards providing protection from hospitalization is 90% which of the…
A: The statement is dealing with only vaccinated people so we can ignore the options related to…
Q: How many milliliters of 0.20M HCl is required to neutralize 80.0 mL of O.80 M NaOH?
A: Given :- molarity of NaOH solution = 0.80 M volume of NaOH solution = 80.0 mL molarity of HCl…
Q: are studying a 3.0 g sample of spinal fluid and discover that it contains 2.27 mg of glucose. What…
A: Given-> Weight of spinal fluid = 3.0 g Weight of glucose = 2.27 mg = 2.27 × 10-3 g=0.00227g (1 g…
Q: Which of the following statement(s) is/are true regarding free energy? (Select all that apply.) A…
A:
Q: What is the pH of a 0.1000M Ca(OH)2 solution? 9.50 13.30 11.50 12.30 0.70
A: Given: Molarity of Ca(OH)2 solution = 0.1000 M
Q: Question 7 of 20 Submit What is the pH of a buffer made from 0.350 mol of HBRO (Ka = 2.5 x 10-º) and…
A:
Q: CHEMWORK Check all the boxes that are true statements. (Select all that apply.) U The removal of the…
A: In this question, we have to choose the correct statement from the below boxes.
Q: Consider the following reversible processes: XY (s) = x* (aq) + Y" (aq) Ksp1 = 0.25 CD2 (s) c2+ (aq)…
A: Given that, two reversible processes are XYs↔X+aq+Y-aq Ksp1=0.25CD2s↔C2+aq+2D-aq…
Q: ___________ in the soil can prevent lakes from becoming acidified due to acid rain. sodium…
A: Limestone can be used to remove acid from the soil because in lime calcium ions can be utilized acid…
Q: A sample of an unknown gas is placed in a vessel with a volume of 7,692 mL at a temperature of 15.5…
A: 17. Given :- volume = 7692 mL pressure = 1.2 atm temperature = 15.5°C To calculate :- no.of…
Q: 34. DETAILS Calcium hydride, Cat, reacts with water to form hydregen gas Caly(s) +2 H,O) - CatOHlaa)…
A:
Q: IUPAC naming cycloalkane
A: IUPAC naming of compound...
Q: Which of the following aqueous solutions are good buffer systems? O 0.14 M hypochlorous acid + 0.18…
A: Buffer solution is combination of weak acid or base with salt of weak acid or base respectively.
Q: The carbocation intermediate formed in step 1 is resonance-stabilized. Drawa different resonance…
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: How many kilojoules of energy are generated when in 82.8 grams of CO(g) reacts according to the…
A: Solution -
Q: Calculate pl if a cell consisting of a saturated calomel reference electrode ( E = 0.244 V) and a…
A: A question based on solutions that is to be accomplished.
Q: Suppose you burn a 2.4 g sample of potato chips. You use the heat given off from that process to…
A:
Q: Hi im still confused how you get the values from the grap
A: As per the given values of K and T, lnK is computed and then a graph is plotted between lnK vs 1/T
Q: Give the electron configurations for the following atoms. Do not use the noble gas notation. Write…
A: Electronic configuration follow (n+l) rule.
Q: Part B If the normal physiological concentration of HCO3 is 24 mM, what is the pH of blood if Pco,…
A:
Q: Below is one of the polypeptide chains in the insulin structure. a. What is the amino acid residue…
A:
Q: Write the formula of the conjugate base of the Brønsted-Lowry acid, HCIO,
A: Given Bronsted - Lowry acid is HClO2 Bronsted Lowry acid => Substance which proton donar.
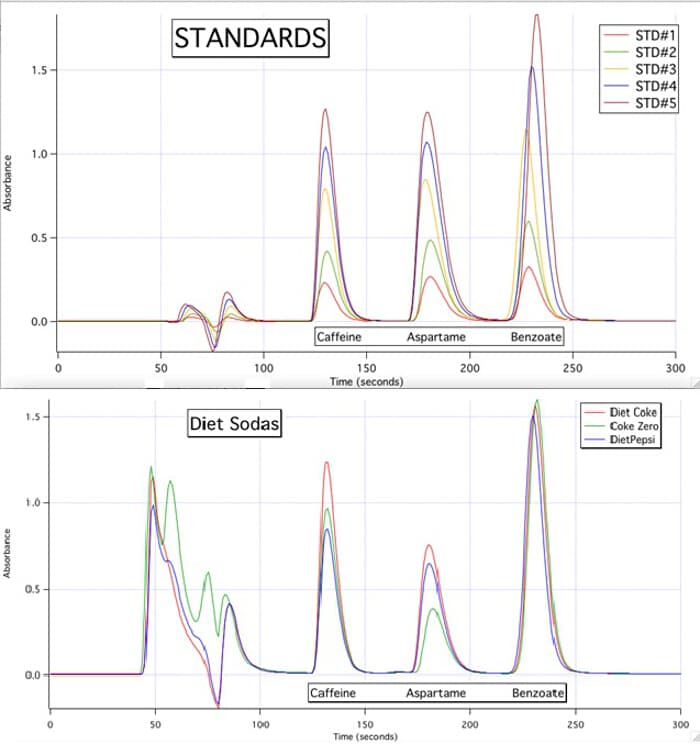
2) Which of the diet sodas have the lowest concentration of aspartame? Which standard best reflects this concentration?
Step by step
Solved in 4 steps with 3 images

- The gravimetric factor used to express CoCBr6·H20 in a sample that is finally weighed as PbClBr is choose below: FW PbClBr / 6 x FW CoCBr6·H20 FW CoCBr6·H20 / FW PbClBr FW CoCBr6·H20 / 6 x FW PbClBr 6 x FW PbClBr / FW CoCBr6·H20There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…
- If 35,000 kg of whole milk containing 4% fat is to be separated in a 6-hour period into skim milk with 0.45% fat and cream with 45% fat, what are the mass flow rates of the two output streams from a continuous centrifuge which accomplishes this separation? (Ans; Cream=464.8335kg/h, Skim milk= 5368.4998kg/h)125.1 mg of streptomycin sulphate are dissolved in 10 ml of water. A GC headspace analysis is carried out in order to determine the methanol content of the drug. A peak for methanol is produced which has 73.2% of the area of a peak for a methanol standard containing 0.532 mg/100 ml of methanol in water analysed under exactly the same conditions, What is the methanol content of the streptomycin sulphate in ppm and %w/w? Answer: 311.3 ppm, 0.3113 % w/w.how??4. A fat sample with combination of acids contain standard hydrochloric acid for blank and sample with 8mL and 5mL respectively. The normality of the standard hydrochloric acid is 0.93N and the weight of the sample is 3 grams. Calculate the saponification value.
- (6.65x10^5)/(6.60x10^-6) (3.73x10^8) Please Calaculition. Only typed solution1. Calculate the experimental density of a salt solution and the percent error (same as relative error percent) using some or all the data given below. solubility of NaCl salt in water: 0.357 g/mLmass of empty graduated cylinder: 25.19g mass of graduated cylinder + salt solution: 30.47g total volume of salt solution: 4.98 mLtrue density of salt solution: 1.07 g/mLDownvoted for wrong solution. A river is carrying water containing 2000 mg/l Magnesium Chloride into a small lake. The lake has a naturally occurring Magnesium Chloride of 50 mg/l. If the river flow is 2500 Lmin and the lake flow rate is 1.5 m³.sec¹, what is the concentration of MgCl2 in the lake after the discharge point? Assume that the flows in the river and lake are completely mixed, that the salt is a conservative substance, and the system is at steady state.
- Please answer fast it’s very important and urgent I say very urgent so please answer super super fast please For the image attached For 1. a Mass of metal: Trial 1 is 35.0228 g Trial 2 is 35.0915 g Trial 3 is 34.0821 g Mass of water: Trial 1 is 20.0177 g Trial 2 is 20.0250 g Trial 3 is 20.0168 g For delta t of water: Trial 1 is 15.5 C Trial 2 is 15.7 C Trial 3 is 15.1 C For delta t of metal Trial 1 is 80.1 C Trial 2 is 80.2 C Trial 3 is 79.5 C For B my calculated Specific heat is: Trial 1 is 0.462 Trial 2 is 0.467 Trial 3 is 0.466An analytical determination for manganese in an aqueous solution on gives thefollowing replicate measurements in ppm as 12.1, 12.3, 12.2, 12.2, 12.4, 12.3, 12.2, 12.4,12.2, and 12.5. Determine the following;a) The range of the datab) The meanc) Mediand) Standard deviatione) Variancef) Relative standard deviation RSD (ppt)An orange juice processing plant now produces essential oil from orange peels. one It is known that 250 kg of peel comes out of 1 ton of oranges and 2.5 g of essential oil comes out of 1 kg of peel. In a laboratory study, 250 g of bark was treated with hexane solvent and 0.548 g of essential oil was obtained in the sample cup of the rotary evaporator. Accordingly, the rotary Calculate the separation efficiency obtained in the evaporator?

