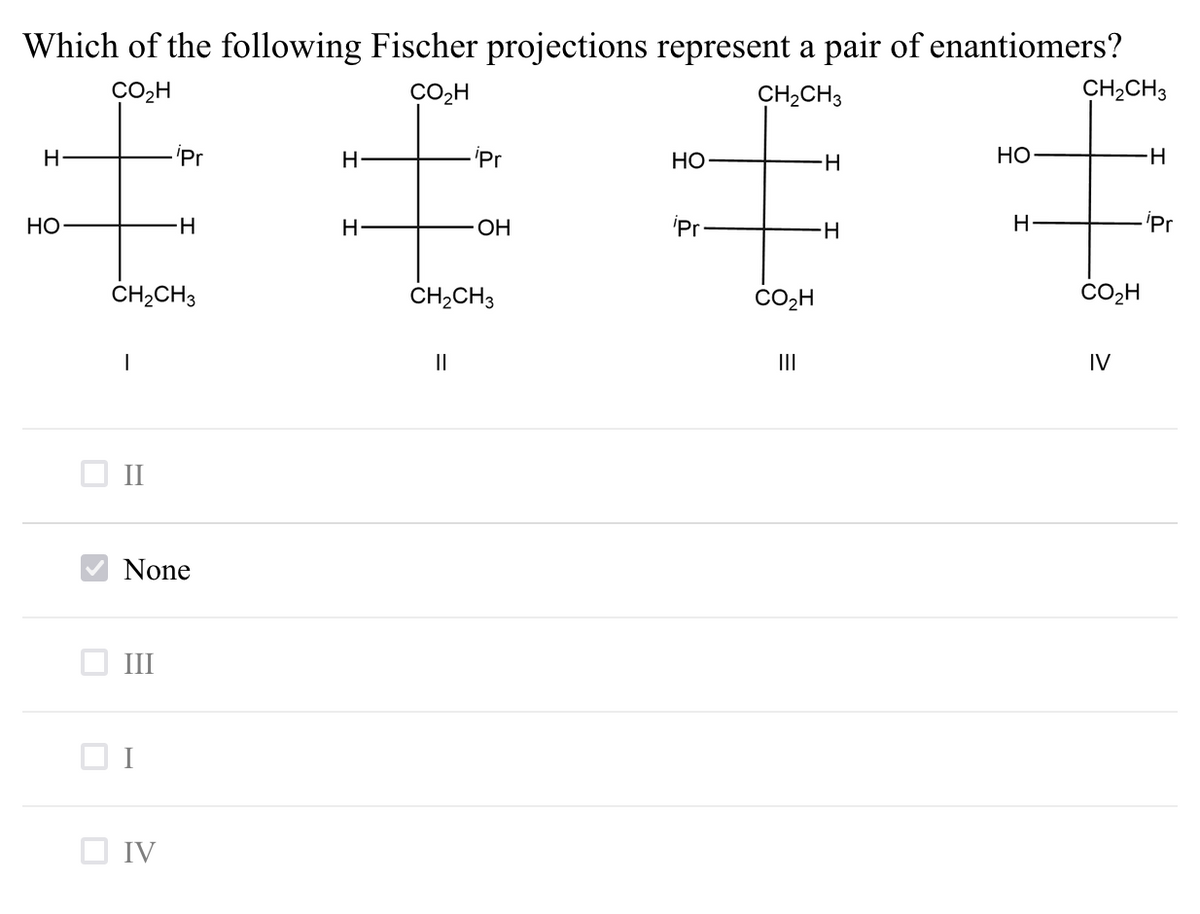
Which of the following Fischer projections represent a pair of enantiomers? CO,H CO2H CH,CH3 CH,CH3 'Pr H 'Pr Но H- НО H- НО H- ОН 'Pr- 'Pr CH2CH3 CH,CH3 CO2H II II IV None III O IV
Q: Which of the following represents enantiomers? CH2OH H CH3 CH3 HD он HO CH3 HO HD IOH HD ICH3 HO…
A: We can assign the enantiomers by considering (R, S) nomenclature or mirror image rule, If a…
Q: Which Fischer projections below are diastereomers? ÇO2H CO,H CO2H CO2H но H- но но CI H- CI F H- H…
A: Diastereomers are the non-superimposable non-mirror image isomers.
Q: Which two Fischer projections represent a pair of enantiomers? CH3 CH3 CH3 HOH H- H- CI но- C₂H5 A O…
A:
Q: . Enantiomers, diastereomers or same molecule? CH3 CH3 Br Br -Br Br- - Br H Br -Br H- H,C- Br H CH3.…
A: Isomer are pair of compound which differ in the structural formula but same molecular formula
Q: Which of the following is the enantiomer of the following substance? HI Br A. Both II and III 8. I…
A: The enantiomers of the substance is given below with explanation.
Q: (a) Br (b) CH3 (c) CH3 H CH3 H Br Br H H OH CH3 H3C OH H °CH3 OH Which of these compounds are…
A: Choral centre in a molecule can be defined as the carbon atom to which four different groups are…
Q: 6. Enantiomers, diastereomers or same molecule? CH3 Br CH3 Br City CH3 City Br CH3 Br
A: Given compounds are : Which are enantiomers or diastereomers or same compounds = ?
Q: en ONO ONO, en Co3 Co3+ ONO CI CI
A: Given compounds are: Optically active isomers have no plane of symmetry and have enantiomers.…
Q: Me Me BH3; H2O2 OH Me Me Me Me
A: the property of a chemical reaction in which a single reactant forms only one product of…
Q: How many stereoisomers of the following molecule are possible in principle? Attachments shoter CH3…
A: we have to calculate the number of stereoisomers determine the relation between the given two…
Q: Which two Fischer formulas represent a pair of enantiomers? C₂H5 CI- -H CH3 HO -H 1D-TH O C₂H5 I III…
A:
Q: I II CH3 Br H-C-Br Н—с—с ČH3 (A) Enantiomers (B) Diastereomers (C) Constitutional isomers (D) Same…
A: Enantiomers which are mirror image to each other or complete configuration should change (R,S)to…
Q: 6) Which of the following terms best describes the pair of compounds shown: enantiomers,…
A: Given :-We have to find which terms best describethe pair of compounds show :…
Q: Convert each of the following Fischer representations to a Haworth formula: CH CH но—с—н H-C-OH…
A: Haworth projection is a way of determining the Fisher projection into the cyclic structure.…
Q: For each pair of molecules, are they identical, enantiomers, diastereomers, constitutional isomers,…
A: Enantiomers contain chiral centers that are non-superimposable & mirror images. They only come…
Q: 1) Identify the stereochemical relationship between the following molecules (enantiomers,…
A: The stereochemical relationships between the given compounds are
Q: Which of the following Fischer projections represent a different stereoisomer? OH он HCH3 HCOH CH3…
A: Since there are multiple questions, we will answer only the first one. If you require the answer to…
Q: 3. A. What will be the number of stereogenic center in following molecules но. H,C, H .CH3 NCH3 i.…
A: i) For compound i) Number of stereogenic centre = 7
Q: For the following pairs of compounds, indicate the relationship (identical compounds, enantiomers,…
A: Isomer are compound having similar molecular formula but different structure
Q: a. Assign the configuration at each stereo center for each of the following reactants. Br:…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Identify each of the following pairs of Fischer projections as enantiomers or identical compounds.…
A: Enantiomers are the compounds which are mirror images of each other.
Q: Define each stereocenter as either (R) or (S). If the molecule does not have a stereocenter select…
A: ->Each chiral center has specific configuration R or S.
Q: Which of the following is the enantiomer of the given compound? HO CHS OH H. CH3 E locA CH2Br H. H.…
A: Two enentiomers have opposite absolute configuration
Q: What is the stereochemical relationship between the following pairs of molecules? Possible answers:…
A:
Q: Indicate whether the following pairs of compounds represent the same molecule, pairs of enantiomers,…
A:
Q: Identify the configuration (R or S) of the stereogenic carbon centre in each of the following…
A: Identify the chiral center (carbon which is having four different groups/atoms attached to it).…
Q: CH3 CH3 CH3 'CH3 CH3 H3C CH3 А E OH OH OH OH HO" HO HO "CH3 HO"CH3 F G H I Is molecule D achiral…
A: The molecule is said to be achiral or chiral based on the stereochemistry they possess if the…
Q: Which are pairs of enantiomers? CH3 CH3 H3C CI CI CH3 H3C ICI ČH2CH2CH3 ČH,CH,CH3 ČH2CH2CH3 I II III…
A: Conditions for pairs of Enantiomers: 1.Enantiomers are non-superimposable mirror images of each…
Q: Identify whether each of the following pairs of compounds are enantiomers or diastereomers, and…
A:
Q: 11) Answer each of the following questions using the Fischer projections below. CH2CH3 CH,CH3 CH2CH3…
A: Since you have asked multiple subpart of a question, we will solve the first three subpart for you.…
Q: A pair of enantiomers of the following is: * CH3 H-a CH3 it ClH HO-H HOH но- HOH H- -CI но- -H CI H…
A:
Q: Assign R or S configuration to each chirality center in the following biological molecules: (a) (b)…
A:
Q: Which of the following terms best describes the pair of compounds shown: enantiomers, diastereomers,…
A:
Q: 43. Which Fischer projections represent the products of this reaction? CH₂ Н=С CH₂CH₂CH₂ CH CH3 H…
A: -> OsO4 / H2O, NaHSO3 is reagent used for syn hydroxilation that means syn diol formed.
Q: Identify the enantiomers, meso compounds, and diastereomers among the stereoisomers below. CH3 CH3…
A: Stereoisomers are the pair of organic compounds that have the same chemical formula but different…
Q: For each pair of molecules, are they identical, enantiomers, diastereomers, constitutional isomers,…
A: Identical: Identical molecules are those which inconvertible by the C2 axis. These molecules have…
Q: For the substituted cyclohexane compound shown, identify the atoms that will interact with the…
A: Concept - In cyclohexane, there are two positions - 1) axial 2) equatorial The groups present in…
Q: Give the relationship between the two sets of compounds below. (enantiomers, diastereomers, meso,…
A:
Q: Which two Fischer projections represent a pair of enantiomers? CH3 CH3 C₂H5 H # H CI C₂H5 B HO H- H…
A: A question based on stereochemistry of organic reactions that is to be accomplished.
Q: Question is attached
A: This concept is explained on the basis of R/S configuration. Give the priority to the atoms…
Q: Draw the stereoisomers of the following molecules in flying wedge representation and Fischer…
A: The Flying-Wedge Projection is a three-dimensional representation of an organic molecule on a…
Q: 44 Assign R or S configuration to each chirality center in the following molecules: HO CH3 (a) OH…
A:
Q: Choose from A - E the term that best describes the isomeric relationship for each of the following…
A: Given these two compounds are chiral, because central carbon is attached to the four different type…
Q: Which of the following sugars has the configuration (2S, 3R, 4R)? сно сно HHOH HO- сно сно HO H HHOH…
A:
Q: Assign R or S configuration to the chirality center in each of the following molecules: (a) CH3 (b)…
A: Some points should kept in mind before R and S nomenclature. CIP rule i.e. Chain Ingold Prelog Rule…
Q: If you convert the 3-D structure below to its Fischer projection, which structure is it? COOH HOH₂C…
A: Priority direction around chiral carbon is anti clock wise then assign S configuration
Q: Translate the wedge-and-hashed structure to its Fischer projection. OH OH H. OH OH Select the…
A: • Draw semicircle goes from 1→2→3 then check clockwise/anticlockwise rotation and 4th Priority…
Q: 7. Which of the following drawings represents the enantiomer of compound X? CH, HO H,C-C-CH, ÓH CH…
A: Hello. Since your question has multiple questions, we will solve the first question for you. If you…
Q: 11. Consider the following compounds: CO,H СНО ОНС ОНС CO,H H;C- -H- H H HO- H,C H, OH H ČO,H ČH3 HO…
A:
Q: Describe the relationship between each of the pairs of isomers shown by recording Constitutional,…
A: We would tell the relation between the compounds based on their structure . Note : As you…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Identify pairs of molecules that represent enantiomers and diastereomers and identify each center stereogenic by writing R or S next to it.Which of the following pairs of structures represent the same enantiomer, and whichrepresent different enantiomers?H3C Cl \ / C=C / \ H2N I (R)-enantiomer (Z) isomer (S)-enantiomer (E) isomer meso compound
- Given the following structure and numbering: (a) Label the stereocenters of A as R or S and draw a Fischer Projection of the enantiomer of A with C1on top and C4 on bottom. (b) Draw a Newman Projection of a diastereomer of A looking down the C2-C3 bond with C2 in front.What is the ee for each of the following mixtures of enantiomers A and B? 95% A and 5% BFor each pair, indicate whether they are constitutional isomers, enantiomers, diastereoisomers, or identical molecules. A BDEF
- Determine the absolute configuration of the following compounds anddraw the structure (Fischer projection) of the other enantiomer.Draw Fischer projections for the four stereoisomers of 2-bromo-3-chlorobutane, label them (2R,3R), (2S,3S), (2R, 3S), and (2S, 3R)Assign R or S configuration to each chirality center in thefollowing molecules, Identify which pairs are enantiomers ordiastereomers
- Locate with an asterisk the stereogenic centers (if any) in the following structure: B. C6H5CH2CH(OH)CO2HIf molecules A and B are isomers of each other, then what kinds of isomers could they be (i.e., enantiomers, diastereomers, or constitutional isomers) under each of the following conditions?(a) Both molecules have the same IHD.(b) Molecule A has a ring but molecule B does not.(c) Molecules A and B contain different functional groups.(d) Molecules A and B share exactly the same functional groups.(e) Molecule A has a plane of symmetry but molecule B does not.Identify each pair of structures below as enantiomers (E), diasteromers (D), or conformers (C).

