Which of the following alkyl halides is essentially unreactive in an S2 reaction? 2-Bromo-2-methylhexane 1-Bromopentane 1-Bromopentane 1-Bromo-4-methylpentane
Q: Consider an E2 reaction between (2-bromoethyl)cyclohexane and base potassium tert-butoxide. Describe…
A:
Q: Which of the following compounds would have a high percentage of para substitution product when…
A: Electron donating groups are ortho para directing. Electron withdrawing groups are meta directing.…
Q: Which of the following alkyl chlorides will undergo SN2 reaction most readily? O…
A: Here, we have to find the alkyl chloride that will undergo SN2 reaction most readily among the given…
Q: What are the products of the following reactions if they proceed via the SN1 mechanism? CH2CH3 %3D…
A: a. SN^1 reaction is passing through the formation of carbocation. As carbocation is planar,…
Q: Which of the following compounds is MOST reactive in an SN1 reaction? I -OTs II -OTs II -OTs IV -OTs…
A: Carbocation will be more stable if it takes part in resonance with a double bond.
Q: Which of the these compounds will undergo an SN2 reaction with NaSH? O CH3CH2CH=CHB O…
A: Nucleophiles are those elements or compounds which are electron rich. These have lone pair of…
Q: Choose the member of each pair that will react faster by the SN1 mechanism.(a)…
A: SN1 reactions are most speedy with tertiary alkyl halides, they happen more gradually with secondary…
Q: Which of the following compounds will give a positive visible result with the Belstein test? I.…
A: The Beilstein test is a flame test for detecting halogens in organic compounds. Polyvinyl chloride…
Q: Which of the following is the product of the reaction of (R)-1-deutero-1-iodopropane with methoxide?…
A:
Q: Which of the following alkyl halides is essentially unreactive in an S2 reaction? A…
A:
Q: Which of these reactions are likely to produce both elimination and substitutionproducts?(a)…
A: In order to predict elimination and substitution reaction products we will use saytzeff rules. Since…
Q: 1. Predict the number of reactive sites that will occur if this compound is reacted with KMNO4 with…
A:
Q: Which of the following is NOT a product of the reaction (Claisen condensation) below? OCH 3 + OCH3…
A:
Q: Which five-membered ring compound results in the two products shown below upon ozonolysis? 1. O3 ?…
A: Ozonolysis of alkene produce carbonyl compound.
Q: What is the product of the reaction of bromoethane with each of the following nucleophiles? a.…
A: Since your question has multiple sub-parts, we will solve first three sub-parts for you. If you want…
Q: What halides would undergo E2 dehydrohalogenation to give the following pure alkenes?(a) hex-1-ene…
A: Since you have posted multiple sub-parts, we will solve the first three sub-parts for you. To get…
Q: What nucleophile could be used to produce each of the following products from 1- bromobutane?…
A: The question is based on the concept of nucleophilic substitution reactions. we have to identify the…
Q: Which of the following is more reactive in E1 reactions? 2-bromopropane 1-bromopropane
A: E1 elimination is more reactive in 2-bromopropane because it forms two 2° carbocation which is more…
Q: Write down the elimination products (E2) of the following reactions. Refer to stereochemistry. a.…
A:
Q: CH3CH2CH=CH-CH-C6H5 CH2 ||…
A: E1 elimination occurs via carbocation intermediate formation. To get stable carbocation,…
Q: Which molecule reacts fastest with ethanol by SN1 mechanism? 1-bromopentane 3-bromopentane…
A: Given molecules: 1-bromopentane 3-bromopentane 2-bromo-2-methylbutane 1-bromo-2,2-dimethylpropane To…
Q: Rank the compounds from most reactive to least reactive under conditions of both SN1 and SN2.…
A: Here we have to rank the compounds from most reactive to least reactive under conditions of both SN1…
Q: Which of the following is NOT a good nucleophile for SN1 reactions? 1) NaOCH3 2) CH3OH 3)…
A: SN1 reactions or unimolecular nucleophilic substitution follows in two steps where in the first step…
Q: NBS hv b. CH3 CH3 carbene
A: N-Bromosuccinamide (NBS) is a reagent used specifically for brominating allylic or benzylic…
Q: The following reaction proceeds by two consecutive E2 eliminations. KOH, 170 °C CICH:- ethylene…
A: First, the given compound goes elimination to give an alkene which further undergoes elimination to…
Q: Which molecule reacts fastest with ethanol by SNl mechanism? 1-bromopentane b. 3-bromopentane…
A: Answer : option c. 2-bromo-2-methybutane react fastest with ethanol by SN1 mechanism.
Q: Markovnikov's Rule is needed to predict that the major substrate product in the reaction between HBr…
A: 1 pentene , 1 bromopentene .
Q: Rank the following alkyl bromides from most reactive to least reactive in an SN2 reaction:…
A: Since for SN2 reaction, the order of rate is Primary halide > secondary halide > tertiary…
Q: Arrange the following alkyl halides in increasing reactivity in E1 reaction. Write 1 for the least…
A:
Q: 6. Which of the following is most likely to undergo rearrangement during reaction with methanol?…
A:
Q: Which of the following molecules would you expect to undergo an S,I reaction the fastest? Look…
A: To answer this question , first we would study about SN1 reaction . Then we would check structure…
Q: In which of the reactions below does the carbocation act as an acid? ÇH3 i. -H CH3-C CH3 II CH3 CH3…
A:
Q: For each of the following alkyl halides, indicate the stereoisomer that would be obtained in…
A: Hey since you have posted sub-part question, we will answer first three sub-parts only. If you want…
Q: Which of these alkyl chlorides undergoes dehydrohalogenation in the presence of a strong base to…
A:
Q: Which of the following haloalkanes will undergo SN1 reaction? 2-chloro-2-methylpropane II…
A: SN1: The SN1 reaction is a substitution reaction in organic chemistry, the name of which refers to…
Q: If 2-fluoropentane could undergo an E1 reaction, would you expect the major product to be the more…
A: Please find below the reaction taking place
Q: Each of the following molecules has two leaving groups (LG) at different positions. If 0.1M each…
A: Nucleophilic substitution reaction goes via two ways One SN1 and other is SN2 Here we have to see…
Q: Which of the following alkyl halides will not react to either Nal in acetone and 2% ethanolic AGNO3?…
A:
Q: Which of the following transformations is an example of a reduction? A CI MgCl Br R1 B R-C=C-R2 R2…
A: Option 3. None of the shown reactions is a reduction reaction.
Q: Which of the following species are likely to be nucleophiles and which electrophiles? OH CH3O CH3CI…
A:
Q: Which of the following alkyl halides undergoes the fastest SN2 reaction? 1-iodohexane 1-fluorohexane…
A: This question belong to nucleophilic substitution reaction. Nucleophilic substitution reaction…
Q: Which of the following molecules can be a starting material in producing 4- Methyl-1-pentyne? Br Br…
A: We have to find out the molecule which can be a starting material in producing 4- methyl 1- pentyne?…
Q: Which of the following alkyl halides gives only one alkene as the product in the E2 reaction? Br 1 2…
A: A multiple choice question based on elimination reaction, which is to be accomplished.
Q: RXN 1 RXN 4 Br H20 H* RXN 2 2H2 RXN 5 BH3 / THF H2O2, NAOH, H2O HO Pd/PtO2 RXN 3 RXN 6 2 HCI HO…
A:
Q: 3. Arrange the following alky halides in order of decreasing reactivity in an S,1 reaction: a)…
A:
Q: Which of the following SN2 nucleophiles will not directly add one or more carbons to the carbon…
A:
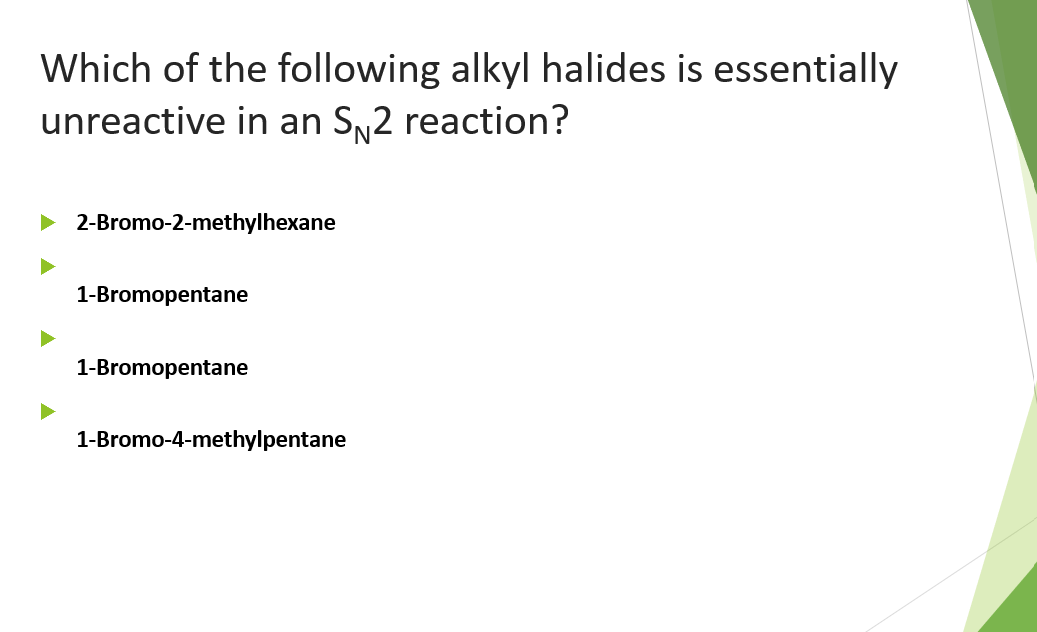
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

- For SN1 Explain the order in which 1o (primary) alkyl halides reacted (fastest to slowest) and explain why. The 1o primary alkyl halides are: (see picture below) 1-chlorobutane 1-bromobutane 1-chloro-2butene benzylchloride7. Circle those of which that are o,p-directors when attached to a benzene ringWhich product is the final product in the reaction below, shown as numbered?
- [IV2] Instructions: Follow the flow of electrons indicated by the curved arrows in each of the following reactions, and predict the products that result: (refer to the photo below)Draw the mechanism ffrom benzaldehyde to this using: i)NaBH4 ii)TsCl, py iii)NaCN iiii)H+, H2ORank the following from most reactive to least reactive in an SN1 reaction.
- For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products. chlorocyclohexane + NaOC(CH3)3 in (CH3)3COHExplain how and why rearrangements occurduring Friedel-Crafts alkylation reactions formingmore than 1 product. Also illustrate therearrangement reaction from the aboveexample.Which of the following pairs of compounds reacts with OH- ions faster in an SN2 reaction? Briefly explain. CH3Br or CH3I? 2-chloro-2-methylpropane or chloromethane?
- Alkyl diazonium salts (shown below) are considered "super" leaving groups; a consequence of this is that they tend to be contact explosives What quailities make alkyl diazonium salts such excellent leaving groups?For SN2 Explain the order in which 1o (primary) alkyl halides reacted and explain why. The 1o primary alkyl halides are: (see picture below) 1-chlorobutane 1-bromobutane 1-chloro-2butene benzylchloride1 Produce the given molecules from-any simple alkyl halide consisting of 4 or less carbons or (use no other functional groups other than these, ethylene oxide, cyclohexane, or cyclopentane. Any other carbon- containing reactants must be synthesized from the reagents gave above... Please explain too!!

