Q: What differences in the IR spectra of the reactant and product would enable you to tell that each of…
A: As you have posted several parts and have not specified which part you want us to solve, hence we…
Q: Q4) Two Isomers have the chemical formula C,H, identify their structure based on their IR spectra…
A: The structure for both the isomers can be identified as follows: First of all degree of unsaturation…
Q: 5. Which IR signals will (a) appear and (b) disappear in the synthesis shown below. Explain your…
A:
Q: Explain how you could use IR spectroscopy to distinguish among the following three compounds. OH…
A: IR spectroscopy is used to determine the functional groups present in a molecule. There will be…
Q: Predict the splitting pattern(s) you would expect for the proton(s) in the molecule below that will…
A: Solution
Q: Which of the following compounds will show sharp IR absorptions at 3300 cm-1 and at 2150 cm-1? (d)…
A: IR spectroscopy is an important tool for the determination of the functional group in the compound.…
Q: Please match the appropriate functional group with the following IR spectra. LOD E 50 4D00 3000 g000…
A: Ir spectra of a compound has specific peaks to specific functional groups based on their vibration.
Q: Provide solution ?
A: (a) Given:
Q: Propanoic acid and methyl ethanoate are constitutional isomers. Show how to distinguish between them…
A: The question is based on the concept of IR Spectroscopy. we need to identify the features which…
Q: For each molecule propose a structure, identify important IR stretches and match each peak in the…
A: Determination of degree of unsaturation Degree of unsaturation Degree of unsaturation(U) =no.of…
Q: 5. Explain how you would distinguish the following compounds by infrared spectroscopy. CH3 -H- CH3…
A: The above compounds can be distinguished by ir spectroscopy by their streaching values. Above…
Q: Which of the following will have a doublet at 3300-3500 cm and a strong singlet at 1650 cmtin its IR…
A: To give the name of functional group , we would check that which bonds give peaks in the areas…
Q: Which of the following compounds has the lowest carbonyl stretching frequency? Explain your a.…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 1. For following IR spectra: A. Identify the functional groups and bond types present in the…
A: Infra-red spectroscopy (IR) involves the transition related to the vibrational modes of the molecule…
Q: 5. Which IR signals will (a) appear and (b) disappear in the synthesis shown below. Explain your…
A: Hydration of alkene gives alcohol which after oxidation gives ketone
Q: Which one of the following five compounds produced the IR spectrum shown below?
A: The predictions from IR spectrum are as follows: Alcohol/Phenol O-H Stretch at 3550-3200 (broad, s)…
Q: Assign as many peaks as possible in the unknown #1 (C8H8O) IR Spectrum
A: the solution is as follows:
Q: What functional group is most likely present if a compound shows IR absorption at these frequencies?…
A: IR spectroscopy gives information on the functional groups present in the sample. The IR spectrum…
Q: Which of the compounds (1-5) depicted below are the best match for the indicated IR spectrum?…
A:
Q: 6. For each of the compounds below, draw a rough estimation of its expected 'H NMR spectrum. CI 0.9…
A:
Q: Compound +4 IR 4000 Unknown #4
A: Infrared ( IR ) spectroscopy is one of the most significant and frequently used spectroscopic…
Q: Which compound is best represented by the following IR spectrum? Major peaks (cm¹); Oa Ob Oc Od Oe…
A:
Q: For each compound, assign likely structures to the fragments at each m/z value, and explain how each…
A: a. The fragments at each m/z value is given by
Q: IR spectra for four of the following compounds are given below. Match the spectra with the most…
A: Spectra 1: 3500 to 3000 cm-1 hydroxy stretch and 1690 cm-1 carbonyl stretch. Therefore the compound…
Q: Which compound below has a strong IR absorption at 1715 cm¹ and shows prominent peaks in the mass…
A: IR spectroscopy is used to determine the functional groups present in the sample. Different…
Q: Which one of the following five compounds produced the IR spectrum shown below?
A: The predictions from IR spectrum are as follows: Alcohol/Phenol O-H Stretch at 3550-3200 (broad, s)…
Q: Which of the following five compounds produced the IR spectrum below? LOH NH₂ OH OH you A 4000 *****…
A: IR spectroscopy is observed at infrared region which is used to identify the functional group from…
Q: Which of the following molecules will have a moderate, strong peak around 2700 - 2830 cm1 and…
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: 6. Which compound would be expected to show intense IR absorption at 2820, 2710 and 1700cm-1? А)…
A: The mentioned IR absorption frequency 2820 cm-1 is a peak related to the Aldehyde and it gives…
Q: Which of these would have a peak in the proton NMR which would disappear when D20 is added? OH но OH…
A: NMR spectroscopy is a very important tool for the determination of the structure of the organic…
Q: Choose the compound with the most intense IR signal at ~1700 cm1. CI A В O A O B o o o
A:
Q: Which compound would be expected to show intense IR absorption at 1640 cm1? O CH3CH2COCH3 O…
A: Infrared spectroscopy is a technique based on the interaction between infrared radiations and…
Q: Which of the following five compounds produced the IR spectrum below? OH NH₂ OH OH NH₂ 430 OA. First…
A:
Q: Choose the structure that best fits this proton NMR spectrum. CH₂(CO)OCH₂CH₂CH3 CH3CH₂(CO)OCH₂CH3 (…
A:
Q: Which of the following molecules will have a moderate, strong peak around 2700 - 2830 m1 and strong,…
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: Which of the following molecules (A and B) matches the IR spectra below. Explain your choice.
A: IR spectroscopy is an important tool for the determination of the functional group in the compound.…
Q: Determine the formula of the hydrocarbon compound where m/z=134. The mass spectrum indicates an M+2…
A: Mass spectroscopy helps in determination of chemical and structural interpretation based on…
Q: What major IR absorptions are present above 1500 cm−1 for each compound?
A: The IR absorption for the following compound can be given as- -C-H absorption occurs at 2900 cm-1…
Q: Q4) Two Isomers have the chemical formula C;H10 identify their structure based their IR spectra…
A: The given chemical formula can correspond to either an alkene or cycloalkane. The given spectrums do…
Q: For each of the following compounds, determine whether or not you would expect the IR spectrum to…
A: Compounds with OH, NH, alkynic H and alkeninc H show IR band left to the 3000 cm-1
Q: For each of the following compounds, determine whether or not you would expect the IR spectrum to…
A: IR spectroscopy is an important tool for the determination of the functional group in the organic…
Q: a) Which of the following molecules best corresponds to the IR spectrum below? OH b) c) HO 0.8 C,H,0…
A:
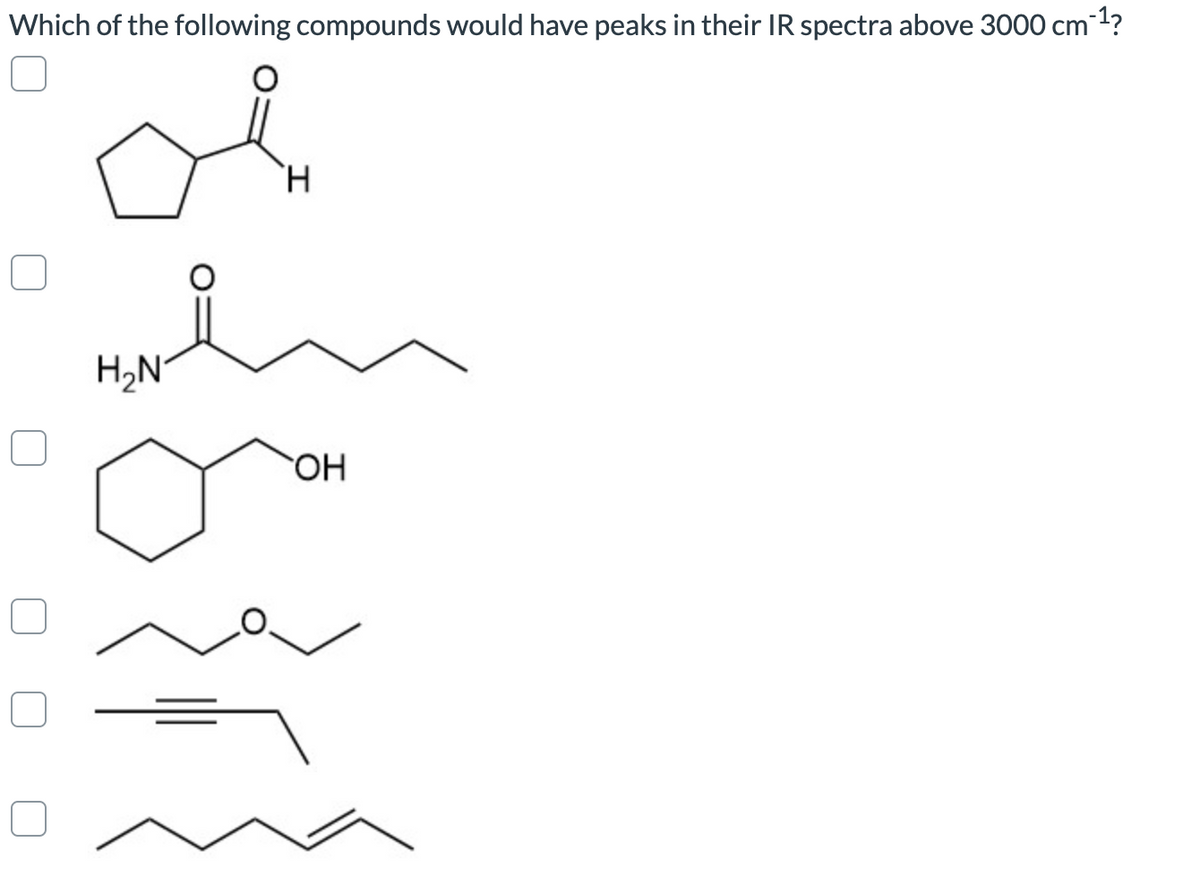
Q: Which of the following compounds will show at least one peak to the left of 3000 cm-1 (more than…
A: We can find out functional groups from IR spectra.
Q: For the following set of compounds, what is the main difference you should look for in their IR…
A: Ir is used to determine the functional group in the molecule
Q: Directions: Assign the IR spectra to the corresponding structures below. Cite the pertinent peaks…
A: For the above mentioned molecules, the carbonyl stretching frequency is common with all molecules…
Q: Which of the compounds match the IR spectrum shown below? Answer choices are shown below the…
A: On clicking pictures using a camera we are able to recognize human species. Similar to this…
Q: Which compound would most likely correspond to the IR spectrum below? OH H. HO 2 5
A: The structure of an unknown organic compound can be observed by knowing the empirical formula…
Q: OH B Making use of the tabulated data in the CHEM 110 spectroscopy datasheet, which compound, if…
A:
Q: Predict the splitting pattern(s) you would expect for the proton(s) in the molecule below that will…
A:
please help?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Please help me! Part 3A Set 1. Can IR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. What significant absorptions would be observed in the IR spectrum?What are the notable peaks in the IR spectra of each molecule?At what approximate positions might the following compounds show IR absorptions?
- Would you expect two enantiomers such as (R)-2-bromobutane and (S)-2-bromobutane to have identical or different IR spectra? Explain.Where might the following compounds have IR absorptions?Treatment of 3,4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation of a product with formula C6H10. Three products are possible. Name each of the three, and tell how you would use 1H and 13CNMR spectroscopy to help identify them. How would you use UV spectroscopy?

