Q: For each of the following reactions, predict the major product. NaOH H20, A EN H2 Raney Ni, A,…
A: The organic reaction for the oxidation of primary and secondary alcohols to carboxylic acids and…
Q: The standard free energy of hydrolysis, DGº of some compounds are given below: Compound DG…
A:
Q: What reaction occurs as a hydrochloric acid solution is added to a solution containing equal…
A: A solution that contain acetic acid and sodium acetate must be present with the following ions which…
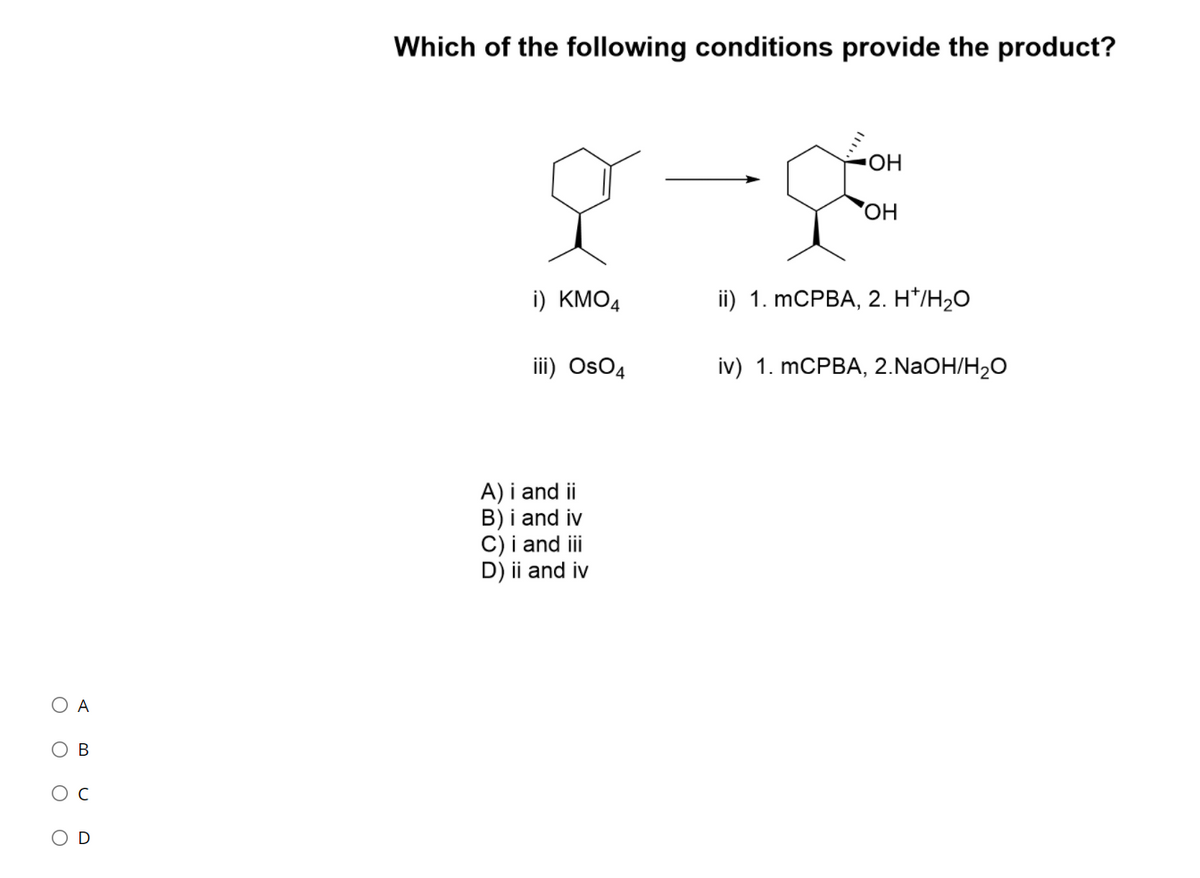
Q: Which of the following sets of conditions would accomplish the reaction shown? Br O OH OH 1. Br2 2.…
A: In this reaction, bromination takes place in alpha position of a carboxylic acid. The special…
Q: (i) Arrange the following compounds in an increasing order of basic strength :C6H5NH2, C6H5N(CH3)2,…
A: Basic strength of amine depends on electron density of nitrogen.
Q: For the reactions below, predict the major product. Hint: Consider whether the Lewis Base has formal…
A: Both the reaction are example of substitution reaction where one functional group is substituted by…
Q: and carbon dioxide after ozonolysis HO. HO after reacting with BH, and H,O, KOH
A:
Q: What is the major organic product of the following reaction? O A O B C A. Problem viewing the image.…
A: Carbonyl compound with secondary amine in acidic medium gives enamine as a product...
Q: Which of the following would be an insoluble product? KNO3 CaCO3 BaSO4 NaC2H3O2 AgBr
A: The completion of a chemical reaction is marked by several factors like change in temperature,…
Q: 10. The following reaction has variation of free energy value of AG = -2.1 kJ/mol CH;Br + H2S 2…
A: The equilibrium reaction taking place is given as, => CH3Br + H2S ------> CH3SH + HBr Given:…
Q: 1. In the boxes draw structures of the product and by-products for each reaction: NaOH(aq) (a) „NH2…
A:
Q: and methanol? OCH3 OCH3 OCH, mixture of a and b with the minor produc
A: E option is correct.
Q: ...|| 1. Hg[OAc)>, Н,0 2. NaBH.
A: Please find your solution below : The given reaction is an example of oxymercuration-demercuration…
Q: R - NH2 NH. HO NH DCC,THF NH RT, N2 (atm.) Вос `NH2 Вос `NH2 6(b-e) R= -CH,CH(CH,),.(2b) -CH,Ph,(2e)…
A: The reaction involves deprotection of amine group and results in formation of desired heterocyclic…
Q: Consider the acid-base reaction below: Ba(HSO4)2 + Ba(OH)2 ----> Compound A + H2O (OR B a(H S O4)2…
A: Ba(HSO4)2 + Ba(OH)2 ----> Compound A + H2O Ba(HSO4)2 + Ba(OH)2 ----> 2 BaSO4 + H2O BaSO4…
Q: What steps are needed to prepare phenylacetylene, CH,C=CH, from C,H,CH,CH,Br. Be sure to answer all…
A: Given steps are : What are the steps to prepare C6H5C≡CH from C6H5CH2CH2Br = ?
Q: In the following reaction in aqueous solution, the acid reactant is and its conjugate base product…
A: An acid is a substance that gives ions in its solution whereas a basic substance gives ions in its…
Q: a. H. Br OH Ph Ph "P HO
A: a. (i) Acid catalyzed amide hydrolysis. (ii) Acid FG to alcohol FG using LAH. (iii) Alcohol FG to…
Q: СООН СООН СООН СООН Br NO2 CH3 CI I II III IV
A:
Q: The reaction below will Select one: O a result in a yellow product Ob produce aryl diazonium ion О…
A: Primary amine reacts with sodium nitrite and hydrochloric acid to form diazonium salt. The aliphatic…
Q: Which of the following reactions would have a positive AS" ? (i) Na₂SO4(s)--> 2 Na*(aq) + SO4² (aq)…
A: Entropy order -> S(gas) > S(liquid) > S(solid) ->Hence , if number of gaseous moles…
Q: Which one of the following reducing agents is likely to be most effective in bringing about the foll…
A: LiAlH4 is the reducing agent for the reduction of ketones/aldehydes.
Q: What is/are the major product(s) of this reaction se 1. BH3 2. H2O2, NaOH 3. PB13 4.…
A: 2- methyl cyclohexene has 2 isomers.
Q: The following acid/base reactions proceed as written: CH3C=C:¯ + CH;OH CH;C=CH + CH3O- (CH3),N- +…
A: Acid - base reaction is a chemical reaction that involves the exchange of one or more ions of…
Q: Identify the suitable reagents for the following: 1) Вн, THF 2)H,O2, NaOH
A:
Q: What major product(s) (from Figure #16) results from the following reaction (from Reaction #16)?…
A:
Q: what will the major organic product of the following Reaction. excess -NaOH (3) с 0 B ventil o O A A…
A:
Q: 2) Predict the tupe of repction that Will product) That will be forned occur and or reactant(S) that…
A:
Q: Explain why the reaction in Problem 18.48 proceeds dramatically more slowly under neutral conditions…
A: The given reaction is,
Q: PbCl2 (s) + 2 KNO3(aq)
A:
Q: Which is true regarding the direction of the following reaction? CH3COOH (aq) + H2PO-4 >> CH3COO- +…
A:
Q: The starred N (of CN) is a heavy isotope of nitrogen (15N). Predict the major product of this…
A: The reaction of an aldehyde with NH4Cl and NaCN followed by hydrolysis will give alpha-amino acids.…
Q: Which of the following will not give Hoffmann bromamide reaction ? (a) CH3 -C-NH2 (Ъ) Ph — С-NH, (c)…
A: Hoffmann Bromamide reaction is fr the conversion of amide to the primary amine with one carbon less…
Q: Which of the following correctly completes the reaction? PbCrO4 + HNO3 → ? PbCrO4 + 2HNO3 → PbH2 +…
A: PbCrO4 + HNO3 ----> ? The balanced molecular eqn for the given reaction is – PbCrO4 + 2HNO3 →…
Q: Predict the product(s) of the reaction below: Al₂O3 → which is it below O2 AI AI + O AI +O2
A: This is the decomposition of Al2O3 it will give Al (aluminium) and oxygen gas (O2) The reaction is…
Q: write the Dwing sequence of reactions: C6H/AICI3 Mg A Cl2 C hv HBr H20 KSH ROOR КОН J H20
A:
Q: Choose the order that has the following carboxylic acid derivatives correctly arranged with respect…
A: The compounds which are synthesized from the carboxylic acid by removing the OH group of the…
Q: Are the following monoprotic or polyprotic H2S04 H2CO3 H3PO4 HNO3 HCl CH3COOH
A: There are two types of bases/acids. One is strong base/acid which is completely dissociable in into…
Q: Which of the following reactions is /are feasible? NaNH2 NaCI H3C CI NH3 H3C `NH2 II SOCI2 OH H3C OH…
A:
Q: 6. Provide the major organic product for the following reaction. 1. LİAIH4 НООС ΝΗ 2. НСІ, Н-0
A:
Q: Acetic acid: k1.75x10 Ammonia: k=1.8x10s Hydrofluoric acid: 7.2x10+ Hydrogen sulficde: ke5.7x10…
A: #2: For acetic acid, CH3COOH(aq), Ka = 1.75*10-5 For hydrogen sulfide, H2S, Ka1 = 5.7*10-8 , Ka2 =…
Q: Which of the following reactions is /are feasible? NANH, NaCI H3C CI NH3 H3C NH2 II SOCI, OH- H3C…
A:
Q: CH3 -с—сн-сн, ČH, ČI CH3OH CH3 -C-CI MeOH ČH3
A: Answer: Since we have methanol as nucleophile so reaction goes via SN1 . Formation of carbonation…
Q: Which of the following lists both acid species involved in the below equilibrium? HS– + CH3COOH ⇌…
A:
Q: (k) CH3-C=¢, C + CH—соон + CH3-COOH
A:
Q: 4, . Consider the following equilibrium: base + -H H-Base Which of the following base(s) would…
A: The chemical reaction that occurs between an acid and base is called acid base reaction.
Q: The standard free energy of hydrolysis, DG° of some compounds are given below: Compound DG…
A:
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A:
Q: Provide the ame reaction associated with each reaction below: Ph-CH2.CH3 Ans 1. R-CO-CH3 + N2H/HO"…
A:
Step by step
Solved in 2 steps

- The methoxy group is reported as having σpara and σmeta values that differ in sign (σmeta =+0.12; σpara = –0.27). What specific experiments are performed to determine these s values?Explain why the σpara and σmeta values are of different sign and what those signs indicate.Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Why does the final product has the opposite configuration compared to the reactant? Shouldn’t it form OMs first, then OMs gets substituted by Cl- via Sn2 (the 1st inversion of configuration) then the Cl- gets substituted by OCH3- (the 2nd inversion of configuration? To my understanding 2 inversions = same configuration
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?at 25C, which of the following reactions of PbS and H2O2 is more favourable?1) PbS + 4H2O2 -> SO2 + PbO2 + 4H2O2) PbS + H2O2 -> PbSO4 + H2OWhich compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaF
- Chemistry please provide the flow of electrons aswell!! thank you! using the starting material (on the left) to determine the sythetic route which will be the most reaosnable and effective to theres none, you have to start witth the begin products to get to finish needed by using the minmium reagents and reactions needed to get to the final productBalence the following half reation under basic conditions: PbO2 → Pb2-Predict the FINAL (?) product for each of the following reaction or synthetic chain:= - MgBr H2O H₂O LiAlH H₂O

