Q: Draw most stable chair conformation for cis-1-tert-butyl-4-methylcyclohexane below. in the image 4 5…
A:
Q: %T 85- 80 70- 60 50 40- 30- 20 10- -0. -1+ 4000 3398.81cm-1 3500 2971.22cm-1 3000 2874.39cm-1…
A:
Q: Which of the following has polar bonds but is nonpolar?
A: Given: formula of compound. Tip : if net dipole moment is zero then molecule is non-polar
Q: Determine the critical cation to anion ratio (Rx/Rz) that separates cubic coordination (8-fold) from…
A: We need to look at the relative sizes of the atoms to see what occurs when one of the relevant ions…
Q: Determine the atomic number of the stable configuration of nuceli with mass number A=167?
A:
Q: What quantity in moles of KBr are there in 30.3 mL of 0.150 M KBr?
A:
Q: Calculate the pH of a formic acid solution that contains 1.43% formic acid by mass. (Assume a…
A: we have to calculate the pH of formic acid solution
Q: 12 A ribbon diagram of a zinc metallo-beta-lactamase protein is shown. Label the secondary…
A:
Q: Determine the pH of a 0.352 M NaOH solution. O A. 13.54 O B. 12 O C. 0.352 O D. 0.45
A:
Q: Balanced chemical equation: nonsupo formado boomein -0-3-3² Ni Cl₂ + Na2CO3 -> 80 HOU+H-0- Balanced…
A: A question based on stoichiometry that is to be accomplished.
Q: Balanced chemical equation: HCI + Naz (03 2 Balanced ionic equation: Balanced net ionic equation:
A: Here is the reaction between HCl which is an acid and Na2CO3 which is a salt. By the reaction a new…
Q: If you have 2.94 × 1027 atoms of silver, how many moles of silver do you have?
A: 2.94 × 1027 atoms of silver
Q: 1st attempt Part 1 See Periodic Table See Hint What mass of potassium nitrate is needed to generate…
A:
Q: The reaction of (S)-2-bromobutane with sodium hydroxide can occur via either an SN1 or SN2…
A: Answer is given below
Q: 6. Write balanced chemical equations as indicated for any observed reactions. Write NR if no…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: draw the products you get with syn- and anti- addition of bromine to z-stilbene. For each of these…
A: When the larger group are on same side of double bond, it is called Z isomer. Let us discuss the…
Q: If you have 0.301 m³ of water at 25.0 °C in an insulated container and add 0.142 m³ of water at 95.0…
A:
Q: A reaction of 34.3 g of Na and 38.9 g of Br₂ yields 47.5 g of NaBr. What is the percent yield? 2…
A: A question based on general chemistry that is to be accomplished.
Q: which one is limiting reagent??
A: This is a two-step reaction. Step-1: 2-bromopropane reacts with Mg to give the Grignard reagent,…
Q: Write the empirical formula for at least four ionic compounds that could be formed from the…
A: A compound is formed by the electrostatic interaction between the cation and the anion. The cation…
Q: Calculate the percent yield if 10.8 g of nitrogen gas is produced from the decomposition of 41.9 g…
A: The percent yield formula is calculated by following formula Percent yield= (experimental yield ÷…
Q: Time (s) 0 1 2 3 4 5 1.7 Find the order of reaction and determine the concentration of the reactant…
A:
Q: 1. Acetic acid is a weak monoprotic acid with Ka = 1.77 x 10¯5. NaOH(s) was gradually added to 1.00…
A: The Henderson-Hasselbalch equation for the acidic buffer solution is: pH=pKa+logA-HA ......(1)
Q: Calculate the grams of solute in each of the following solutions: (a) 27.5 mL of 1.50 M KMnO4 (b)…
A:
Q: Determine the pH during the titration of 14.9 mL of 0.149 M nitric acid by 7.12x10-2 M potassium…
A:
Q: The percent yield for the reaction PCl3 + Cl2 → PCl5 is 67.7 percent. What mass of PCl5 would be…
A:
Q: How many signals would be present in the ¹HNMR? O 4 02
A: 1HNMR is also known as proton NMR. NMR is nuclear magnetic resonance. It is a technique that is use…
Q: A 24.6 mL sample of 0.216 M ammonia, NH3, is titrated with 0.399 M hydrochloric acid. After adding…
A: Here we are required to find the pH of the solution.
Q: How many signals would be present in the ¹HNMR? 05 04 OH
A: NMR spectroscopy is a very important tool for the determination of the structure of the organic…
Q: For the general reaction shown below the average rate of disappearance of the reactant "A" over the…
A:
Q: Compare acids, bases, and neutral substances in terms of their relative H+ and OH- concentrations.…
A: We have to compare acids ,bases , neutral substances in terms of their relative H+ and OH-
Q: 20 mL of 0.1M titrand titrated with 0.1M titrant. Data points at 1 mL increments. 14.0 12.0 10.0 Hd…
A: From the pH vs volume of titrant plot we can figure out whether titrant and titrand are acid or…
Q: 10-³ M NaCHO₂ (sodium formate) added to water
A:
Q: the second half of the questio was not answered and that was the part I was really struggling with
A: Answer-2 When work is done by the system against a constant external pressure, it will be the…
Q: Why is it important that we are very careful in how much acid is added to neutralize the reaction?…
A: If too much acid is added during crystallization after neutralization value then acid will increase…
Q: Calculating Avogadro’s Number Using Electrochemistry Background As discussed in lecture,…
A: In the experimental setup, we have taken an aqueous solution of H2SO4(aq) at the cathode and Copper…
Q: If your lab partner had failed to look up the vapor pressure of water and assumed that it was zero,…
A: Given Experiment: Calculating Avogadro’s Number Using Electrochemistry The Vapor pressure of water…
Q: Calculate pressure using the ideal gas law and the van der Waals equation. A 1.88-mol sample of…
A:
Q: Calculate the number of moles in 7.40 μg of Si.
A: mole = massmolar mass 1 μg = 10-6 g
Q: A 28.4 mL sample of 0.379 M trimethylamine, (CH3)3N, is titrated with 0.318 M hydrobromic acid. At…
A:
Q: Balance the chemical equation for the important industrial process: C2H4 + O2 + HCl → C2H4Cl2 + H2O.…
A: Given - C2H4 + O2 + HCl -> C2H4Cl2 + H2O Weight of C2H4 =710.0kg = 710000 gm (1kg = 1000gm)
Q: How many grams of N₂(g) is reacted if 19.0 L of NH3(g) at 0.80 atm and 23.0°C formed? N₂(g) + 3H₂(g)…
A:
Q: Complete each nuclear reaction. a. 21084Po→206Pb b. Fe → 52Mn + β+ + γ c. 208Tl…
A:
Q: The combustion of octane, C,H₁s. proceeds according to the reaction shown. 2C₂H₁ (1) +250₂(g)…
A:
Q: Select the monomer that would make the following polymer: ty O
A: A polymer is a large molecule that is formed by the repetition of small units called a monomer, and…
Q: Chemistry Question
A: Diels-Alder reaction : It is a [4+2] cycloaddition reaction between a diene and dienophile to form a…
Q: A diprotic acid, H, A. has acid dissociation constants of K₁1 = 4.63 x 10 and K₂ = 5.82 x 10-12.…
A:
Q: what will the structure be? and if the bands can be given like C-H band or N-H band along with their…
A: Carbonyl stretching frequency of amides appear at 1630-1680 cm-1. Here the peak is at 1694.82 cm-1.…
Q: Calculate pressure using Dalton's law of partial pressures. A gas mixture is made up of CH4 (4.29…
A:
Q: 2. A lead pellet having a mass of 26.47 g at 89.98 °C was placed in a calorimeter containing 100.0…
A:
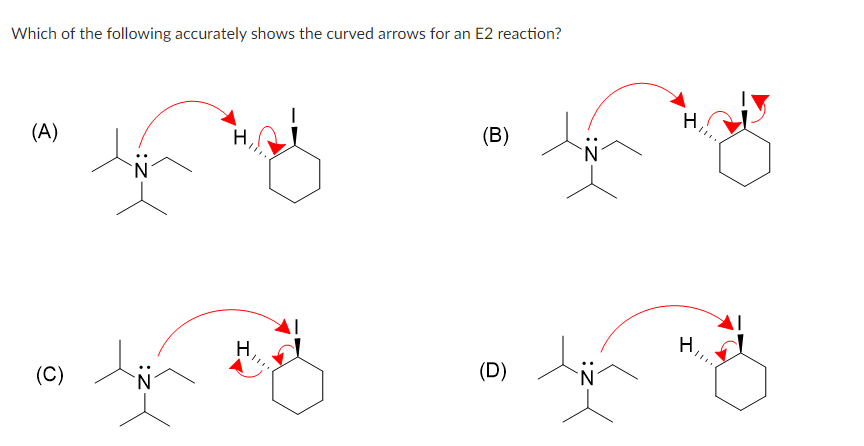
Step by step
Solved in 2 steps with 2 images

- Which of the following compounds (A or B) would experience a faster SN1 reaction?Is this an E1, E2, SN1, or SN2 and is it elimination or substitution.Arrange the following alkyl halides in increasing reactivity in E2 reaction. Write 1 for the least reactive, then 2 and 3 for the most reactive Answer: A. _____ B. _____ C. _____
- Rank A, B, and C in order of increasing SN1 reactivity.Provide the complete mechanism using Curved Arrow Formalism for the reaction of 1-chloro-1-methylcyclopentane treated with NaOMe in MeOH. If more than one product is formed, which is the major, minor, and very minor product? Is the reaction SN1, SN2, E1, or E2. I appreciate the help on this question.Which of the following are achiral? And identify the relationship between each of them. Ex. A-B, B-D
- what mechanism is involved? SN1, E2, E1, or SN2Draw a detailed mechanism for the FeBr3@catalyzed reaction of ethylbenzene with bromine, and show why the sigma complex (and the transition state leading to it) is lower in energy for substitution at the ortho and para positions than it is for substitution at the meta position.How does each of the following changes affect the rate of an E2 reaction? (a, b, and c)
- Which of the products will react the fastest given an SN2 reaction ?Of the two molecules below select which molecule would be predicted to react faster via an E2 elimination. (hint: stereoelectronic arguments can help)Arrange the following according to INCREASING reactivity towards E2: 1st ( least reactive)? 2nd? 3rd? 4th (most reactive)?

