Q: CH3 CH3 1. BH3, THF 2. NaOH, H,O, Н-02 H3C CH2 H3C Alkenes can be hydrated via the addition of…
A:
Q: 4. Predict the product and the curved-arrow mechanism for the following reaction at both 20°C and…
A:
Q: Oxidation of cholesterol converts the alcohol to a ketone. Under acidic or basic…
A: The structure of cholesterol and cholest-4-ene-3-one are given below.
Q: Which of the following is the major organic product in the reaction sequence? CH;CH,M9CI diethyi…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: Which of the following set of alcohols can form a carbocation that is prone to…
A: Answer 4
Q: Complete the following substitution reaction by providing the missing product. Indicate…
A:
Q: 5. Predict the structure of the product formed (with correct stereochemistry) for each of following…
A: Since you have asked multiple questions, we will solve the only first question for you. If you want…
Q: 1. NaOCH,CH, 2. H,O* CH,CH,OH
A:
Q: Q2. When (CH3CH2)3CB is added to CH3OH at room temperature, the major product is (CH3O)C(CH2CH3)3…
A: Alkyl halides undergo nucleophilic substitution through, SN1 and SN2 mechanism. The given alkyl…
Q: cis-3,4-Dimethylcyclobutene undergoes thermal ring opening to form the two products shown. One of…
A: The ring opening reaction of given compound occurs in presence of thermal conditions forms 2…
Q: 04. Give the expected major product or products in each of the following transformations. Clearly…
A: In this question, we want to determine the major product (s) and also clearly mentioned the…
Q: 5) 2-methyl-1,3-cyclohexadiene is treated with 1 equivalent of HCl. There are two major products.…
A: Applying concept of 1,2 and 1,4 addition reaction at conjugated diene.
Q: Which of the following carbocations would NOT be likely to undergo rearrangement? CH3 а. CH,CCH,CH,…
A:
Q: Predict the b-elimination product(s) formed when each bromoalkane is treated with sodium ethoxide in…
A: beta-elimination: The elimination reaction takes place from beta-carbon of haloalkane is known as…
Q: Which of the following alkenes will yield a meso dihalide when reacted with Br/CCh at room…
A: Alkene reacts with bromine to form dibromoalkane. The reaction proceeds by the bromonium ion…
Q: Which of the following set of alcohols can form a carbocation that is prone co…
A: We have to predict the alcohol that can undergo rearrangement of carbocation.
Q: Explain the different products of the following two reactions by considering the mechanism by which…
A: In literature two different type of substitution reaction of alkenes are reported depending on the…
Q: Name the kind of sigmatropic rearrangement that occurs in the following reaction.
A: The given reaction is represented as follows:
Q: C\ Rearrangement the following structures by increasing melting point? CH3 CH3 ОН D\ Rearrangement…
A: With increasing in molecular weight, both melting point and boiling point increases. H- bonding is…
Q: Which of the following is the major product of the rapid reaction of cyclohexene with bromine in…
A:
Q: H30* ?
A: In the presence of acid alcohol gets protonated and then water is eliminated to produce carbocation.…
Q: In the mechanism of the following transformation of the cyclic ether to the corresponding dibromide,…
A: This reaction passes through 6 transition state and 5 intermediate.
Q: -C=C-CH,
A: Given
Q: Fill in the blank: The reaction of alkene with peroxyacid (RCO3H) is: A) a radical mechanism with…
A: The reaction of alkene with peroxyacid (RCO3H) is
Q: COOME CHO CO2ME
A: Electrocyclic reaction is unimolecular... And diels alder reaction is biomolecular... Diels alder…
Q: This molecule will undergo a bimolecular nucleophilic substitution reaction with hydroxide ion. Draw…
A:
Q: 2. Complete the following reactions of alkenes by drawing the structures of the major products.…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Identify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a…
A: Since it is given that the carbocation rearrangement is not taking place. Hence we need to look for…
Q: Which of the following reaction forms an alkene in a one-step concerted process involving anti…
A: The reaction which involves the elimination of two substituents from the organic molecule is called…
Q: In the mechanism of the following transformation of the cyclic ether to the corresponding dibromide,…
A:
Q: The attached reaction does not afford the major product that is given.Explain why this is so, and…
A: In the given reaction a tertiary alkyl halide is undergoing a substitution reaction, where a bulky…
Q: of the following alkenes will undergo a carbocation rearrangement when reacting with H2O in the…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the…
A: Bulky bases prefer elimination whereas non-bulky bases prefer substitution reaction.
Q: NBS (CH3)2CHCH2CH=CHCH3 ? Cl4
A: NBS is N-bromosuccinimide is an alternative to bromination. It is used for allylic bromination i.e.…
Q: Two products are formed in the following [1,7] sigmatropic rearrangement, one due to hydrogen…
A: Sigmatropic reactions are those reactions where formation of new bond takes place when another bond…
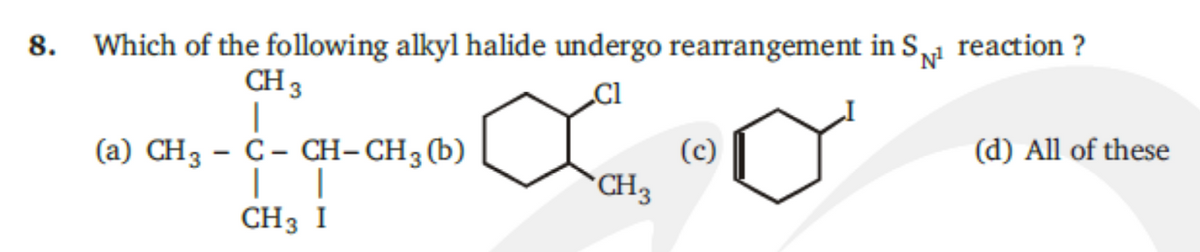
Q: Which of the following compounds has the greatest tendency to undergo rearrangement during SN1…
A: SN1 reaction involes the two step reaction first is formation of stable carbocation and second is…
Q: a) CH3 b) CH3 – CH – CH- CH3 CH3 с) СНЗ — СН2 —- CH - СHЗ
A:
Q: Q3. 2-Bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C.…
A: Dehydrohalogenation involves the removal of the halogen atom together with a hydrogen atom from a…
Q: Each of the following transformations is believed to proceed by a sequence of reactions concerted.…
A: A pericyclic reaction takes place via a concerted mechanism in which the old bond breaking and a new…
Q: 7. Which of the following alkenes is most stable? А. В. С. D.
A: The stability of alkenes depends on the substituents attached to it. More the number of substituents…
Q: Addition of HBr to which of the following alkenes will lead to a rearrangement?
A: Addition of HBr to alkene take place according to the Markovnikov's Rule ( Addition of H+ take place…
Q: a) What product is formed from the [1,7] sigmatropic rearrangement of a deuterium in the following…
A: (a) The reactant in the equation undergoes [1, 7] sigmatropic rearrangement of a deuterium to…
Q: The major product formed by addition of HBr to (CH3)2C=CH– CH=C(CH3)2 is the same at low and high…
A: Chemical reaction in which an atom or a group of atoms are added to each carbon atom of a…
Q: How many of the following will undergo a favourable rearrangement to a lower energy carbocation? H3C…
A: We know the stability of carbonation is tertiary> secondary> primary.Again if a carbonation…
Q: What are the possible starting materials of the given alkene below, if the starting material is…
A: In this question, the stability of intermediate towards the formation of a specific product in…
Q: Which of the following alkenes will yield a meso dihalide when reacted with Br2/CCI4 at room…
A: Meso compounds are those compound which have more than one stereocenters. They are superimposable.…
Q: provided a detailed sythesis of this mechanism starting with reactants leading up to this product? F…
A: Methyl substitution of BACE-1 Inhibitor has a profound effect on it's potency
Q: С. CH2CH2CH3 CH3OH H3CH2C,] H3C" 25 °С d. CH3O (CH3)3Br 0 °C е. CH3O- -Br 75 °C
A: Following are the product with appropriate stereochemistry.
Q: е. CH3O- -Br 75 °C f. CH3 CH3O 'CI 55°С CH3
A: In this question, we will draw major product or Products with stereochemistry. You can see details…
Step by step
Solved in 2 steps with 2 images

- M 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :What is the expected major product of reacting cyclohexane carbaldehyde with (CH3)2NH?For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products. chlorocyclohexane + NaOC(CH3)3 in (CH3)3COH
- Complete the flowchart by drawing the resulting structures of each reaction. A. B. C. D. E. F. G. H. I.Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d belowThe major product formed by addition of HBr to (CH3)2C=CH– CH=C(CH3)2 is the same at low and high temperature. Draw the structure of the major product, and explain why the kinetic and thermodynamic products are the same in this reaction.
- 2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3Q2. What are the products of the following reactions? Q3. 2-Bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C. Identify A, B and C. Which is predominant? When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed. What is the detailed reactin scheme for the transformation? Describe the purification procedure.
- Reaction of 2° alcohol A with HCl forms three alkyl chlorides, all of which result from rearrangement of the 2° carbocation initially formed. Draw the structures of these products and a mechanism that illustrates how each is formed.N(CH2CH3)3 + HNO3 --------> a.) rewrite the reaction using bond-line structure of reagents and products of the reaction b.) supply the curved arrows explaining the mechanism of the reactionWhat order of reagents should be used to synthesize the following product from methylcyclopentane?(A) Br2(B) Br2, hv(C) KOH, heat

