Q: Consider the protons that are in the position ortho- to the amine group in the structure shown…
A: Downfield means proton at higher chemical shift value and upfield means proton at lower chemical…
Q: There are three possible products in the reaction below. Study the 'H NMR spectrum on the next page…
A: H NMR means Proton Nuclear Magnetic Resonance. In this method we can determine the number of…
Q: _3. Fill in the missing precursor to complete the transform below. он ? а. CН,Br b. CH;MgBr с.…
A: The missing precursor is CH3CH2MgBr
Q: The molecule ethylbenzene is seen to have the three highest fragments at m/z = 106, 91, and 65. The…
A: In mass spectrum the stable molecular fragmentation will lead to give base peak and which is 100 %…
Q: give the mass spectroscopy interpretation of the peaks Propan-1-amine / Propylamine
A: First , we will do the molecular ion fragments and then assign the peaks to the particular…
Q: Circle the peaks on the COSY spectrum that confirm your previous assignment of the vinyl hydrogen…
A: SOLUTION: Step 1: The major compound of 2-hexenal is Trans isomer and the minor product is cis…
Q: 3.11. What is the importance of 3000 cm-1? a. This is the frequency of the natural harmonic of the…
A: For question 1 Option 3 is correct as medium appearance at 3000 to 3100cm-1 indicates the alkene C-H…
Q: Consider the reaction as described below, then identify what change(s), if any, would be observed…
A:
Q: Which of the following molecules is responsible for this IR spectrum?
A:
Q: 15) Which one of the compounds corresponds to this IR spectrum. NH2 1000 2000 3000 4D00
A:
Q: Consider the protons that are in the position ortho- to the carbonyl containing group in the…
A: When nucleus is surrounded by lots of electron density then it is said to be shielded or upfield.…
Q: 2. Briefly describe how you could use IR spectroscopy to determine if the following reactions…
A: IR spectroscopy is an important tool for the determination of the functional group in the compound.…
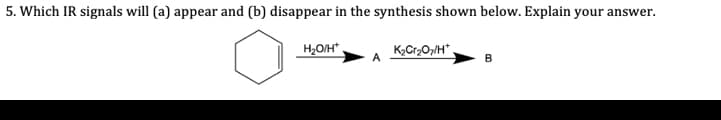
Q: 5. Which IR signals will (a) appear and (b) disappear in the synthesis shown below. Explain your…
A: Hydration of alkene gives alcohol which after oxidation gives ketone
Q: Draw the fragmentation pattern of this molecule. The major peaks are: m/z = 136 m/z = 135…
A: A question based on mass spectrum that is to be accomplished.
Q: Explanation on how chemical effects (for example, the conjugation of C=O with C=C double bond) alter…
A: Conjugation decreases the bond length of Bonds and hence there occurs decreases in Wavenumbers of…
Q: Which of these molecules best corresponds to the IR spectrum below?
A: given Ir spectrum find out structure
Q: What is the expected IR absorptions you would expect to see from the following molecule? OH If you…
A: Among all the techniques used commonly in recent days for structural evaluation, IR is one of the…
Q: The McLafferty rearrangement of the molecule below will produce an ion that will be detected at a…
A: McLafferty rearrangement: It is a cleavage of the α-β sigma bond by the transfer of γ-H from the…
Q: For each molecule below, draw what its 1. How many peaks you would see (how many different kinds of…
A:
Q: Where are you likely to find a new peak related to the product of this reaction? „Br Na* NH2 3500…
A: NaNH2 is a strong base and it converts alkyl halides to alkenes.
Q: How can IR spectroscopy be used to determine when the following reaction is complete?
A: IR stretching frequency : it is called infra red stretching frequency . when a molecule absorbs at a…
Q: Но- HN- Sketch out the C-NMR for the molecule above and discuss why the peaks are located where they…
A: From the 13C-NMR spectrum we can identified the type and nature of carbons present in the molecule.…
Q: How can IR spectroscopy distinguish between 1-hexyne, 2-hexyne, and 3-hexyne?
A: Depends upon the absorption bands for each compound, IR spectroscopy can distinguish 1-hexyne,…
Q: A student obtains an IR spectrum with an intense, broad absorption at about 3350 cm-1. The student…
A: Alcohols have a different IR absorption with the stretching vibration (OH and CO) OH = peak at…
Q: 25 20 15 PPM 10
A: In 13CNMR spectrum number of signals depict the different type of carbons in a compound. Carbons…
Q: Suggest how UV-vis spectroscopy could be used to determine whether each of the following reactions…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Q1. Why is it common to use CCl4 as a solvent in IR spectroscopy?
A: Infrared spectroscopy is the study of measurement of infrared radiations with matter. The…
Q: hat information can be obtained from the UV-visible spectrum for compound 1 and 2?
A: Two UV-Visible spectra for the compounds 1 and 2, which are to be analyzed.
Q: A. Below is a “Name Reaction” - Clemensen reduction. Describe how the IR spectrum could be used to…
A: In Clemmensen reduction, the reactant is a ketone, containing carbonyl functional group (-C=O). The…
Q: R; = 0.70 R = 0.55 R; = 0.47 -+ ------ Eluent: ELOAC `OH OH 1 2 3 5. Which compound corresponds to…
A: Option number C means compound 1 has higher rf value 0.70 because like dissolve like. Ethyl acetate…
Q: please help, i need to assign IR peaks with frequencies greater than 1500 cm^-1 ro a specific bond…
A:
Q: influence
A: Dear student this question is related to organic chemistry.
Q: Identify the important peaks in the following MS spectral data and draw the structure of the…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: s000 2000 1500 1000 500 NAVENUMBERI
A:
Q: Consider the spectra shown below and match them to the following compounds: CH, CH, CH, CH, CH, CH,…
A: As we know that both 13C and 1H NMR are identical according to their principles, the major…
Q: Choose the compound that best matches the IR spectra given below. Note: In case the labeled tick…
A:
Q: spectroscopy
A: The bond energy and the force constant of the conjugated C=O is comparatively lower than the…
Q: assign a structure to each of the following three spectra. The information is here
A: The matching is as follows:
Q: 3. C3HB You have seen these peak locations before. What does the coupling pattern tell you.…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: A low-intensity peak at m/Z = 31 can be seen in the mass spectrum of methyl acetate in Figure 3.…
A: The mass spectrum of methyl acetate has a low intensity peak at m/z = 31. The fragment that is…
Q: Which of the following molecules (A and B) matches the IR spectra below. Explain your choice.
A: IR spectroscopy is an important tool for the determination of the functional group in the compound.…
Q: how could you use ir spectroscopy to determine if the following reaction was successful
A: IR spectroscopy is an important tool for the determination of the functional group in the compound.…
Q: For each compound: i. Calculate the mass of the molecular ion. ii. Draw the most likely…
A: As per rule i should have to answer only three subparts so please ask remaining questions in another…
Q: 2) Here is an IR spectrum of one of the molecules below. Which one? Describe what specific features…
A: The ir spectrum is related to 1 butanol. Here the streaching frequency SP3 C-H streaching and C-H…
Q: Match a structure with the correct ¹H-NMR. You will, of course, notice that there are more…
A:
Q: simulate the NMR spectra and try to signal the 1H and 13C NMR signals of these 2 reactions.
A: IR spectroscopy stands for infrared spectroscopy. It involves studying the absorption of infrared…
Q: Fill in the table with all ¹H NMR characteristics for each signal and write down pertinent…
A:
Q: 3. What structures correspond to theses peaks? Show your reason.
A: The structure of ketones can be derived using mass spectral analysis. In case of ketone, the…
Step by step
Solved in 2 steps with 2 images

- I need help with this 13C NMR, please explain how to solve it.Given the Mass Spectrum, IR spectrum, and C/H NMR spectrum, what is the spectroscopy unknown or a molecule that matches this data.hi, can someone please help me interpret this IR? it was done in the lab and I've also attached the reaction we did to obtain this IR. which peaks correspond to the product/reactant/impurities? Thanks!
- hello, thanks so much for this!! i also have an IR and Mass Spec spectra with this assignment. I figured out the IR to match these same signals and come up with this same compound, but I cant seem to figure out how to do the fragmentation on the Mass Spec. It is given then M+ = 116, but there's baRely a peak thereThe molecule ethylbenzene is seen to have the three highest fragments at m/z = 106, 91, and 65. The base peak is at 91. Explain why this occurs and draw the structure of the fragmentsA. Below is a “Name Reaction” - Clemensen reduction. Describe how the IR spectrum could be used to tell if the reactions had been successful. Provide approximate wave numbers and the corresponding functional groups for the key absorption bands and/or peaks in IR. You might want to describe from two aspects: which peak(s) of what functional group(s) should appear in the IR of the product (that is different from the reactant); which peak(s) of what functional group(s) should disappear in the IR of the product (that is present in the reactant B. For the Clemensen reduction (same reaction), describe how the 1H NMR spectrum could be used to tell if the reactions had been successful. Provide approximate chemical shifts and the integral and the “splitting” of the “unique” in the NMR spectrum. You might want to describe from two aspects: which peak(s) should have a change in chemical shift(s); which peak(s) should have a change in integral; which has a splitting change; which peak(s) will be…
- Q1. Why is it common to use CCl4 as a solvent in IR spectroscopy? Q3. After completion of the previous reaction, you end up seeing the following peaks: 3300 cm-1 (broad intense peak), 3060-3040 cm-1 (2 medium intensity peaks), 3030-3020-3010 cm-1 (3 small peaks), 1640 cm-1 (low intensity peak), 1585 cm-1 (medium intensity peak), 1410 cm-1 (medium intensity peak), 1100 cm-1 (strong intensity peak), 1010 cm-1 (medium intensity peak), 900-690 (many peaks, medium intensities). Give the molecular structure of the actual final product for the previous reaction. (Hint: it is not a carboxylic acid, nor the ketone previously drawn.) Q4. You want to take the IR spectrum of acetylene. How would you prepare your sample? Mention all important details of the sample preparation (no need to describe the acquisition).Morning, Please help Draw what you would expect to see in the 1H NMR of this compound attached. Please show the number of signalsA low-intensity peak at ?/? = 31 can be seen in the mass spectrum of methyl acetate in Figure 3. Propose a fragment that could originate from methyl acetate and be responsible for this peak.

