Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter24: Catalytic Carbon-carbon Bond Formation
Section: Chapter Questions
Problem 24.39P: E. J. Coreys 1964 total synthesis of -caryophyllene (essence of cloves) solves a number of problems...
Related questions
Question
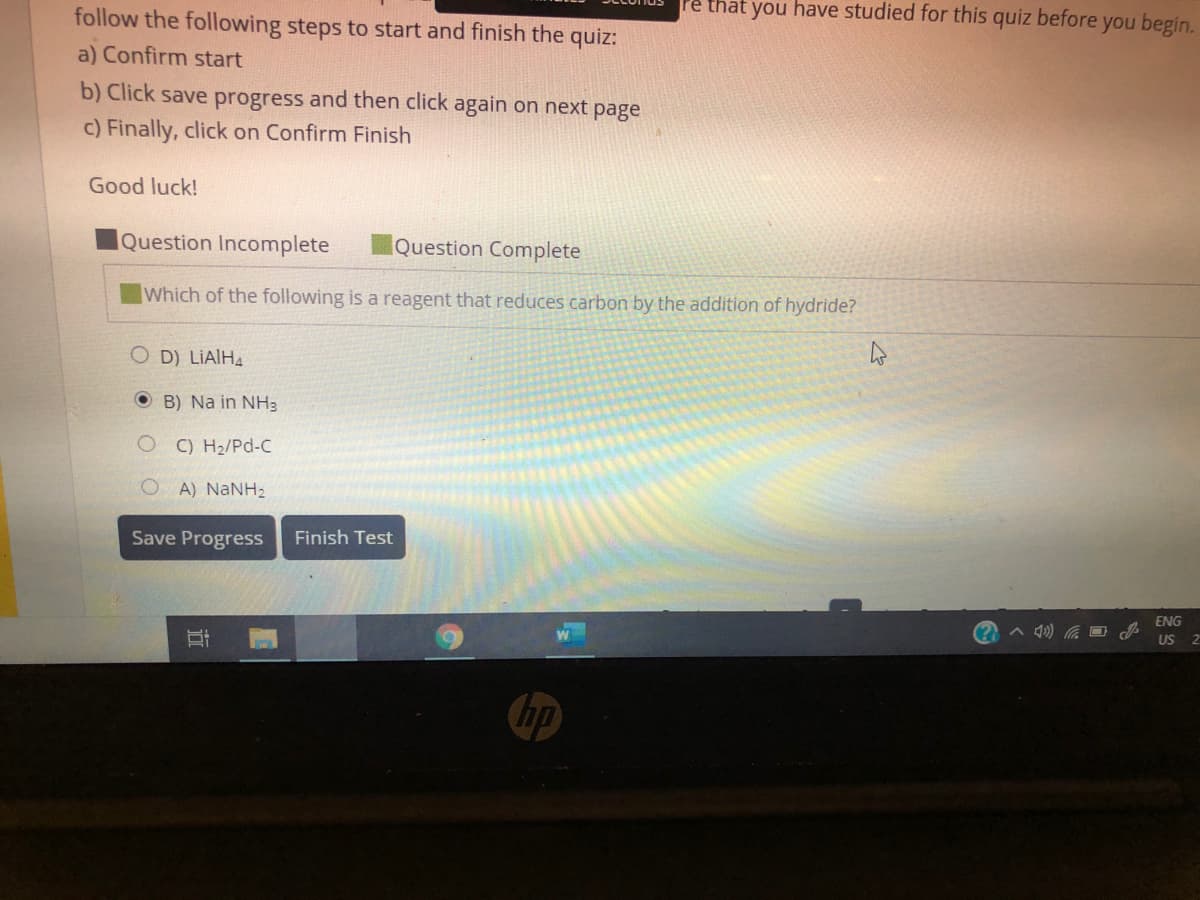
Transcribed Image Text:follow the following steps to start and finish the quiz:
re that you have studied for this
quiz before
you begin.
a) Confirm start
b) Click save progress and then click again on next page
c) Finally, click on Confirm Finish
Good luck!
Question Incomplete
Question Complete
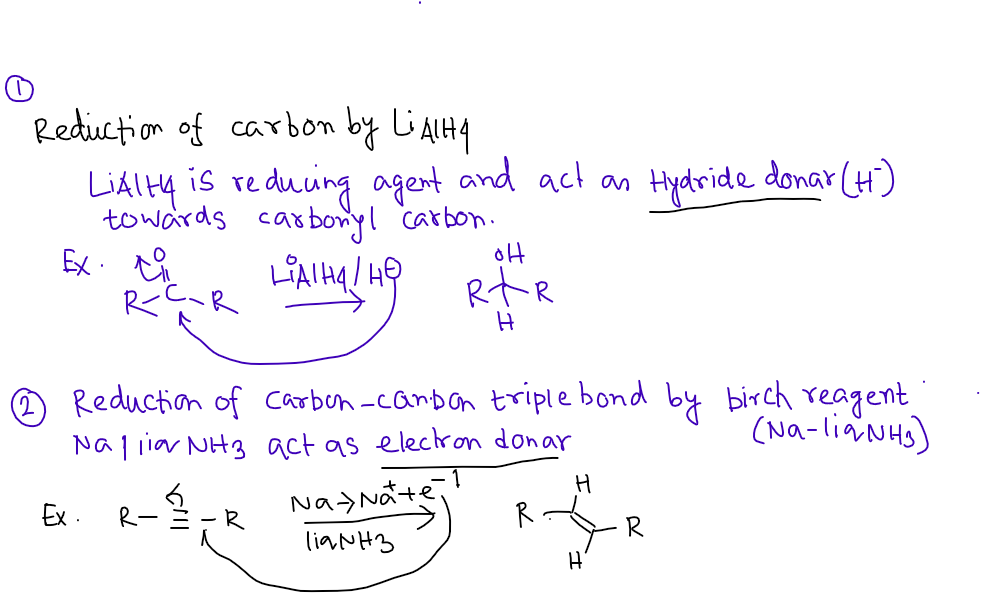
Which of the following is a reagent that reduces carbon by the addition of hydride?
O D) LIAIHA
O B) Na in NH3
C) H2/Pd-C
OA) NANH2
Save Progress
Finish Test
ENG
US 2
Expert Solution
Step 1
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning