Q: Nitration of benzene is carried out by which of the following reactive ?species NO3 O HNO3 O "NO2 O…
A:
Q: Which of the following is not a nucleophile? NH4+ NH3 CH3OH OH-
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which molecule can loose a hydrogen and become a nucleophile? Select one: a. H3C– ECH b. H;C- -CH3…
A: The terminal alkynes or 1-alkynes are weak acids and loose a hydrogen when treated with strong base…
Q: Which of the following anions is the most nucleophilic in polar protic solvents? а. F- b. Cl- С. Br-…
A:
Q: Which of the following are most likely to behave as electrophiles, and which as nucleophiles?…
A: Br2 can undergo heterolytic cleavage to form Br- and Br+ ions. Br+ is very unstable due to lack of…
Q: How many nucleophilic centers are present in this molecule? oho „NH2
A:
Q: Rank the nucleophiles in following group in order of increasing nucleophilicity. H2O, −OH, CH3CO2-
A: Water is a weak nucleophile because it is neutral. Since acetic acid is more acidic than water, the…
Q: Which of the following cannot be a nucleophile? он Br III IV O II
A: Nucleophile is an species that is able to attack on the electrophile Nucleopge is an electron rich…
Q: ?Which of the following is not a nucleophile اخترأحد الخیارات a. NH3 -b. Br c. FeBr3 d. CH30CH3
A:
Q: Which of the following is the strongest nucleophile? O H2S O NH3 O PH3 O CH3OH O H2O
A: The compounds given are H2S, NH3, PH3, CH3OH and H2O.
Q: Which of the following nucleophiles would add to an alpha, beta- unsaturated ketone via direct…
A: Given Reactant Alpha,beta-unsaturated ketone reagent which give Direct addition = to be determined
Q: Which of the following are most likely to behave as electrophiles? * (a) NH4+ (b) C=N- (c) Br+ (d)…
A: Answer
Q: Which of the following correctly represents a nucleophilic attack? :o: :CN ö: : :CN ..O :
A: Nucleophile is reactive species that contains negative charge and attacks on electron deficient…
Q: In the following reaction, which chemical species is acting like a nucleophile? [ H,CO CH,O H;CO b)…
A: Nucleophile is the one which has the ability to donate a pair of electrons or possising negative…
Q: CH3 CH3-CECH CH3-C-CH3 CH3-0-CH3 EN: I II III IV
A: Electrophiles have low electron density while nucleophiles have high Electron Density.
Q: Which of the following nucleophilic substitution reactions will take place?
A: Nucleophilic substitution takes place only when a group attacks and one group leaves.
Q: Which of the following cannot react as a nucleophile? I. CH3NH2 II. (CH3)2N III. (CH3)3N IV.…
A: Nucleophiles: A nucleophile is defined as a substance that donates the electron pair to…
Q: Select which molecule is the better nucleophile in the following pair: a). Br- or Cl- in H2O b).…
A: A better nucleophile is one which can easily donate lone pair and negative charge should be stable…
Q: Select the strongest nucleophile from the list below. OH O CF O HS-
A: The species which have tendency to donate electron pair and are electron rich in nature is known as…
Q: Which of the following is NOT a good nucleophile for SN1 reactions? 1) NaOCH3 2) CH3OH 3)…
A: SN1 reactions or unimolecular nucleophilic substitution follows in two steps where in the first step…
Q: In the following reaction, which chemical species is acting like a nucleophile? 5 H,CO N- CH;O* a)…
A: The chemical species can be divided as electrophile and nucleophile on the basis of the electron…
Q: Which of the following is the least reactive towards a nucleophile? O C A Н3С о A Н Н С O B CH3 C…
A:
Q: In the following reaction which species acts as the nucleophile? OCH,CH3 (H3C),N, CH;CH,OH OCH,CH3…
A: Since you asked multiple question we will solve first question for you. If you want to specify…
Q: Which of the following species is likely to be a nucleophile and which an electrophile? (a) CH3CI…
A: Electrophile is species which loves negative charge and nucleophilehile is species which loves…
Q: Which of the following is the strongest nucleophile? OH- H20 H2S HS-
A: Answer :- HS- -------------------------------------
Q: In the presence of a nucleophile, which of the following will most likely undergo conjugate…
A:
Q: A nucleophilic substitution reaction is shown below. Identify the nucleophile. I A) CH:S- B) Na II…
A: Nucleophile: In a chemical reaction a nucleophile is a species that forms bonds with electrophiles…
Q: Which of the following are most likely to behave as electrophiles? * (a) NH,+ (b) C=N- (с) Brt (d)…
A: Br2 can undergo heterolytic cleavage to form Br- and Br+ ions. Br+ is very unstable due to lack of…
Q: Identify ALL of the nucleophiles in each of the following molecules. (а) (c) `NH2 tu (b) -Li (d)…
A: a) The nucleophiles in the given compound is O. b) The nucleophiles in the given molecule is…
Q: For each of the following pairs of species, which is the stronger nucleophile in ethanol? Explain.…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Which of the following is the STRONGEST nucleophile? -H- -OH I II II IV а. I b. II с. Ш d. IV
A: a) The compound I is the weakest nucleophile because there is no heteroatom as well as it is…
Q: Which of the following is the best (strongest) nucleophile? NH3 H20 NH2 CH30 II II IV Select one:…
A:
Q: Which of the following species is likely to be a nucleophile and which an electrophile? (a) CH3CI…
A: This question belong to reaction mechanism organic chemistry. Electrophile : all positively charged…
Q: Which of the following is the strongest nucleophile ? О а. СНз O b. NH2 С. O C. HO O d. F O e. NH3
A: Nuclephile are species which are able to donate lone pair of electron
Q: Which of the following reactions take place more rapidly when the concentration of the nucleophile…
A: Given reactions:
Q: 1. For each of the following molecules, put a box around the nucleophilic atom(8). MgBr SH H.
A: A nucleophile is a species which is highly electron rich in nature while an electrophile is defined…
Q: Which one of these provides the strongest nucleophile in an SN2 reaction? a) HC=N b) NaC=N c) NH3 d)…
A: The nucleophilicity of the above mentioned compounds depends on the extent of the dissociation of…
Q: Which molecule can loose a hydrogen and become a nucleophile? Select one: a. H3C- ECH b. H3C- -CH3…
A: Solution:
Q: Rank the following compounds in order of increasing reactivity with nucleophiles (from least…
A: In this question, we want to arrange all three Compound in the Increases order of reactivity. How…
Q: Which of the following species are likely to be nucleophiles and which electrophiles? Which might be…
A:
Q: Which is the strongest nucleophile? 2 3 4 O 1 O 2 O 4
A:
Q: Which of the following are most likely to behave as electrophiles? (a) NH4+ (b) C=N- (c) Br+ (d)…
A:
Q: Which of the following species is likely to be an electrophile and which is nucleophile a) Hcl…
A: Electrophiles are those species which are electron deficient. And Nucleophiles are those species…
Q: Which of the following species are likely to be nucleophiles and which electrophiles? Which might be…
A:
Q: Which of the following is the most reactive to a nucleophile? O ethyl ethanoate O ethanoyl chloride…
A: The nucleophilicity of the considered compounds has been compared below.
Q: Rank the nucleophiles in following group in order of increasing nucleophilicity. −OH, −NH2, H2O
A: The word 'nucleophile' comes from two words- nucleus and 'philos' means love. Nucleophiles are…
Q: Which of the following are electrophiles, and which are nucleophiles? H− CH3O− CH3C=CH CH3CHCH3…
A: Given, H− CH3O− CH3C=CH CH3CHCH3 NH3
Q: List the following in increasing order of nucleophilic strength.…
A: Kindly get the answer given below.
Not a nucleopihile
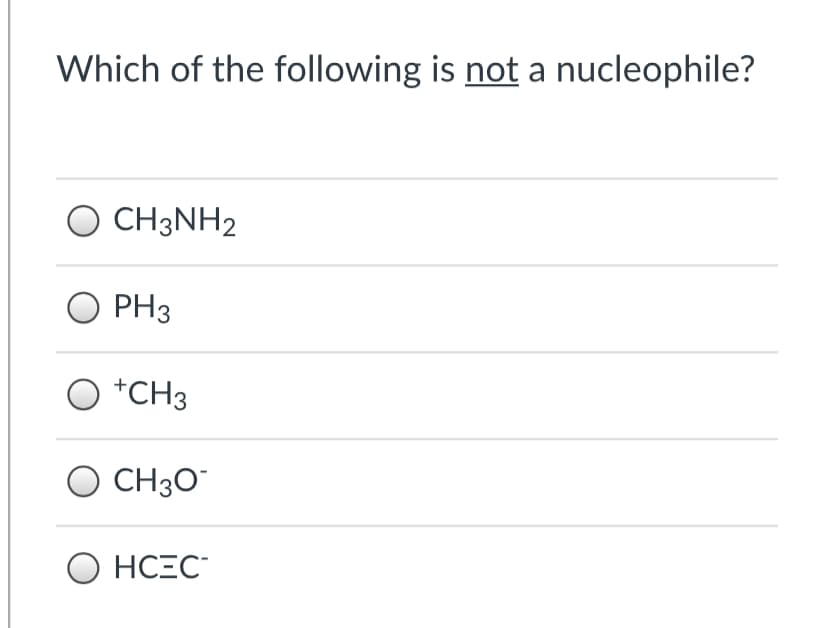
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Which of the following species is likely to be an electrophile and which is nucleophile a) Hcl b) CH3NH2 C) CH3SH D)CH3CHOWhich of the following are most likely to behave as electrophiles, and which as nucleophiles? Explain.(a) NH4(b) CqN (c) Br (d) CH3NH2 (e) HOCqCOHWhich is the strongest nucleophile among the following? CN- , OH-, CH3OH, NH3
- Are the following nucleophiles, electrophiles, they be both a nucleophile or electrophile, or neither? pyridine alkenes Hg+ (ex. Hg(OAc)2 CH3ONa PBr3In the following reactions; identify substrate, nucleophile and leaving groupWhich of the following SN2 nucleophiles does not lengthen the carbon-carbon chain? KC≡C-C6H5 LIC≡N NaC≡CH KOCH3 NaC≡CCH(CH3)2

