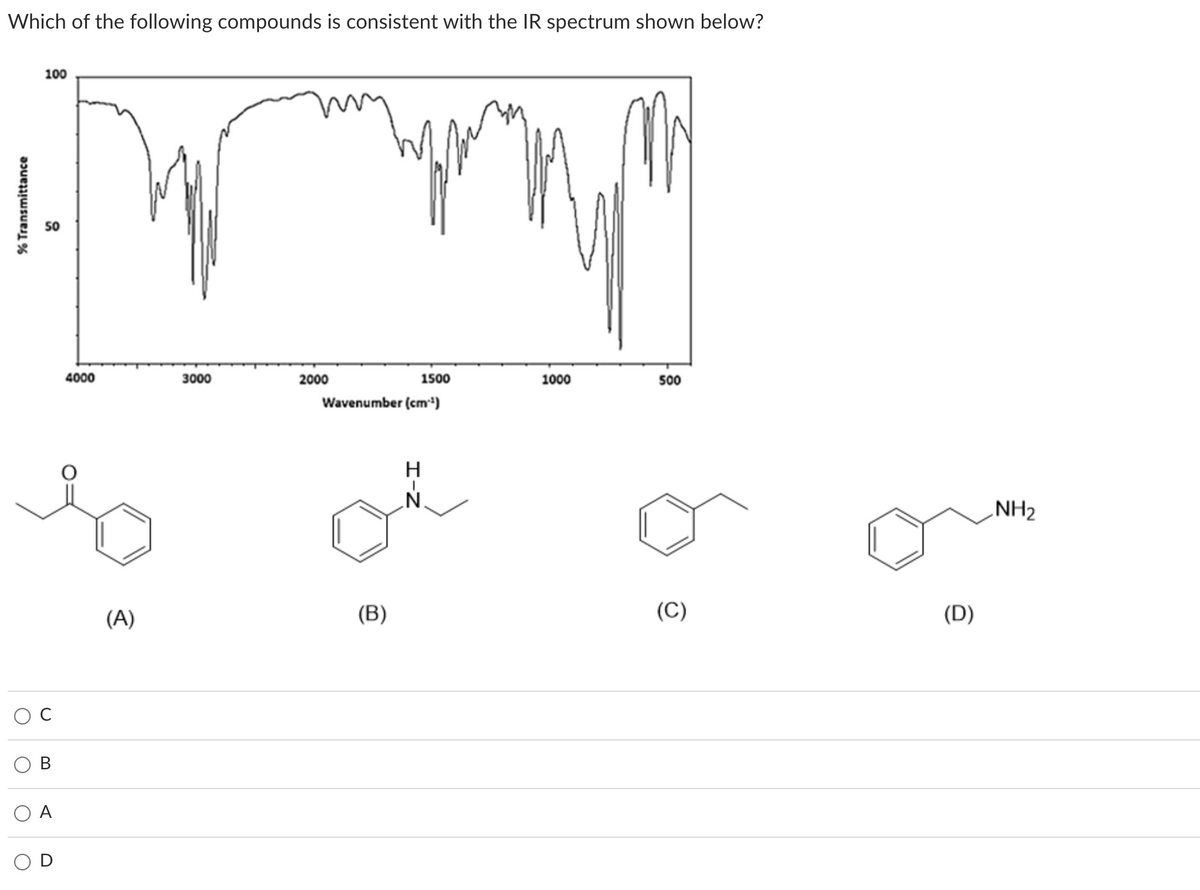
Which of the following compounds is consistent with the IR spectrum shown below? 100 2000 1500 1000 500 Wavenumber (cm) H (C) % Transmittance 50 B O A O D 4000 (A) 3000 (B) (D) NH₂
Q: 2. Write the IUPAC name and three common names for each of the following chalcones. A. Br CI B. CI
A:
Q: Complete the changes in concentrations for each of the following reactions: d. Mg3(PO4)2 (s) → 3Mg2+…
A:
Q: [References] The specific rotation, [a]p, for sucrose is +67. What is the observed rotation for a…
A:
Q: IR Spectrum (KB/ disc) 4000 ent % of base peak 3000 44 100 80 60 40 20 40 13C NMR Spectrum (100.0…
A:
Q: The following concentrations are found in mixtures of ions in equilibrium with slightly soluble…
A: If AmBn is slightly soluble solid then Ksp can be written as - AmBn. -------> mAn+ + nBm- Ksp =…
Q: What are the products for the following ozonolysis reaction? 1.0₂ 2. DMS III A. I and II B. II and…
A:
Q: In an experiment, 0.500 mol of Fe(NO3)3 is dissolved in water to produce an 2.00 L solution. The…
A: Molarity is the number of moles of solute present in per litre of solution. It is denoted by letter…
Q: What are the objectives of computational differences chemistry? Explain the between molecular…
A:
Q: 0/50 mixture of cyclohexanamine and cyclohexane and asked NH₂ Cyclohexanamine Cyclohexane onent of…
A: Given: 50/50 mixture of cyclohexanamine and cyclohexane
Q: Statement Analysis: Statement 1: AG" is the difference in free energies of the products and…
A:
Q: MeS MeCN dentify the correct product(s) from the reaction above SMe Et O Et Et J... L.C SMe SMe
A: It is a SN2 type reaction as nucleophile is very strong.
Q: Which keto product would be isolated from the reaction shown? 1. BH3/THF = 2. HO, H2O2. 20 OH OH C A…
A:
Q: The chemical reaction of 2-butene and HCI yields what product? O CH3CH₂CH₂CH3 CI CH3 CH2 CHCH 3 CI…
A: The chemical reaction of 2-butene and HCl yields the product
Q: Will a precipitate be observed if 0.10 mol Ag* and 0.001 mol SO4²- are added to make 1.0 L of…
A: Here we are required to find whether precipitation will occur or not
Q: Solve the following stoichiometry problem using this balanced equation. 3NO2+H2O->2HNO3+NO How many…
A:
Q: CI How many enantiomers does the following molecule have? H III. ...
A:
Q: Please calculate and mke the numbers ledgibe... I GOTTHIIS ANWER YOU PROVIDED WRONG AND 2 SIG…
A: M
Q: Rank the following ¹H NMR proton signals from lowest ppm chemical shift to highest: I. II. III. HHH…
A: To answer the given MCQs.
Q: Calcium hydroxide and lead chloride have very limited solubility in neutral water (?? ≈ 7.0).…
A: Le Chatelier's principle states that " When a settled system is disturbed, it will adjust to…
Q: Using the balanced equation below to determine the molar ratio between the chemical pair: O2 and…
A:
Q: In the reaction aA + bB → CC + dD where a, b, c, and d are stoichiometric coefficients, which of the…
A: -> A redox reaction is a reaction in which one specie get oxidized and other specie get reduced.…
Q: Pls help on both questions.
A: Here, we have to find the shape of BrF5. Also, we have to find the correct option when 6.41 of H2S…
Q: 2NH3 all in the gas state. If the equilibrium constant is 2.73x10-8,what concentration of ammonia…
A:
Q: When the equation Fe2(C204)3 → FeC204 + CO2 is balanced with the smallest integer coefficients, the…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: Which of the following conversioon proesses DOES NOT depend on the molar mass of a substance?
A: The compound is SF6 Molar mass of the compound is 146.07 g/mol Mass of the compound 235.7 g Number…
Q: B. Where does the amino acid have a net charge of -1? above pH 9.69 at pH=9.69 at pH=6.01 at pH=2.34…
A: The given titration curve has 2 different points where sharp rise in pH takes place. Hence the…
Q: IZ N NH₂ CO CI سلمك SH OH pyridine OH (1 eq)
A:
Q: Use VSEPR to predict the geometry of the following ion: SO32 Answer: trigonal planar linear…
A:
Q: In an experiment, 0.500 mol of Fe(NO3)3 is dissolved in water to produce an 2.00 L solution. The…
A:
Q: What is the name for a 50:50 mixture of enantiomers such as a 50:50 mixture of (S)- -utanol and…
A: a 50:50 mixture of enantiomers such as 50:50 mixture of (S)-2-butanol and (R)-2-butanol are called…
Q: to form carbon dioxide and water vapor. Write a balanced equation for this reaction. _CgH₁8 (1) + O₂…
A:
Q: 8. Assume a concerted process is taking place and draw the mechanism of the following reaction. OTS…
A: A question based on stereochemistry of organic reactions that is to be accomplished.
Q: A 1.52 M solution of (R)-2-butanol is mixed with an equal volume of a 0.760 M solution of racemic…
A:
Q: Which of the following statements are consistent with compound X below? лятно NH₂ X I. X has an odd…
A:
Q: four common types of radioactive decay, differences between them and a detailed comparison,…
A:
Q: 3. Calculate the density of sulphur dioxide at STP. 4. A 1L steel tank is fitted with a safety valve…
A:
Q: 11. A suspension of milk of magnesia, Mg(OH)2, is analysed by back titration. A 1.0391 g sample is…
A: Solutions Calculate the weight percentage of magnesium hydroxide in the sample..
Q: Ammonia, NH3 (g), is produced when nitrogen gas reacts with hydrogen gas at 304 kPa and 25.0°C.…
A: Here we are required to find the percentage yield of ammonia formation
Q: The following reactions happen simultaneously when HCl is added to a mixture of Mg(OH)2 and MgCO3:…
A: Volume of 37 wt% HCl required can be calculated from the balanced reaction of HCl to MgCO3 and…
Q: Br product 4 + 2 18 B .Br product 3 Br 7 3 product 2 + 5 00 product 1
A:
Q: [References] Draw the organic product(s) of the following reaction. OH -CN excess NaOH • You do not…
A: In this question, we will draw the product for this reaction. Here NaOH act as base and it will…
Q: A balanced chemical reaction that includes the energy change during the reaction is called the…
A: chemical reactions are of two types exothermic and endothermic reaction.
Q: The aluminum sample is not cut into small pieces but rather left as one large piece. a. How will…
A: Introduction : Percent yield of the reaction is the percent ratio of actual yield to the…
Q: Consider the following reaction schemes involving the reaction of an imine with an organolithium…
A: We have find out the reaction of an imine with an organolithium compound.
Q: Which of the following represents the equilibrium in a saturated solution of Cr2(SO4)3?? Cr₂(SO4)3…
A:
Q: Which reaction is faster in each pair? For each blank, type A or B. blank 1 blank 1 H N3 B A NaN3…
A:
Q: 7. Which of the following is more stable? 8. Name the compound according to IUPAC nomenclature. (Use…
A: We know between cis and trans isomer, trans isomer is more stable compare to cis, as in trans isomer…
Q: Translate the Fischer projection to a wedge-and-hashed bond structure in a zig-zag chain. Be sure…
A: Stereochemistry is branch of chemistry in which we deal with the arrangement of atoms in three…
Q: Chemistry 2. The pks of tye amino acid Arg are 1.82, 8.99, and 12.48. If Arg was used as a buffer,…
A: We have find out the ratio of conjugate base to weak acid in a pH of 7.00 Arg buffer.
Q: Draw the structual condensed formula of 4-ethyl-1-heptyn.
A: Draw the structural condensed formula --
give answer asap
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- The IR frequency of C≡N¯ in fac-[IrCl3(C≡N)3] is 2200 cm-1. Estimate the IR frequency of C≡N¯ for fac-[IrF3(C≡N)3] with an explanation.What are the major peaks in the IR spectra? Disregard fingerprint region <1500cm^-1You are a forensic chemist working in a crime lab. You have received a white powder from investigators that is suspected to be a drug of abuse. Based on the FTIR spectrum, make a decision and justify it based on peak assignments above 1500 cm-1 . Which of the three is it?
- Using a 60 MHz spectrometer, a chemist observes the following peak: doublet, J = 7 Hz, at 2.50 ppm What would be the chemical shift of this same peak as measured by a 300 MHz spectrometer? (Include units.)A calibration curve for a newly developed dye was prepared using a cuvette of pathlength 1.0 cm and illumination at 421 nm. What is the concentration (μM) of dye in a sample if an absorbance of 2.14 was recorded upon placement into a spectrometer?In determining the concentration of a sample X, it was found that it has maximum absorbance at 275 nm. Compound X, has molar absorptivity at 275 nm of 8400 L / mol.cm. Using a spectrophotometer with a 1 cm cuvette, an absorbance of 0.70 was found. What is the concentration of compound X in the sample?
- Calculate the HDI of each FM and propose 2structures for two molecules that are isomers.Specify what the indicated absorption (s) shouldbe to.a.C7H15NO with strong absorptions in 3400, 3270and 1670, 1390, and 1370 cm-1b. C6H10O with absorptions in 2065 (m), 1685 (f),1650 (m) and 970 (f) cm-1c. C9H12O, with absorptions in 1600 and 1500 and1245 cm-1.Convert the accompanying transmittance data to absorbances: (a) 25.5% (b) 32.8% (c) 0.085The absorbance, A, of a stock CuSO4 solution was 0.214. What is the %Transmittance of the solution?
- Draw block diagram of IR spectrophotometer and describe the basic components.A solution containing two different fluorescent compounds, Ben and Jerry, were analyzed for their individual concentrations in the mixture. Standards of pure Ben and pure Jerry were prepared at a concentration of 500.0 mM and were run in a UV-Vis Spectrophotometer to determine their absorption properties. Absorbance Wavelength Compound Ben 500 mM Compound Jerry 500 mM 400 nm 0.137 0.136 450 nm 0.312 0.113 500 nm 0.154 0.078 550 nm 0.076 0.079 600 nm 0.227 0.148 650 nm 0.230 0.230 700 nm 0.151 0.357 750 nm 0.157 0.246 800 nm 0.154 0.154 A standard curve of the standards was also prepared to help determine the concentration of each component in the solution. The solution produced an absorbance reading of 0.486 at the λmax of Ben, and 0.463 at the λmax of Jerry. STD CURVE BEN Λmax Ben Λmax Jerry STD CURVE JERRY Λmax Ben Λmax Jerry CONC (mM) ABS ABS CONC (mM)…A solution containing two different fluorescent compounds, Ben and Jerry, were analyzed for their individual concentrations in the mixture. Standards of pure Ben and pure Jerry were prepared at a concentration of 500.0 mM and were run in a UV-Vis Spectrophotometer to determine their absorption properties. Absorbance Wavelength Compound Ben 500 mM Compound Jerry 500 mM 400 nm 0.137 0.136 450 nm 0.312 0.113 500 nm 0.154 0.078 550 nm 0.076 0.079 600 nm 0.227 0.148 650 nm 0.230 0.230 700 nm 0.151 0.357 750 nm 0.157 0.246 800 nm 0.154 0.154 A standard curve of the standards was also prepared to help determine the concentration of each component in the solution. The solution produced an absorbance reading of 0.486 at the λmax of Ben, and 0.463 at the λmax of Jerry. STD CURVE BEN Λmax Ben Λmax Jerry STD CURVE JERRY Λmax Ben Λmax Jerry CONC (mM) ABS ABS CONC (mM)…

