Q: Is 2-pyridone aromatic?
A:
Q: Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the…
A: Given; Reaction of naphthalene with CH3CH2COCl and AlCl3
Q: Which of the following reactions is a substitution reaction? C6H6 + Cl2 → C6H5Cl + HCl CH2=CH2 + Cl2…
A:
Q: When a substituted benzene undergoes an electrophilic aromatic substitution reaction, where doesthe…
A: When substituted benzene undergoes an electrophilic aromatic substitution reaction, where does the…
Q: SN2 reaction is an example of a nucleophilic substitution reaction. Imagine that 1-iodobutane reacts…
A: Answer:
Q: Which of these is an aromatic compound? :S: :: S: H. H :0: :0:
A: The compound which contains (4n+2)π electrons, and obeys Huckels rule. According to this rule the…
Q: Consider the following aromatic .compounds ONHS N(CH N(CH32 OCH NON NO2 NO2 OCH ARO MAT TIC HYD ROC…
A: Electrophilic aromatic substitution (EAS) is the type of organic reaction in which electrophile…
Q: Which (if any) lone pairs are participating in aromaticity?
A: Since the lone pair which is present in the single bonded atom is in conjugation with the ring,…
Q: Which is least reactive in electrophilic aromatic substitution? (A) (B) CH3 (C) CHO (D) C3
A:
Q: AIC +. CH3CH2CI 24111S O A) electrophilic substitution B) electrophilic addition C) nucleophilic…
A: Applying concept of fredalcraft acylation reaction.
Q: I3D :O と
A: For the compount to be aromatic, it should be cyclic, planar and obeys (4n+2)pi electrons.
Q: 1. The multifunctional molecule below can undergo both nucleophilc addition reactions and…
A: In the given molecule, the free aldehyde group and the free ketone group will undergo Nucleophilic…
Q: Once reacted with NaNH2 in NH3, which forms the smallest number of methylated anilines? А. С. В. D.…
A:
Q: MCQ 26: Introducing a halogen atom into an alkene can greatly reduce its A. polarity B. volatility…
A: Alkenes are the hydrocarbons with atleast one double bond between two carbons. When a halogen is…
Q: All aromatic compounds have __________ as a building block. A. benzene B. aniline C.…
A: Please find your solution below : There are certain criteria that an aromatic compound must follow.…
Q: Chemistry c) 4 d) 5 2) What is the correct order for the following molecules according to their…
A: Electrophilic aromatic substitution means one electrophile is substituted by other electrophile.
Q: Which of the following compounds are NOT aromatic? N. A A only OC, D, and E A and C C and D O B, C,…
A:
Q: Which of the following compounds are aromatic? :N A N B D N-N E F О В, С, F O B, F О В, С, F, G O B,…
A: According to huckel rule , when (4n+2) pi electrons( 2,6,10... etc.) are in complete cyclic…
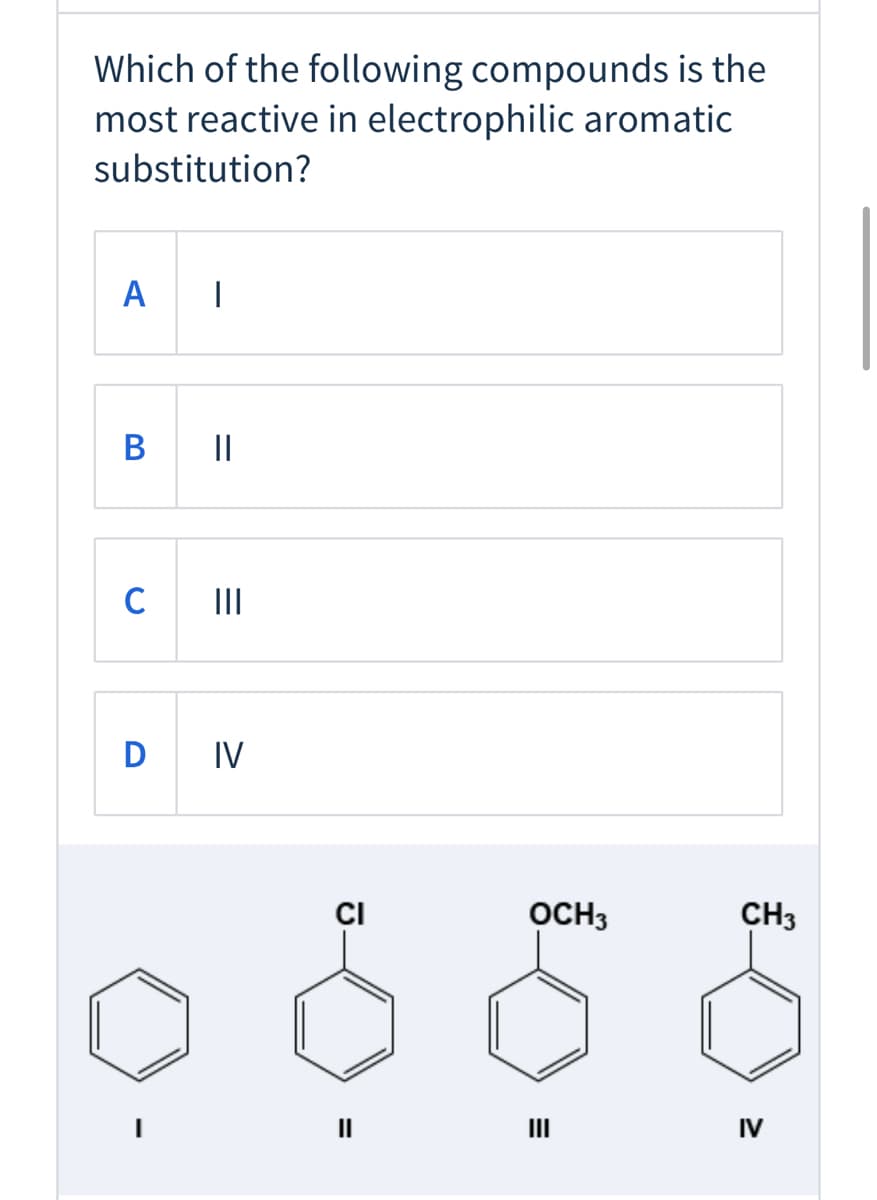
Q: Rank group of compounds from most reactive to least reactive toward electrophilic aromatic…
A: When the benzene ring has electron-donating groups, then the benzene ring has more reactive to…
Q: Of Hückel's original rules for aromaticity, one needed to be revised. Which one is it? The ring…
A: The important criteria for the aromaticity, such as ring must be planar, fully conjugated or pi…
Q: CH3 CH,Br +Br2 +HBr (a) Nucleophilic addition (c) Electrophilic addition (e) Free radical…
A:
Q: In which of the structures A through D shown below is the chlorine substituent replaced at the…
A: The replacement of chlorine in the given reaction would proceed through addition elimination (…
Q: Which of the following is aromatic? A) B) HN- C) D) ZI
A:
Q: Trensproduc is formed 5. Draw the structures of A and B, and use the curved arrow formalism to show…
A: Given,
Q: Which structure is NOT aromatic? d. Do .. H3C :Br:
A: In order to decide a compound to be aromatic the following points are considered. The compound…
Q: A bicyclic heterocycle aromatic ring with four N atoms. a Pyridine b Pyrazine c…
A:
Q: NaOtBu HOLBU heat Brg, H20 compound A compound B Br
A:
Q: Which one of the following is an aromatic compound? a) b) A d) i. ii. ii. d iv. a IZ
A:
Q: II
A: Aromatic compounds : compounds follows huckels rule are aromatic according to huckels rule a…
Q: The most reactive among the other hydrocarbons. O Alkynes O Alkenes Alkanes O Arenes These compounds…
A: Hydrocarbons are the compounds containing only carbon and hydrogens. If the reactivity of…
Q: electrophilic aromatic substitution
A: Ans. Option B is correct: Electrophilic aromatic substitution/replacement responses occur naturally…
Q: Which compounds are aromatic? (A) (B) (C) (D) (E) (F) B. (H) (1) (G) H-
A: The following conditions must satisfied in order to becomes aromatic. Must be planar Must be…
Q: Which (if any) lone pairs are participating in aromaticity?
A: Since the lone pairs which are present on the double bonded atoms do not participate in resonance…
Q: NANH2 1) HgSO, H,SO, 2) H,0 Br 1)NANH2(xs) 2)H,0 Br 2 1)Hg(OAc)2, HO 2)NOBH tautomerization Br…
A: Mercury (2) acetate is used as oxidising agent.
Q: Explain why electrophilic aromatic substitution in Pyrrole takes place at C-2 positions whereas, in…
A: Pyrrole is more reactive towards electrophilic reagents in comparison to pyridine because of its…
Q: H-N: || II IV V
A: Aromatic compounds are a class of organic compounds which has continuous delocalization of π…
Q: Meta directors in electrophilic aromatic substitution reactions have A) unpaired electrons in the…
A: Electron withdrawing group (through -I effect,-M effect,-R effect) attached to the benzene ring.…
Q: Which structure Is aromatic and has six electrons In the conjugated system? Click on a letter A…
A: Aromatic compound which follows huckel's rule i.e. 4n+2 rule where n is natural number.
Q: Which of the following is a 3-ring aromatic molecule? Anthracene quinoline indole purine
A: Which of the following is a 3-ring aromatic molecule?
Q: Which of the structures shown below is not aromatic? Click on a letter A to D to answer. CH, CH, B D
A: Aromaticity of a compound is defined as a property of delocalization of pi electrons which enhances…
Q: What is the major organic prou 0₂N 0000 D + HNO3 NO₂ H₂SO4 OH B NO2 NO₂ 66 с D
A: Organic reactions are those in which organic reactant react to form organic products.
Q: Synthesize the following compounds starting from benzene. Reactions contain more than two steps.…
A: The compound a can be synthesized by the following steps starting from benzene:
Q: The general formal for alkanes - 21 .compounds is A - CnH2n. B - CnHn+2. C-C₂H₂n+2. A O B General…
A: (21)Alkanes- Alkanes are saturated hydrocarbons. This means that their carbon atoms are joined to…
Q: Classify each of the following compounds as aromatic or non-aromatic. SH A B. D. C
A: The aromatic compounds are those compounds which should contain alternate single and double bonds…
Q: N. (A) (B) H 'N' „N. HN. B'
A: A compound is said to be aromatic if it follows the huckle's rule of aromaticity which are as shown…
Q: Which of the following compounds are aromatic? E B
A:
Q: This reaction is classified as: NCH3 CH3NH2 O A) Nucleophilic substitution B) Nucleophilic addition…
A: The answer is given as follows Ans: nucleophilic addition
Q: Which of the following is sn electrophile in electrophilic aromatic substitution? OH- NH3- Cl-…
A: ->Electrophile is the molecule or ion that is electron deficient and in some way could accept a…
Q: Which of the following lone pairs of purine participate in its aromaticity? N. C
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Draw the organic products formed when cyclopentene is treated withfollowing reagent. [1] CH3CO3H; [2] H2O, HO−Draw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. NaNO2, HClBenzene undergoes electrophilic aromatic substitution reaction with alkyl halide in the presence of Lewis acid like AlCl3
- Name and Draw the Phenolof all possible chemical (Electrophilic aromatic substitution) reactions of the compound namely; Halogenation (Chlorination or Bromination) NitrationDraw the organic products formed when cyclopentene is treated withfollowing reagent. (CH3)3COOH, Ti[OCH(CH3)2]4, (−)-DETCan hemiacetals cleave and form a carbonyl without acidic conditions? Or will PPh3 act as an acid and make a carbonyl?
- Draw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. CH3CHO, NaBH3CNAnswer all questions Q1. What are the various ways by which alkenes maybe synthesized. Q2. Give two examples each of unsymmetrical alkenes and reagents. Q3. Give three examples of reactions of alkenes which tesult in anti markovnikov additionBenzopyrene, naphthalene and pyrene are members of these group of aromaticcompounds: *Non-benzenoid aromatic compoundsHeterocyclic aromatic compoundsBenzenoid aromatic compoundsHeteronuclear compoundsWhat type of aromatic compound is pyridine? *Benzenoid aromatic compoundNon-benzenoid aromatic compoundHomonuclear cyclic compoundHeterocyclic aromatic compoundWhat property of aromatic rings prevent the involvement of the conjugated structure toaddition reactions? *Radical stabilizationResonance stabilityInductive effectAromatic effect
- What reagents would you use to prepare each of the following from 3-hexene? a. b.CH3CH2CH2CH2CH2CH3 c. d.why a CH3 group directs electrophilic aromatic substitution to theortho and para positions ?Based on the characteristics of the carbonyl group (C = O), what reactions or transformations take place with aldehydes and ketones? a. nucleophilic additions by oxygenb. electrophilic additions by carbon attackc. nucleophilic additions by carbon attackd. electrophilic substitutions through a carbocationand. acid-base because carbonyl can act as both an electrophile and a nucleophile



