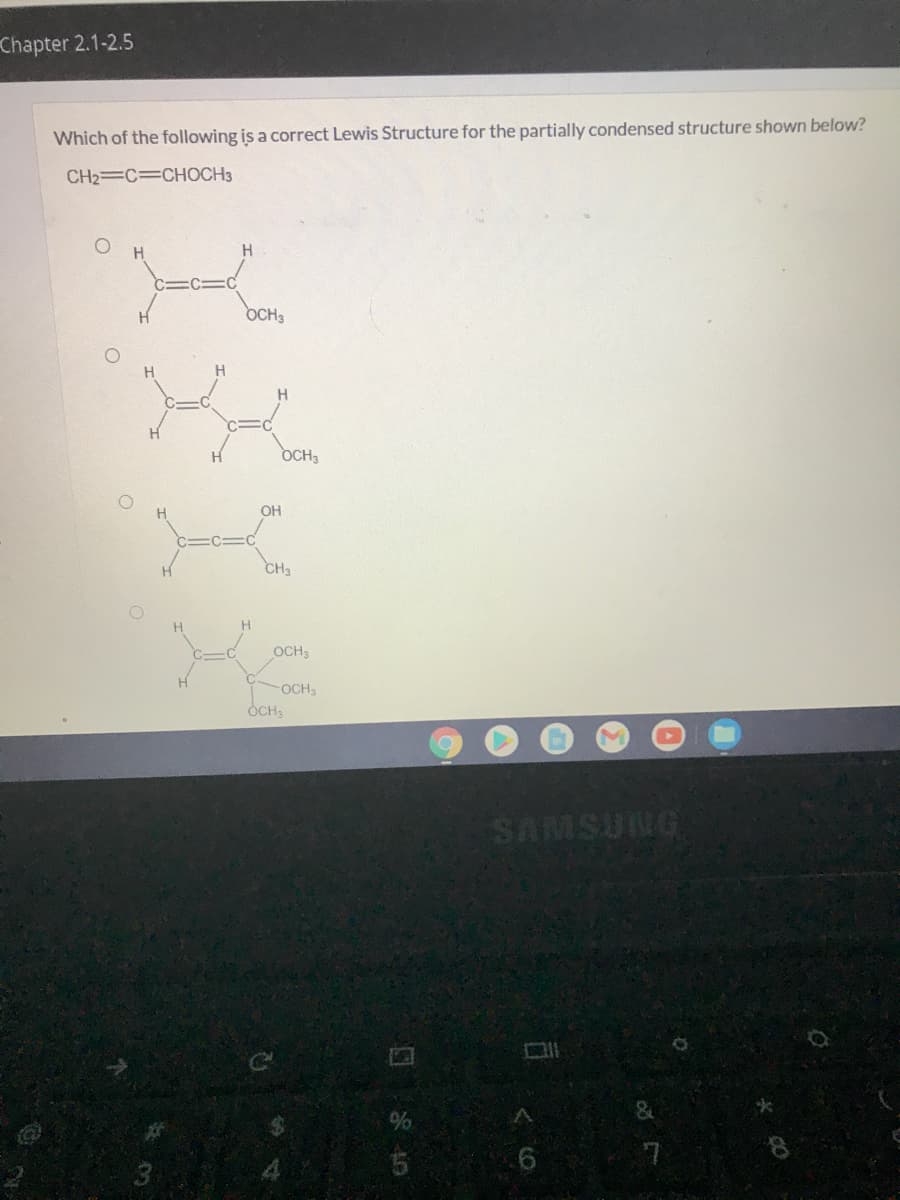
Which of the following is a correct Lewis Structure for the partially condensed structure shown below? CH2=C=CHOCH3 O H H. C=C=C OCH3 H H. H. OCH3 H. OH C=C=C CH3 H OCH3 H OCH ÓCH,
Q: VSEPR MODEL AND GEOMETRY MOLECULAR CH3NO2 C3H6O N2H4 CHCl3 CH6BN HCN C2H3LiO2
A: Hi, since there are multiple subparts posted we will provide you with answers for the first-three…
Q: 1. H,S 2. N,0 (the skeletal structure is N-N-O) 3. HNO3 4. PH3
A: Answer is given below But only first 3 we can Allow.
Q: Use bond energy data (based on the energy bond) to approximate the ∆H for the following chemical…
A: Given reaction is : C6H12O6 (s) → 2CH3CH2OH (l) + 2CO2 (g), ∆H = ? Change in enthalpy of the…
Q: Which of the organic compounds shown below exhibit cis-trans isomerism? Select one: O a.…
A: In organic chemistry , there are different type of compounds which are related with each other as…
Q: 1 Consider the ways in which two-dimensional structural formulas of the type shown above can provide…
A: Two- dimensional structures are a way to represent molecules in the sheet of paper and ease down the…
Q: 19. How many valence electrons are in a molecule of formaldehyde (CH,0)? A) 16 B) 12 C) 10 D) 8 E) O…
A: Explanation to the correct answer is given below.
Q: Describe the type of bonding between two carbons atoms in the red box of the structure below? H H ..…
A: This question belong to chemical bonding. We have calculate the bond between two carbon atom in the…
Q: How lone pairs of electrons many are present in CH3 CH 20 CH 2 CH3?
A: We have a structure of ether compound and we have to predict the number of lone pair in given…
Q: The chemical formula for hexane is C6H14. How many different isomers does a hexane have? Note:…
A: The compounds with the same molecular formula but different bond connectivity are structural…
Q: Ethambutol is a drug used to treat tuberculosis. Determine the shape around the indicated atoms in…
A: To find : Shape and add lone pairs to heteroatoms while determining the shape. Solution : Shape is…
Q: 3. The compound C4H6 has several isomers. Draw each of the isomers and determine the molecular…
A:
Q: Condensed Structure Skeletal Structure IUPAC Namе CH,CH,CHCH,CHCH,CH,CH3 ČH3 ČH3 CH3 CH;CH2-C-CH,CH2…
A: The skeletal structure of molecule is also called as line-angle formula. It is shorthand formula to…
Q: How many valence electrons are there in a correctly drawn Lewis structure for formamide, HCONH2 (…
A: Valence electrons are the electrons present in the outermost shell of an atom. Example: Valence…
Q: compound hydrogen-bonding force Between Between molecules of the compound and molecules of water?…
A: Hydrogen bonding is a special type of dipole-dipole attraction between molecules, not a covalent…
Q: Determine the number of valence electrons in acetone (CH,C(0)CH3) and then draw the corresponding…
A: Lewis structures is used to represent the covalent bonds formed between the atoms using valence…
Q: What type of molecular geometry does the molecule shown below have S=C=S
A: For the compound , CS2 the central atom is Carbon which forms 4 bonds and it forms 4 bonds. Hence,…
Q: Before we jump into functional groups let us practice drawing structures first. Fill in the table…
A: Molecular Formula is the way of representation of number of different kind of atoms in the compound.…
Q: How many sigma and pi bonds ( in total) are present in the following organic molecule? Give the…
A: A molecule can be formed by a combination of atoms. The atoms in a molecule are bonded by atoms. The…
Q: Carbons: S CH3OCO NC2 57
A: Condensed structural formulas are the same as structural formulas, with the only difference that…
Q: H H H H H H H H of a) Name the molecule diagramed above.
A: The name of the diagram is
Q: Draw a Lewis structure for the molecule below, showing all lone pairs. You may abbreviate any methyl…
A: A Lewis Structure is a simple representation of the valence shell electrons in a molecule. In the…
Q: O REPRESENTATIONS OF ORGANIC MOLECULES Drawing a skeletal structure from a Lewis structure Convert e…
A:
Q: Determine the molecular formulas and then write line-angle (skeletal) structures for the following…
A:
Q: In complete sentences, describe why someone might expect the bond angles in methane (CH4) to be 90°,…
A: Methane has tetrahedral tetrahedral structure in which the four hydrogen atoms gets bonded to the…
Q: Which molecules are NONpolar? :F: H F C=0: :NEN : :F: O SIF4 O N2 O CH20 O CH3CI O C2H4
A:
Q: но. HO HO. Но Figure 1. Citric Acid 1. Citric acid has eight hydrogens, but only four are shown in…
A: Solution: The formula for citric acid is C6H8O7 . In the given structure four hydrogens are…
Q: and skeletal structures of the following molecules shown in ral formula. 2. Η Η Η Η ΗΗ…
A: Condensed and Structural formula for the given compounds are shown below:
Q: Draw a skeletal ("line") structure of this molecule: CH3 HO-CH2 C CH2
A: This is a four carbon compound.
Q: 2. Complete the following for methane, the simplest saturated hydrocarbon. blimil.. Methane, CH4…
A: Polar compound are those in which there is electronegative difference between atoms. In these…
Q: 41. Which of the following is the appropriate conversion of the condensed structure, CH;COCH3, to a…
A: In skeletal form we don't show carbon and hydrogen we show only hetro atom ,and hydrogen when it…
Q: ID the molecules( XeF2O, CIO4, HNO2, CH3CH(NH2)COOH) with the following labels (1 is more than one…
A: Oxyacids are those in which acidic proton comes from O-H bond which attached with any heteroatom.
Q: H3C CH3 -CH3 Hl Br (а) (b) (с) HO CI H3C H3C- CH3
A: Given that : We have to name the following molecules :
Q: pper right corner of this assigni Questions: ) Draw the skeletal structure of the following co H H.…
A: 1) given molecule is in skeleton form so no need to draw again 2)R and S given by CIP rule in which…
Q: Formula Model Molecular geometry BeCl, ball & stick labels PO,(OH); ball & stick labels NO;CI ball &…
A:
Q: What are the polarities of CH₂Cl2 and CF₂Cl₂? + t O Both are nonpolar. O Both are polar. O CH₂Cl₂ is…
A: In chemistry, the polarity of molecule is decided by the net dipole moment of the molecule in the…
Q: Which formula represents an organic compound? A) NaCl B) BaSO4 C) PH3 D) C4H10 How many covalent…
A:
Q: 76. This skeletal structure corresponds to the molecular formula : Но HO. Ago HO OH
A: The Molecular structure can also be made as: C-atom will have 4 bonds O-atom will have 2 Bonds…
Q: Draw the skeletal structure of the follow H. H H. HO, H. H. H2N H.
A: Interpretation- To draw a skeletal structure of given compound so basically asked only skeletal…
Q: In complete sentences, describe why someone might expect the bond angles in methane (CH4) to be 90°…
A: Methane has tetrahedral tetrahedral structure in which the four hydrogen atoms gets bonded to the…
Q: Although we usually don't show lone pairs on skeletal structures, sometimes it's necessary, for…
A:
Q: ОН HOČCH,CH200CH2CH2CH,ČHCHCH,F ČH=CH2
A: The bond line structure of the given compound =
Q: b) For those compounds that can exist as cis and trans isomers, draw and label the isomers. 1.…
A:
Q: Convert the Lewis structure below into a skeletal structure. H H C-H :0: H H H H || | H-0-C -C=C-…
A: Rules to draw skeletal structure from Lewis structure: 1) Draw 2-dimensional structural formula. 2)…
Q: Determine if each of the following molecules is polar or non-polar. H HH HH H HHH HH HH H. Br H…
A: It is a non-polar molecule as there is no extra atom attached other than C and H. Here, the electron…
Q: Draw a skeletal structure for the molecules in parts (a) and (b), and a condensed structure for the…
A: The organic compounds are composed of carbons and hydrogens. The structures of the organic compounds…
Q: HO, The condensed structure for the molecule shown in the figure is C6H1602 CH3CH2CH(CH3)3CO2H…
A: In condensed structure, the covalent bonds present on each carbon are omitted and each structural…
Q: The chemical structure of glycine (C₂H₂NO₂) is shown below. Highlight each atom that is in a…
A:
Q: Convert the Lewis structure below into a skeletal structure. H H-C-H H н :0: Н H || | H C-C -C-C-…
A: We have to draw the skeletal structure of given structure.
Q: 1. Write the structural formulas for four isomers of the molecular CaHs. Include the cis and trans…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Condensed structure for 2-methyl-2-propanol. Also do you know the defintion or an idea of what a condensed structure is? I can't find it anywhere and it was never explained to us. Thank you in advanceConsider the following three molecules, A-C:Make a continuous model for C4H10 by using 4 black 4 hole carbon atoms, 10 white one hole hydrogen atoms, and 13 pink bonds. Then write in wedge dash notation C4H10 and add in missing hydrogen atoms.
- Can you help me with the explanation of the line bond structure, condensed structure, skeletal structure and all these terms give me examples too?I need to make a skeletal strcuture for this formula and i have no idea how. im very new to organic chem and i did the lewis structure but idk how to turn it into a skeletal structure. help please (CH3)3COO(CH2)2CCOCH2NHCHOConsidering each of the given ball-and-stick models: [1] Does the dark red atom have one or more lone pairs of electrons on it? Explain your choice. [2] Give an example of a molecule that has the indicated geometry.
- Between 1865 and 1890, other possible structures were proposed for benzene, such as those shown here. Considering what nineteenth-century chemists knew about benzene, which is a better proposal for benzene’s structure: Dewar benzene or Ladenburg benzene? Why?Below each structure in the previous question is a “condensed structure” that tells you somethingabout how the atoms are arranged. Draw complete Lewis structures for each of the followingcondensed structures. (The net charge, if any, on each molecule is given at the end.) a. CH3CH2 b. CH2CH2 c. CH2CCH2 d. C( CH3)3+ e. BF4 f. NCO g. CH2OH+ (two different acceptable answers) h. CH2CHCHCHCH2+ which may alsobe written as CH2(CH)3CH2+ (more than one acceptable answer)Consider the molecule below. Determine the molecular geometry at each of the 2 labeled carbons.
- 7. For the compounds, C13 H28 and C15 H30 , that are alkanes, draw the line/skeletalstructure. A. C13 H28 : B. C15 H30 :For SCN-1, find: 1. number of valence electrons 2. Draw the Lewis Diagram 3. number of electron groups 4. number of lone pairs 5. electron group geometry drawing & name 6. molecular geometry drawing & name 7. bond angle(s)? 8. polar? 9. resonance?pls help ASAP! “which term best describes the relationship between the following two molecules?”



