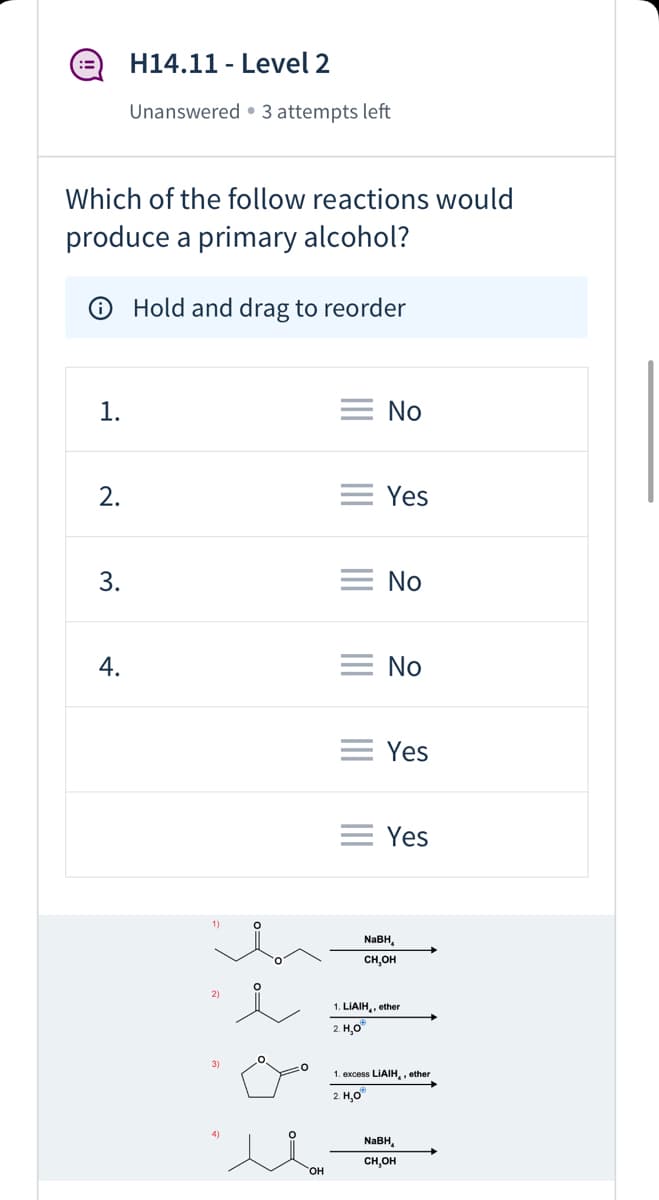
Which of the follow reactions would produce a primary alcohol? Hold and drag to reorder 1. No 2. = Yes 3. No = No = Yes = Yes 1) NABH, CH,OH 2) 1. LIAIH,, ether 2. H,0" 1. excess LIAIH,, ether 2. Н,о NABH, CH,OH но, 4.
Q: which of the following is not an alcohol? 2. CH2OHCH,OH 1. CH2=CHCH,OH 3. C6H5CH2OH 4. CGH5OH Select…
A: A chemical compound where hydrocarbon is attached to -OH functional group is called an alcohol.
Q: Which molecule shown is a secondary alcohol? O-4 O-H H-O
A: The classification of alcohol is given on the basis of the number of carbon atoms attached with the…
Q: Complete the following statements about the compounds below: OH HO ÓH A. В. C. D. 1. Compound A is a…
A: In primary alcohol , -OH attached with 1° carbon. In secondary alcohol , -OH attached with 2°…
Q: H+ НО
A:
Q: Classify the alcohol shown. CH H,C-C-CH,CH, O primary O secondary O quaternary tertiary None of the…
A: The structure of the primary alcohol is--- Here, the alcoholic -OH group attached with carbon, that…
Q: 3. Molecules with more than one alcohol group can react with thionyl chloride (SOCI,) in a way that…
A: Given is reaction between diol and thionyl chloride.
Q: the following questions. propyl alcohol? alcohol is only H-C-OH H. Methyl atcohol C- H H H HO…
A: Given structures are : a). b). c). Identify the functional groups in the given compounds = ?
Q: 1. Primary and secondary alcohols can be oxidized with chromic acid, but tertiary alcohols cannot.…
A: Oxidation of primary alcohols: primary alcohols are oxidized to aldehyde or carboxylic acid. The…
Q: HO 7. OH The above is an example of a a. primary alcohol b. secondary alcohol c. tertiary alcohol…
A:
Q: Pernyataan berikut yang manakah tidak benar mengenai alkohol dan fenol? Which of the following…
A: Given : Different different statements and reaction of alcohol. To find : incorrect statement and…
Q: Consider the five esters below with formula C ,H 1202: (a) (b) (c) (d) (e) (i) Which is/are mad e…
A: The ester is made from the condensation reaction between a carboxylic acid and an alcohol.
Q: Give each of the following a systematic name and indicate whether each is a primary, secondary, or…
A: Primary, Secondary and tertiary alcohol: Depending on the carbon (primary, secondary and tertiary)…
Q: Make sure to include all the alcohols possible n
A: Primary alcohols upon oxidation with mild oxidising agents like pyridinium chloro chromate (PCC)…
Q: 4. Based on Zaitsev's rule, write the major/minor products, as well as the side product, for the…
A:
Q: H3C-CH=CH2 H20 +
A: In the hydration reaction of alkenes, the alkene first attacks on the water molecules and takes…
Q: (e) Provide the product of each of the following reactions. (f) For each reaction, write the name of…
A:
Q: Draw stuctures for the following compounds, and upload an image (.png or .jpeg) of the structures.…
A:
Q: A CH3- CH-CH=CH2 + H20 Br B + H20 1 LIAIH4 || CH3- CH2-C- OH 2 H*
A: The main organic product are formed by following reaction is shown in below reaction.
Q: In order to form a tertiary alcohol, excess CHsMgBr could react with which one(s) of these…
A: Note: As per our guidelines, we are supposed to answer only one question when multiple questions are…
Q: Design syntheses to produce the following compounds starting with any alcohol with 3 or less carbons…
A: We have to design the synthesis for the following given compounds starting with any alcohol with 3…
Q: To practice working through the early parts of a multistep synthesis, devise syntheses of(a)…
A: a.
Q: Complete the example of Fischer esterification. Assume that the alcohol is present in excess.
A: The acetic acid is treated with 3-methyl-1-butanol in the presence of an acid to form isopentyl…
Q: Structures (II) and (II) are secondary alcohols Structure (1) is a carbocation Structure (IV) is an…
A: Primary alcohol - OH group is attached to that carbon which is further directly attached to one…
Q: 3. Alcohols Functional group: -OH Eg: CH₁ CH₂ -CH, -CH₂-OH Fill in the missing info. 1 2 3…
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: Q.10. To 0.5 mL of an unknown alcohol, 5 mL of distilled water and 1 mL of 6M NaOH was mixed in a…
A: Inorganic chemistry the Iodoform test is used to test the presence of alcohol and carbonyl…
Q: Question 15 Points out of 1.00 P Remove flag Which of the following molecules will have two…
A: Concept: If a molecule has it's two mirror image form then the molecules are called enantiomer. In…
Q: Which reaction in the Figure below will give the alcohol [1] as the main product? BH, A 2 H,0, NaOH…
A:
Q: ORG-CHEM-LEC.-MODULE-6 - Compatibility Mode Word O Search John esign Layout References Mailings…
A: Here we have to determine above alcohols are primary or secondary or tertiary-
Q: Draw structural formulas for the alkene that gives each alcohol upon hydroboration-oxidation.
A: Hydroboration-oxidation reaction is the chemical reaction in which the formation of alcohol molecule…
Q: what are the IUPAC names of the alcohols that could be used to produce this compund?
A:
Q: 2. Arrange the following compounds in order of decreasing boiling point: (a) 3- hexanol; (b)…
A: Boiling point is directly proportional to H-bonding. Less hindered alcohol will more boiling point…
Q: In order to form a tertiary alcohol excess CH3MgBr could react with: HO. A A, C, and D A, B, and D A…
A: Ketones react with Grignard reagent forms tertiary alcohol. In the case of acid and cyclic ester…
Q: Draw structural formulas for the alkene that gives each alcohol upon hydroboration-oxidation.
A: Hydroboration-oxidation: Hydroboration-oxidation is a method of producing alcohol from alkenes. The…
Q: 62. Which is a tertiary alcohol? CH3 H3CH,C-0-H он но HO II II IV а. I b. II с. II d. IV е. V
A:
Q: 4. Which of the following has a higher boiling point? Explain your answer: (a) 2-propanol or…
A: Hydrogen bonding can increases boiling point.
Q: produce a primary alcohol? O Drag and drop options on the right- hand side to reorder and match with…
A:
Q: What is the appropriate ester and Grenar ?reagent for preparing alcohols a) 2-Phenyl-2-propanol , b)…
A:
Q: Draw structural formulas for organic products A and B in the window below. Br Mg CH3OH CH3CH2ČCH3…
A: Alkyl bromide reacts with Mg metal to produce Grignard reagent which after reaction with methanol…
Q: Which of these compounds is a properly named 2° alcohol? a) 1,3-Propandiol но он b)…
A:
Q: Question 2 of 2 Submit Alcohols undergo dehydration reactions in the presence of an acid catalyst.…
A: Dehydration of Alcohols to Yield Alkenes: One way to synthesize alkenes is by dehydration…
Q: Question 10: Which alkene is required to complete the reaction in Figure 10? * A O All of these…
A:
Q: 51. From the following list, select the two molecules that are isomers: 1. CH;CH-CH-CH-CH; 3.…
A: We have given that Which of the following is secondary alcohol ? a) 1-pentanol b) 2-pentanol c)…
Q: OH OH II III IV HOʻ HO HO V VI VII
A: Answer:- This question is answered by using the simple concept of primary, secondary and tertiary…
Q: Draw structural formulas for organic products A and B in the window below. Br CH₂CH₂CCH3 CH3 Mg…
A:
Q: Label each functional group as an alcohol, ether, acetal, or hemiacetal. LOCH3 а. OCH3 (select) OH…
A:
Q: Which would be a secondary alcohol? . a) CH3 – CH2 – CH2OH b) CH3 – CH – CH3…
A:
Q: Consider the following compounds: он OH dd g HO NIC ORG GAN WRITE THE ANSWER IN CAPITAL LETTERS.…
A: Classification of the alcohol based on the carbon atom of the alkyl group bonded to the hydroxyl…
Q: 2002 Wadsworth Group, a part of Cengage Learning 3. Consider the alcohol hexyl alcohol (1-hexanol),…
A:
Q: In what proportions should alcohols of 90% and 50% strengths should be mixed to make 70% alcohol?
A: Percentage w/v means that that that much grams of substance is there in 100 mL of the total…
Q: la) A student placed 2 drops of an unknown sample in a test tube and added 2 mL of ethanol to the…
A: The alcohol functional group is furnished as R-OH, where R can be an alkyl or aryl group. When R is…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- #B: Methyl acetate has methoxy, -OCH3 as the remaining of the alcohol part in the ester. Isopropyl acetate has isopropoxy, -OCH(CH3)2 as the remaining of the alcohol part in the ester. -OCH(CH3)2 is more electron donating than the methoxy, -OCH3 group due to the presence of two electron-donating -CH3 group in the former. Hence saponification reaction of Isopropyl acetate is much slower than methyl acetate. Hence the rate of saponification of methyl acetate, CH3CCO2CH3 is 50 times greater than that for isopropyl acetate.4. draw the structure of the organic product for each alcohol reacting with permanganate. Look on the internet for an explanation of why the tertiary alcohol in presence of KMnO4 might react. (hint may involve dehydration.) - methanol - ethanol - propanol - 2-methyl-2-propanolI have this task in organic chemistry (book: Brown's introduction to organic chemisty, global edition). Task 10:42. In (a) I have to tell what the funcion of K2CO3 is in step 1. Is it that CO32- take the hydrogen atom in 1-napthol? Will it then be a SN2 mechanism? In (b) I have to name the amine used in step 2 to form Propanolol. But I can't really find out how to come up with an amine that will make that reaction. Here are two pictures of the task:
- We have covered several oxidants that use a multi-valent atom (Cr, Cl, S, or I) as their active species, going from a higher oxidation state before the oxidation to a lower oxidation state after oxidizing the alcohol. Draw the structure of the following atoms, before and after the oxidation of an alcohol to a ketone or aldehyde. How many bonds to oxygen does each atom have before and after the oxidation? (a) the I in the DMP reagent (b) the carbinol C in the alcohol that is oxidizedTunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):1. An unsaturated hydrocarbon reacts with acidic KMnO4 to give isobutyric acid as the only organic product. The unsaturated hydrocarbon is most probably: a. none of these b. 2,2-dimethyl-3-hexene c. 3,4-dimethyl-3-hexene d. 4-methyl-2-hexene e. 2,5-dimethyl-3-hexene 2. Which of the ff. halides will most probably react via an SN1 mechanism? a. CH2=CHCl b. CH2=CHCH2CH2Cl c. CH3CH=CHCH2Cl d. CH3Cl 3. Which of the following will decolorize potassium permanganate solution at room temperature? a. 2-bromobutane b. 1-propanol c. chlorobenzene d. 1-hexyne
- Assuming a 56.8% yield, how kany ml of alkene is required to produce 22.5 mL of 2,3-dimethyl-2-butanol? MW of alkene: 84.16, d: 0.653 MW of 2,3-dimethy-2-butanol: 102.17, d:0.823For compound D to E, what is the reaction of a primary alcohol to an aldehyde and what can be the potential reagent? (reduction / oxidation / elimination / substitution) (mCPBA / PCC / CrO3 / LiALH4 with hydronium quench)A hydrocarbon of unknown structure has the formula C8H10. On catalytichydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. Onhydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.(a) How rnany degrees of unsaturation are present in the unknown?(b) How many triple bonds are present?(c) How many double bonds are present?(d) How many rings ar e present?(e) Draw a structure that fits the data.
- Which of these compounds is most soluble in water at pH 7.0? (Note that the non-ionized forms are shown, and remember that ionization will impact solubility!) a CH3-CH2-HCO-O-CH3 (an ester) b CH3-CH2-CH2-CH2-CHO c CH3-CH2-CH2-CH2-CH3 d CH3-CH2-CH2-CH2-NH2Solution:- 3. Determine the amount, in of a (2.20 M s olution of dichloromethane needed to completely react with 15.72g cyclohexene to give 1,2-dibromocuclohexanw. Assume 12% excess is needed in order to react completely. a . How much 1,2 -dibromocyclohexane would theoretically be produced ? c. How many ML of the 2.20 M Br2 solution are required?Which of these compounds is most soluble in water at pH 7.0? (Note that the non-ionized forms are shown, and remember that ionization will impact solubility!) a CH3-CH2-CH2-CH2-CH2OH b CH3-CH2-CH2-CH2-COOH c CH3-CH2-CH2-CH2-CH3 d CH3-CH2-CH2-CH2-CHO

