Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 48SST
Related questions
Question
100%
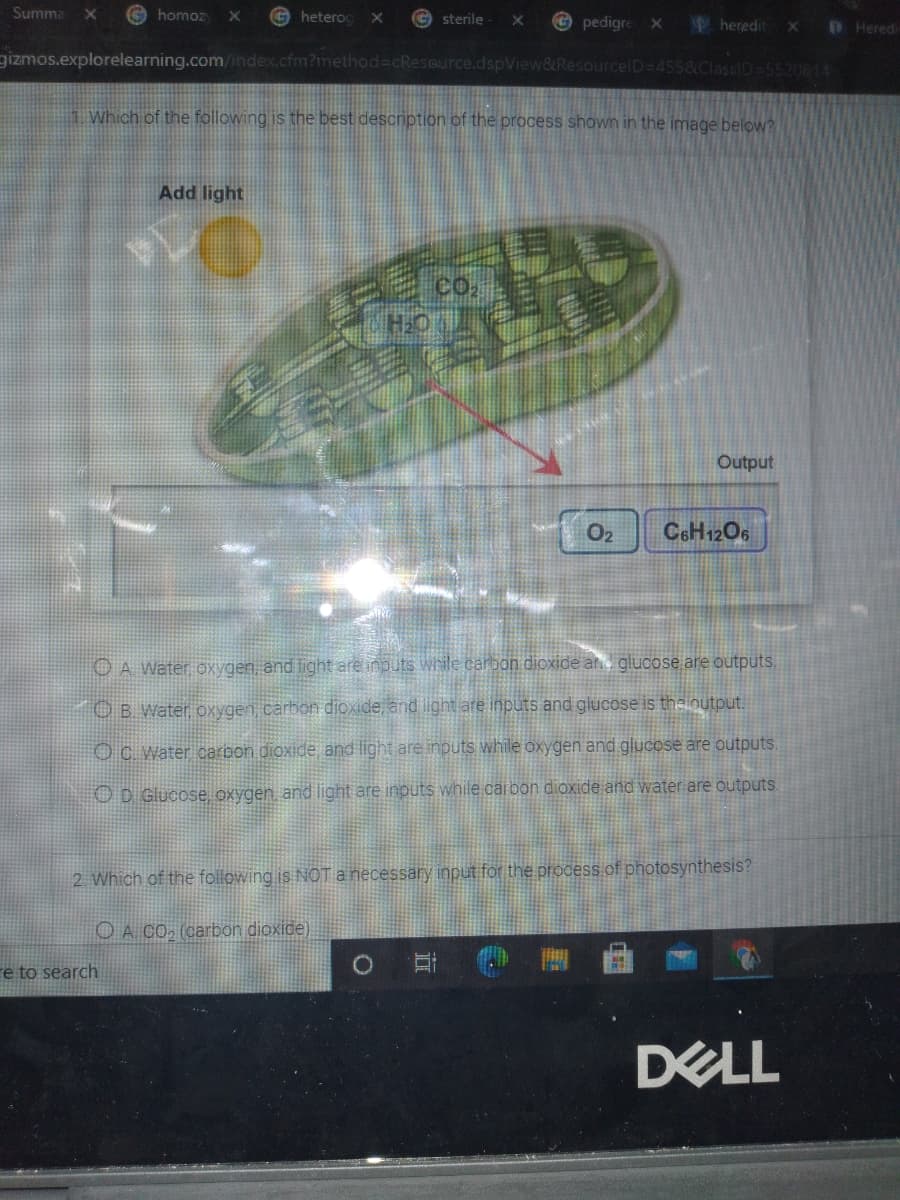
Which of the following is the best description of the process shown in the image below?
Transcribed Image Text:Summa
homoz
G heteroc X
G sterile
pedigre x
heredit
D Heredi
gizmos.explorelearning.com/index.cfm?method3cReseurce.dspView&ResourcelD=4558ClassID-5520614
1 Which of the following is the best description of the process shown in the image below?
Add light
co,
Output
O2
C6H12O6
O A Water oxygen and light are inputs while carbon dioxide and glucose are outputs,
O B. Water oxygen carbon dioxide, and light are inputs and glucose is theloutput.
OC Water carbon dioxide and light are inputs while oxygen and glucose are outputs.
OD Glucose oxygen and light are inputs while carbon dioxide and water are outputs.
2 Which of the following is NOT a necessary input for the process of photosynthesis?
O A CO, (carbon dioxide)
re to search
DELL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning


Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning