Which of the following is a correct practice in handling reagents? A Return any excess reagent to its source container. B Clean up any spills after finishing the laboratory work Never insert spatulas, spoons, or knives into a bottle containing solid chemicals D Dispose any liquid chemicals in the sink
Which of the following is a correct practice in handling reagents? A Return any excess reagent to its source container. B Clean up any spills after finishing the laboratory work Never insert spatulas, spoons, or knives into a bottle containing solid chemicals D Dispose any liquid chemicals in the sink
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
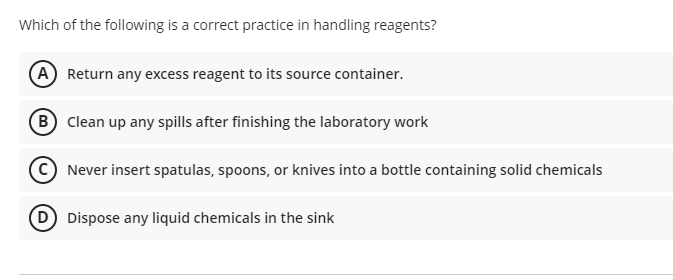
Transcribed Image Text:Which of the following is a correct practice in handling reagents?
A Return any excess reagent to its source container.
B Clean up any spills after finishing the laboratory work
© Never insert spatulas, spoons, or knives into a bottle containing solid chemicals
D Dispose any liquid chemicals in the sink

Transcribed Image Text:Francis A. Santos
A chemist obtained the following data for the percent compound Z in triplicates (n=3) of an insecticide preparation: 7.47, 6.98, and
7.27. Calculate the 90% confidence limit for the mean of the data assuming that only information about the precision of the method
is the precision for the three data points.
A 7.24 + 0.74%
B) 7.24 + 0.27%
7.24 + 0.13%
7.24 + 0.42%
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 6 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

